Abstract
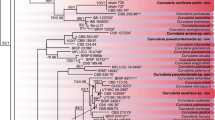
Phytophthora capsici and P. tropicalis are the two species of Phytophthora associated with foot rot disease of black pepper in India. High genetic diversity amongst the Phytophthora species contributes to its wide host range and variability in the virulence pattern. In the present study, genetic diversity of Phytophthora species infecting black pepper was analysed using RAMS (Random Amplified Microsatellites) and REP (Repetitive Extragenic Palindromic)-PCR fingerprinting. Forty-eight isolates, 24 each of P. capsici and P. tropicalis collected from major black pepper growing states, such as Karnataka, Kerala, Tamil Nadu and Goa, were used in the study. The analyses revealed a total of 160 loci of which 150 (93.75%) were polymorphic. UPGMA cluster and PCoA analysis based on combined RAMS and REP-PCR data clearly grouped the P. capsici and P. tropicalis isolates into two clusters which were further divided into four sub-clusters viz., I & II (P. capsici) and III & IV (P. tropicalis). The study clearly indicated that all the isolates were genetically unique and the entire population was heterogeneous. REP-PCR primers showed more polymorphic loci than RAMS primers. Further, sixteen isolates were selected for morphological and infectivity analyses under in vitro conditions. The isolates exhibited varied colony morphology, sporangial shapes and belonged to A1 mating type. Under in vitro conditions, all the sixteen black pepper Phytophthora isolates could infect nutmeg, tomato, chilli, pumpkin, and cucumber and few of the isolates could infect cardamom. None of the isolates could infect coconut, areca nut and vanilla.








Similar content being viewed by others
Data availability
All data generated in this study are available within the paper and also as supplementary information.
References
Aragaki M, Uchida J (2001) Morphological distinctions between Phytophthora capsici and P. tropicalis sp. nov. Mycologia 93:137. https://doi.org/10.1080/00275514.2001.12061285
Bhai RS, Jeevalatha A, Biju CN, Vinitha KB, Cissin J, Rosana OB, Fayad A, Praveena R, Anandaraj M, Eapen SJ (2022) Sympatric occurrence of sibling Phytophthora species associated with foot rot disease of black pepper in India. Braz J Microbiol 53(2):801–818. https://doi.org/10.1007/s42770-022-00716-2
Bilung LM, Pui CF, Su’ut L, Apun K (2018) Evaluation of BOX-PCR and ERIC-PCR as molecular typing tools for pathogenic Leptospira. Dis Markers 2018(6):1–9. https://doi.org/10.1155/2018/1351634
Borba MP, Ballarini AE, Paulo J, Witusk D, Lavin P, Sand SVD (2020) Evaluation of BOX-PCR and REP-PCR as molecular typing tools for Antarctic Streptomyces. Curr Microbiol 77:3573–3581. https://doi.org/10.1007/s00284-020-02199-6
Bouws H, Finckh MR (2007) Effects of cropping history and origin of seed potatoes on population structure of Phytophthora infestans. Eur J Plant Pathol 117:313–327. https://doi.org/10.1007/s10658-006-9099-z
Brasier CM (1967) Physiology of reproduction in Phytophthora. Hull, UK: University of Hull, PhD thesis
Brumat ACL, MayDe Mio LL, Tessmann DJ, Duarte HSS, Auer CG, Dos Santos ÁF (2022) Phytophthora tropicalis: causal agent of persimmon fruit rot in Brazil. J Phytopathol 170:428–436. https://doi.org/10.1111/jph.13094
Burdon JJ (1993) Genetic variation in pathogen populations and its implications for adaptation to host resistance. In: Jacobs T, Parlevliet JE (eds) Durability of disease resistance. Curr Plant Sci Biotechnol Agric, vol 18. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-2004-3_4
