Abstract
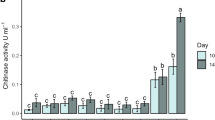
Coffee pulp (CP) is a significant agro-industrial waste generated during coffee bean processing, which possess substantial environmental contamination and is rich in pectin. The primary objective of this study was to investigate the conversion of pectin extracted from coffee pulp into pectic oligosaccharides (POS) using native microbial strains. The study aimed to optimize the growing conditions, including temperature, time, and pectin concentration, to assess the productivity of pectinase. Two fungal strains that exhibited the highest growth on CP were isolated and subsequently identified as Aspergillus fumigatus P-1007 and A. fumigatus HA1, employing 5.8S rRNA gene sequencing. The optimization of temperature for the organism was carried out between 25 and 45 °C; compared to the other temperatures at 45 °C the productivity of pectinase was high; the exact temperature was used for the time experiment where we found that compared to the A. fumigatus P-1007, A. fumigates HA1 was showed high enzyme productivity on 6th day. Hence, the highest productivity of endo-pectinase was seen at a temperature of 45 °C on the 6th day using isolated A. fumigates HA1 in the CP with 1% of coffee pectin. Additionally, the produced POS were screened and confirmed through TLC and HPLC analysis. The antioxidant activity of the POS derived from the separated CP demonstrated an effective concentration (EC50) of 400 µg/ml. The study indicates that the efficient utilization of CP waste for producing potentially valuable functional food ingredients, such as POS, holds promise for commercial development.





Similar content being viewed by others
Data availability
All the research data generated and analysed will be made available on reasonable request basis.
References
Acuña-Argüelles ME, Gutierrez-Rojas M, Viniegra-González G, Favela-Torres E (1995) Production and properties of three pectinolytic activities produced by Aspergillus niger in submerged and solid-state fermentation. Appl Microbiol Biotechnol 43:808–814
Adebola O, Corcoran O, Morgan WA (2013) Protective effects of prebiotics inulin and lactulose from cytotoxicity and genotoxicity in human colon adenocarcinoma cells. Food Res Int 52:269–274
Aguilar G, Huitrón C (1990) Constitutive exo-pectinase produced by Aspergillus sp. CH-Y-1043 on different carbon source. Biotechnol Let 12(9):655–660
Aneja KR, Sharma C, Joshi R (2012) Antimicrobial activity of Terminalia arjuna Wight & Arn.: an ethnomedicinal plant against pathogens causing ear infection. Braz J Otorhinolaryngol 78:68–74
Babbar N, Roy SV, Wijnants M, Dejonghe W, Caligiani A, Sforza S, Elst K (2016) Effect of extraction conditions on the saccharide (neutral and acidic) composition of the crude pectic extract from various agro-industrial residues. J Agric Food Chem 64(1):268–276
Bhattacharyya U, Paul P, Mustafi S, Sen C, Gupta M, Roychoudhury B (2019) Isolation of pectinolytic fungi from rotten tomato, brinjal and banana. In: Biotechnology and biological sciences: Proceedings of the 3rd international conference of biotechnology and biological sciences (Biospectrum 2019), August 8–10, 2019. CRC Press, Kolkata, India, p 374
Blumenthal CZ (2004) Production of toxic metabolites in Aspergillus niger, Aspergillus oryzae, and Trichoderma reesei: justification of mycotoxin testing in food grade enzyme preparations derived from the three fungi. Regul Toxicol Pharmacol 39(2):214–228
Brand-Williams W, Cuvelier ME, Berset CLWT (1995) Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci Technol 28:25–30
Bressani R, Elías LG (1979) Nutritional role of polyphenols in beans. Polyphenols in cereals and legumes: proceedings of a symposium. IDRC, Ottawa