Chowdappa P, Chandramohanan R (1997) Occurrence and distribution of mating types of Phytophthora species causing black pod disease of cocoa. Indian Phytopath 50(2):256–260
da Silva MR, Martinelli JA, Federizzi LC, Chaves MS, Pacheco MT (2012) Lesion size as a criterion for screening oat genotypes for resistance to leaf spot. Eur J Plant Pathol 134:315–327. https://doi.org/10.1007/s10658-012-9991-7
de Bruijn FJ (1992) Use of repetitive (repetitive extragenic palindromic and enterobacterial repetitive intergeneric consensus) sequences and the polymerase chain reaction to fingerprint the genomes of Rhizobium meliloti isolates and other soil bacteria. Appl Environ Microbiol 58(7):2180–2187. https://doi.org/10.1128/aem.58.7.2180-2187.1992
Donahoo RS, Lamour KH (2008) Interspecific hybridization and apomixis between Phytophthora capsici and Phytophthora tropicalis. Mycologia 100(6):911–920. https://doi.org/10.3852/08-028
El-Esawi MA, El-Zaher Mustafa A, Badr S, Sammour R (2017) Isozyme analysis of genetic variability and population structure of Lactuca L. germplasm. Biochem Syst Ecol 70:73–79. https://doi.org/10.1016/j.bse.2016.11.003
Erwin DC, Ribeiro OK (1996) Phytophthora diseases worldwide. American Phytopathological Society Press, St Paul
Farhana MDSN, Bivi MR, Khairulmazmi A, Wong SK, Sariah M (2013) Morphological and molecular characterization of Phytophthora capsici, the causal agent of foot rot disease of black pepper in Sarawak, Malaysia. Int J Agric Biol 15:1083–1090
Food and Agriculture Organization of the United Nations (2023) https://www.fao.org/home/en. Accessed 19 April 2023
Fu C, Qiu Y, Kong H (2003) RAPD analysis for genetic diversity in Changium smyrnioides (Apiaceae), an endangered plant1. Bot Bull Acad Sin (taipei) 44:13–18
Goodwin SB (1997) The population genetics of Phytophthora. Phytopathology 87:462–473
Hantula J, Dusabenyagasani M, Hamelin RC (1996) Random amplified microsatellites (RAMS)—a novel method for characterizing genetic variation within fungi. Eur J for Pathol 26:159–166
Hema M, Kumar R, Singh NP (2007) Volatile price and declining profitability of black pepper in India: disquieting future. Agric Econ Res Rev 20:61–76
Hong CX, Richardson PA, Kong P, Jeffers SN, Oak SW (2006) Phytophthora tropicalis isolated from diseased leaves of Pieris japonica and Rhododendron catawbiense and found in irrigation water and soil in Virginia. Plant Dis 90(4):525. https://doi.org/10.1094/PD-90-0525C
Hong C, Patricia A, Richardson KP (2008) Pathogenicity to ornamental plants of some existing species and new taxa of Phytophthora from irrigation water. Plant Dis 92(8):1201–1207. https://doi.org/10.1094/PDIS-92-8-1201
Jara-Arancio P, Carvalho CS, Carmona-Ortiz MR, Bustamante RO, Schmidt Villela PM, Andrade SCS, Peña-Gómez FT, González LA, Fleury M (2022) Genetic diversity and population structure of Jubaea chilensis, an endemic and monotype gender from Chile, based on SNP markers. Plants 11(15):1959. https://doi.org/10.3390/plants11151959
Jeevalatha A, Biju CN, Bhai RS (2021) Ypt1 gene-based recombinase polymerase amplification assay for Phytophthora capsici and P. tropicalis detection in black pepper. Eur J Plant Pathol 159:863–875. https://doi.org/10.1007/s10658-021-02211-0
Jibat M, Alo S (2021) Characterization of Phytophthora capsici foot rot disease of black pepper in Ethiopia. J Plant Pathol Microbiol 12:542
Li P, Liu D (2021) Genetic diversity amongst asexual and sexual progenies of Phytophthora capsici detected with ISSR markers. Plant Protect Sci 57:271–278