Brody H, Griffith J, Cuticchia AJ, Arnold J, Timberlake WE (1991) Chromosome-specific recombinant DNA libraries from the fungus Aspergillus nidulans. Nucleic Acids Res 19:3105–3109
Chen H, Yan X, Zhu P, Lin J (2006) Antioxidant activity and hepatoprotective potential of agaro-oligosaccharides in vitro and in vivo. Nutr J 5(1):31
Combo AMM, Aguedo M, Goffin D, Wathelet B, Paquot M (2012) Enzymatic production of pectic oligosaccharides from polygalacturonic acid with commercial pectinase preparations. Food Bioprod Process 90:588–596
Cortesao M, Holland G, Laue M, Meyer V, Moeller R (2019) Aspergillus niger colony microstructure analysis with scanning electron microscopy (SEM). Vaam 19:21
El Enshasy HA, Elsayed EA, Suhaimi N, Malek RA, Esawy M (2018) Bioprocess optimization for pectinase production using Aspergillus niger in a submerged cultivation system. BMC Biotechnol 18:1–13
FAO/WHO (2001) Evaluation of health and nutritional properties of powder milk and live lactic acid bacteria. Food and agriculture organization of the united nations and world health organization expert consultation report. FAO, Rome
Garthoff JA, Heemskerk S, Hempenius RA, Lina BA, Krul CA, Koeman JH, Speijers GJ (2010) Safety evaluation of pectin-derived acidic oligosaccharides (pAOS): genotoxicity and sub-chronic studies. Regulatory Toxicol Pharmacol 57:31–42
Gómez B, Gullón B, Yáñez R, Parajó JC, Alonso JL (2013) Pectic oligosacharides from lemon peel wastes: production, purification, and chemical characterization. J Agric Food Chem 61(42):10043–10053
Gullón B, Gómez B, Martínez-Sabajanes M, Yáñez R, Parajó JC, Alonso JL (2013) Pectic oligosaccharides: manufacture and functional properties. Trends Food Sci Technol 30(2):153–161
Holck J, Hjernø K, Lorentzen A, Vigsnæs LK, Hemmingsen L, Licht TR, Meyer AS (2011) Tailored enzymatic production of oligosaccharides from sugar beet pectin and evidence of differential effects of a single DP chain length difference on human faecal microbiota composition after in vitro fermentation. Process Biochem 46(5):1039–1049
Hosseini-Abari A, Emtiazi G, Jazini M, Kim J, Kim BG (2019) LC/MS detection of oligogalacturonic acids obtained from tragacanth degradation by pectinase producing bacteria. J Basic Microbiol 59(3):249–255
Hussain A, Afzal A, Irfan M, Malik KA (2015) Molecular detection of aflatoxin producing strains of Aspergillus flavus from peanut (Arachis hypogaea). Turkish J Agric Food Sci Technol 3(5):335–341
Kasai A, Horie H, Sakamoto W (2004) Selection of food sources by Ruditapes philippinarum and Mactra veneriformis (Bivalva: Mollusca) determined from stable isotope analysis. Fish Sci 70:11–20
Khatri BP, Bhattarai T, Shrestha S, Maharjan J (2015) Alkaline thermostable pectinase enzyme from Aspergillus niger strain MCAS2 isolated from Manaslu Conservation Area, Gorkha, Nepal. Springerplus 4(1):1–8
Kleerebezem M, Vaughan EE (2009) Probiotic and gut lactobacilli and bifidobacteria: molecular approaches to study diversity and activity. Annu Rev Microbiol 63:269–290
Liu H, Wei X, Zu S, Lin X, Zhang J, Shi A, He N (2021) Separation and identification of neutral oligosaccharides with prebiotic activities from apple pectin. Food Hydrocoll 121:107062
Machenahalli S (2018) Microbes from coffee pulp waste and their pectinolytic enzyme activity for biodegradation. Int J Microbiol Res 10:1109–1110
Mahto RB, Yadav M, Muthuraj M, Sharma AK, Bhunia B (2023) Biochemical properties and application of a novel pectinase from a mutant strain of Bacillus subtilis. Biomass Conv Bioref 13:10463–10474
Manasa V, Padmanabhan A, Appaiah KA (2021) Utilization of coffee pulp waste for rapid recovery of pectin and polyphenols for sustainable material recycle. Waste Manag 120:762–771
Martínez Sabajanes M, Yáñez R, Alonso JL, Parajó JC (2012) Pectic oligosaccharides production from orange peel waste by enzymatic hydrolysis. Int J Food Sci Technol 47(4):747–754
Muthaiyan A, Hernandez-Hernandez O, Moreno FJ, Sanz ML, Ricke SC (2012) Hydrolyzed caseinomacropeptide conjugated galactooligosaccharides support the growth and enhance the bile tolerance in Lactobacillus strains. J Agric Food Chem 60(27):6839–6845
Prasad B, Goya S (2019) Effects of carbon and process parameters on the production of pectinase by Aspergillus spp. isolated from rotten citrus fruits. Int J Innov Biol Chem Sci 12:1–10
Pu J, Zhao X, Wang Q, Wang Y, Zhou H (2016) Development and validation of a HPLC method for determination of degree of polymerization of xylo-oligosaccharides. Food Chem 213:654–659
Rao KR, Vipin AV, Venkateswaran G (2020) Molecular profile of non-aflatoxigenic phenotype in native strains of Aspergillus flavus. Archiv Microbiol 202:1143–1155
Rebello S, Anju M, Aneesh EM, Sindhu R, Binod P, Pandey A (2017) Recent advancements in the production and application of microbial pectinases: an overview. Rev Env Sci Biotechnol 16:381–394
Rivero-Urgell M, Santamaria-Orleans A (2001) Oligosaccharides: application in infant food. Early Hum Develop 65:43–52
Sabater C, Blanco-Doval A, Montilla A, Corzo N (2021) Optimisation of an enzymatic method to obtain modified artichoke pectin and pectic oligosaccharides using artificial neural network tools In silico and in vitro assessment of the antioxidant activity. Food Hydrocoll 110:106161
Silva DM, Batista LR, Rezende EF, Fungaro MHP, Sartori D, Alves E (2011) Identification of fungi of the genus Aspergillus section Nigri using polyphasic taxonomy. Braz J Microbiol 42:761–773
Soares MMCN, Da Silva R, Carmona EC, Gomes E (2001) Pectinolytic enzyme production by Bacillus species and their potential application on juice extraction. World J Microbiol Biotechnol 17:79–82
Torimiro N, Adediwura VA, Ojo ST, Oluwadare AO, Okonji RE (2018) Pectinolytic activities of pectinase produced by some bacterial isolates cultured from deteriorating fruits. Nigerian J Biotechnol 35(2):91–98
Turzhanov AS, Rukavitsyna IV, Khapilna ON, Kalendar RN (2018) Optimization of DNA extraction from filamentous fungi Alternaria sp. and Fusarium sp. Eurasian J Appl Biotechnol 3:35–41
Ubaidillah S, Siswanto Muzakhar K (2017) Decomposition of coffee pulp polysaccharides by Aspergillus niger extracelluler enzyme. Int Conf Life Sci Biotechnol 7:148–153
Wang J, Hu S, Nie S, Yu Q, Xie M (2016) Reviews on mechanisms of in vitro antioxidant activity of polysaccharides. Oxid Med Cell Long 64:1–13
Wu DT, He Y, Yuan Q, Wang S, Gan RY, Hu YC, Zou L (2022) Effects of molecular weight and degree of branching on microbial fermentation characteristics of okra pectic-polysaccharide and its selective impact on gut microbial composition. Food Hydrocoll 132:107897
Xue L, Long J, Lu C, Li X, Xu X, Jin Z (2021) Immobilization of polygalacturonase for the preparation of pectic oligosaccharides from mango peel wastes and assessment of their antibacterial activities. Food Biosci 39:100837
Yeung YK, Kang YR, So BR, Jung SK, Chang YH (2021) Structural, antioxidant, prebiotic and anti-inflammatory properties of pectic oligosaccharides hydrolyzed from okra pectin by Fenton reaction. Food Hydrocoll 118:106779
Zhang S, Hu H, Wang L, Liu F, Pan S (2018) Preparation and prebiotic potential of pectin oligosaccharides obtained from citrus peel pectin. Food Chem 244:232–237
Acknowledgements
The authors are thankful to Department of Biotechnology (BT/PR/7570/BCE/8/1022), New Delhi and Director, CSIR-CFTRI, Mysore for encouragement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Manasa, V., Appaiah, K.A.A. Indigenous fungal strains isolation and molecular identification from coffee pulp for the production of pectic oligosaccharides. 3 Biotech 13, 410 (2023). https://doi.org/10.1007/s13205-023-03811-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03811-9