Li P, Liu D, Guo M, Pan Y, Chen F, Zhang H, Gao Z (2017) A PCR-based assay for distinguishing between A1 and A2 mating types of Phytophthora capsici. J Am Soc Hortic Sci 142(4):260–264. https://doi.org/10.21273/JASHS04013-17
Majid MU, Awan MF, Fatima K, Tahir MS, Ali Q, Rashid B, Rao AQ, Nasir I, Husnain T (2016) Phytophthora capsici on chilli pepper (Capsicum annuum L.) and its management through genetic and bio-control: a review. ZEMDIRBYSTE 103:419–430
Mansfeld BN, Colle M, Zhang C, Lin YC, Grumet R, Grear JS, Kuhn A (2020) Developmentally regulated activation of defense allows for rapid inhibition of infection in age-related resistance to Phytophthora capsici in cucumber fruit. BMC Genomics 21:628. https://doi.org/10.1186/s12864-020-07040-9
Markert JA, Champlin DM, Gutjahr-Gobell R, Grear JS, Kuhn A, McGreevy TJ, Roth A, Bagly MJ, Nacci DE (2010) Population genetic diversity and fitness in multiple environments. BMC Evol Biol 10:205. https://doi.org/10.1186/1471-2148-10-205
Masanto HA, Wibowo A, Subandiyah S, Shimizu M, Suga H, Kageyama K (2019) Genetic diversity of Phytophthora palmivora isolates from Indonesia and Japan using rep-PCR and microsatellite markers. J Gen Plant Pathol 85:367–381. https://doi.org/10.1007/s10327-019-00853-x
Meitz J, Linde C, Thompson LSD, Mcleod A (2010) Phytophthora capsici on vegetable hosts in South Africa: distribution, host range and genetic diversity. Australas Plant Pathol 39:431–439. https://doi.org/10.1071/AP09075
Mishra RK, Pandey BK, Pathak N, Zeeshan M (2015) BOX-PCR- and ERIC-PCR-based genotyping and phylogenetic correlation among Fusarium oxysporum isolates associated with wilt disease in Psidium guajava L. Biocatal Agric Biotechnol 4(1):25–32. https://doi.org/10.1016/j.bcab.2014.09.012
Mohammad B, Jose RS (2018) Chemical management of Phytophthora capsicin pumpkin in Illinois. Mod concepts dev agron 1(4)
Nath VS, Basheer S, Jeeva ML, Veena SS (2016) Genetic and phenotypic characterization of Phytophthora colocasiae in Taro Growing Areas of India. J Plant Pathol Microbiol 7:383. https://doi.org/10.4172/2157-7471.1000383
Orlikowski LB, Trzewik A, Nowak K, Szkuta G (2006) Phytophthora tropicalis, a new pathogen of ornamental plants in Poland. J Plant Prot Res 46
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 28:2537–2539
Pereira D, McDonald BA, Croll D (2020) The genetic architecture of emerging fungicide resistance in populations of a global wheat pathogen. Genome Biol Evol 12(12):2231–2244. https://doi.org/10.1093/gbe/evaa203
Rai GS, Liew ECY, Guest DI (2020) Survey, identification and genetic diversity of Phytophthora capsici causing wilt of chilli (Capsicum annuum L.) in Bhutan. Eur J Plant Pathol 158:655–665. https://doi.org/10.1007/s10658-020-02108-4
Rohlf FJ (2000) NTSYS-PC. Numerical Taxonomy and Multivariate Analysis System. Version 2.10e. Department of Ecology and Evolution, State University of New York at Stony Brook, Stony Brook
Roy SG, Bhattacharyya S, Mukherjee SK, Khatua DC (2009) Molecular identification of Phytophthora spp. affecting some economically important crops in Eastern India through ITS-RFLP and sequencing of the ITS region. J Phytopathol 157:666–674. https://doi.org/10.1111/j.1439-0434.2008.01537.x
Shej C, Renu SG, Balaji S, Anandaraj M (2009) Ribosomal DNA analysis of three Phytophthora species occurring in India. Indian Phytopathol 62: 155–162. https://epubs.icar.org.in/index.php/IPPJ/article/view/12540
Shirzad-Aski H, Tabatabaei M (2016) Molecular characterization of Pasteurella multocida isolates obtained from poultry, ruminant, cats and dogs using RAPD and REP-PCR analysis. Mol Biol Res Commun 5(3):123–132
Spices Board (2023) http://indianspices.com/. Accessed 19 April 2023
Stephenson LW, Erwin DC, Leary JV (1974) Hyphal anastomosis in Phytophthora capsici. Phytopathology 64:149–150
Syed-Ab-Rahman SF, Xiao Y, Carvalhais LC, Ferguson BJ, Schenk PM (2019) Suppression of Phytophthora capsici infection and promotion of tomato growth by soil bacteria. Rhizosphere 9:72–75. https://doi.org/10.1016/j.rhisph.2018.11.007
Truong NV, Burgess L, Liew ECY (2008) Prevalence and aetiology of Phytophthora foot rot of black pepper in Vietnam. Australas Plant Pathol 37:431–442. https://doi.org/10.1071/AP08034
Truong NV, Liew ECY, Burgess LW (2010) Characterisation of Phytophthora capsici isolates from black pepper in Vietnam. Fungal Biol 114(2–3):160–170. https://doi.org/10.1016/j.funbio.2009.11.005
Truong NV, Burgess LW, Liew ECY (2012) Cross-infectivity and genetic variation of Phytophthora capsici isolates from chilli and black pepper in Vietnam. Australas Plant Pathol 41:439–447. https://doi.org/10.1007/s13313-012-0136-4
Versalovic J, Koeuth T, Lupski JR (1991) Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Res 19:6823–6831
Versalovic J, Kapur V, Mason EO, Shah U, Koeuth T, Lupski JR, Musser JM (1993) Penicillin-resistant Streptococcus pneumoniae strains recovered in Houston: identification and molecular characterization of multiple clones. J Infect Dis 167:850–856
Wang M, Zhang X, Yang T, Han Z, Yanagimoto T, Gao T (2009) Genetic diversity in the mtDNA control region and population structure in the Sardinella zunasi Bleeker. Afr J Biotechnol 7:4384–4392. https://doi.org/10.5897/AJB2022.17528
Welsh J, Honeycutt RJ, McClelland M, Sobral BW (1991) Parentage determination in maize hybrids using the arbitrarily primed polymerase chain reaction (AP-PCR). Theor Appl Genet 82(4):473–476. https://doi.org/10.1007/BF00588601
Yeh F, Boyle T, Yang R, Ye Z, Xiyan JM (1997) POPGENE: Microsoft Window-based International Forestry Research, Edmonton, Canada
Yu-Huan Gu, Ko WH (2000) Occurrence of parasexual cycle in Phytophthora parasitica following protoplast fusion. Bot Bull Acad Sin (taipei) 41:225–230
Zaccardelli M, Campanile F, Moretti C, Buonaurio R (2008) Characterization of Italian populations of Xanthomonas campestris pv. campestris using primers based on DNA repetitive sequences. J Plant Pathol 90:375–381
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20(2):176–183. https://doi.org/10.1006/geno.1994.1151
Acknowledgements
The authors would like to acknowledge Director (ICAR-IISR), Head—Division of Crop Protection (ICAR-IISR) for the facilities and support, Mr. K Jayarajan, Chief Technical Officer (ICAR-IISR) for the statistical analysis. University Grants Commission (UGC), Government of India for the fellowship.
Author information
Authors and Affiliations
Contributions
FZ—Implementation of the research, analyses and interpretation of results, draft manuscript preparation, AJ—Conceptualization, experimental design, analysis and interpretation of results, manuscript editing, CNB—Analyses and interpretation of results, manuscript editing. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the author.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zumaila, F., Jeevalatha, A. & Biju, C.N. Genetic diversity, mating type and pathogenicity of two Phytophthora species infecting black pepper in India. 3 Biotech 14, 1 (2024). https://doi.org/10.1007/s13205-023-03843-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03843-1