Abstract
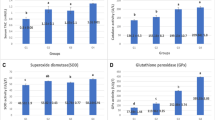
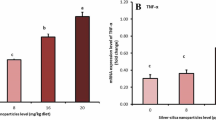
This study aimed to evaluate the effectiveness of the embryonic injection of silver nanoparticles (SilNPs) on some productive traits and hepatic gene expression of lipopolysaccharide (LPS)-challenged broilers after a 42 d rearing period. 560 fertile eggs were randomly allocated to four groups and received either of the following treatments at d 7 of incubation, control (no injection), placebo (1 mL saline), SilNP20 (20 mg/kg silver nanoparticles), or SilNP40 (40 mg/kg silver nanoparticles). After the incubation, 320 hatchlings experienced a 42 d standard rearing period. Live body weight (LBW), feed intake (FI), and feed conversion ratio (FCR) were weekly recorded. At the end of the experiment, two birds from each replicate (n = 8 per treatment) were exposed to LPS intraperitoneal injection at 48, 24, and 12 h before slaughter time. They were also used for blood, intestinal, and microbial evaluations. The hepatic mRNA levels of tumor necrosis factor-alpha (TNF-α), interleukin 6 (IL-6), transforming growth factor beta (TGF-β), and insulin-like growth factor I (IGF-I) were assessed at d 1 and 42 of the experiment. Adminstration of SiLNPs improved LBW, FI, and FCR and also enhanced liver and spleen weights (P < 0.05). SilNP20 birds had significantly lower bursa of Fabricius weight (P < 0.05). SilNP20 had lower total cholesterol levels than others. There was a significant difference (P < 0.05) between SliNP40 and SilNP20 in the ratio of villus height to crypt width. Compared to control groups, chicks of SilNP20, but not SilNP40, showed a significant increase in the relative expression of TNF-α, IL-6, TGF-β, and IFG-I genes at d 1. On d 42, however, both SilNP20 and SilNP40 had significantly higher TNF-α and TGF-β levels than both controls. Silver nanoparticles did not significantly affect the microflora of the ileum and cecum in the current study. In summary, SilNPs administration to chick embryos showed a long-term positive effect on their productive performance.

Similar content being viewed by others
Availability of data and material
All data have been included in the manuscript.
References
Abd El-Ghany WA (2019) Nanotechnology and its considerations in poultry field: an overview. J Hell Vet Med Soc 70:1611–1616. https://doi.org/10.12681/jhvms.21783
Abd El-Hack ME, El-Saadony MT, Salem HM, El-Tahan AM, Soliman MM, Youssef GB, Taha AE, Soliman SM, Ahmed AE, El-Kott AF, Al Syaad KM (2022) Alternatives to antibiotics for organic poultry production: types, modes of action and impacts on bird’s health and production. Poult Sci 101:101696. https://doi.org/10.1016/j.psj.2022.101696
Abdelsalam M, Al-Homidan I, Ebeid T, Abou-Emera O, Mostafa M, Abd El-Razik M, Shehab-El-Deen M, Abdel Ghani S, Fathi M (2019) Effect of silver nanoparticle administration on productive performance, blood parameters, antioxidative status, and silver residues in growing rabbits under hot climate. Animals. https://doi.org/10.3390/ani9100845
Abou-Kassem DE, El-Abasy MM, Al-Harbi MS, Abol-Ela S, Salem HM, El-Tahan AM, El-Saadony MT, Abd El-Hack ME, Ashour EA (2022) Influences of total sulfur amino acids and photoperiod on growth, carcass traits, blood parameters, meat quality and cecal microbial load of broilers. Saudi J Biol Sci 29:1683–1693
Almamury A, Hassanabadi A, Zerehdaran S, Nassiri-Moghaddam H (2021) Effects of dietary supplementation of a herbal product (NBS Superfood) on growth performance, intestinal morphology, immune status and blood metabolites in broiler chickens. Poult Sci J 9:245–254. https://doi.org/10.22069/psj.2021.19078.1691
Andi MA, Hashmei M, Ahmadi F (2011) Effects of feed type with/without nanosil on cumulative performance, relative organ weight and some blood parameters of broilers. Glob Vet 7:605–609
Bhanja SK, Hotowy A, Mehra M, Sawosz E, Pineda L, Vadalasetty KP, Kurantowicz N, Chwalibog A (2015) In ovo administration of silver nanoparticles and/or amino acids influence metabolism and immune gene expression in chicken embryos. Int J Mol Sci 16:9484–9503. https://doi.org/10.3390/ijms16059484
Chen X, Schluesener HJ (2008) Nanosilver: A nanoproduct in medical application. Toxicol Lett 176:1–12. https://doi.org/10.1016/j.toxlet.2007.10.004
Chwalibog A, Sawosz E, Hotowy A, Szeliga J, Mitura S, Mitura K, Grodzik M, Orlowski P, Sokolowska A (2010) Visualization of interaction between inorganic nanoparticles and bacteria or fungi. Int J Nanomedicine 5:1085–1094. https://doi.org/10.2147/IJN.S13532
Dosoky WM, Fouda MM, Alwan AB, Abdelsalam NR, Taha AE, Ghareeb RY, El-Aassar MR, Khafaga AF (2021) Dietary supplementation of silver-silica nanoparticles promotes histological, immunological, ultrastructural, and performance parameters of broiler chickens. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-021-83753-5
Edens FW (2003) An alternative for antibiotic se in poultry: probiotics. Brazilian J Poult Sci 5:75–97
Elfaky MA, Sirwi A, Ismail SH, Awad HH, Gad SS (2022) Hepatoprotective effect of silver nanoparticles at two different particle sizes: comparative study with and without Silymarin. Curr Issues Mol Biol 44:2923–2938. https://doi.org/10.3390/cimb44070202
Elkloub K, El-Moustafa ME, Ghazalah AA, Rehan A (2015) Effect of dietary nanosilver on broiler performance. Int J Poult Sci 14:177–182. https://doi.org/10.3923/ijps.2015.177.182
Faedmaleki F, Shirazi F, Salarian AA, Ahmadi Ashtiani H, Rastegar H (2014) Toxicity effect of silver nanoparticles on mice liver primary cell culture and HepG2 Cell line. Iran J Pharm Res 13:235–242
Farzinpour A, Karashi N (2013) The effects of nano-silver on egg quality traits in laying Japanese quail. Appl Nanosci 3:95–99
Fatholahi A, Khalaji S, Hosseini F, Abbasi M (2021) Nano-Bio zinc synthesized by Bacillus subtilis modulates broiler performance, intestinal morphology and expression of tight junction’s proteins. Livest Sci 251:104660
Gallocchio F, Belluco S, Ricci A (2015) Nanotechnology and Food: brief overview of the current Scenario. Ital Oral Surg 5:85–88. https://doi.org/10.1016/j.profoo.2015.09.022
Guernec A, Berri C, Chevalier B, Wacrenier-Ceré N, Le Bihan-Duval E, Duclos MJ (2003) Muscle development, insulin-like growth factor-I and myostatin mRNA levels in chickens selected for increased breast muscle yield. Growth Horm IGF Res 13:8–18. https://doi.org/10.1016/s1096-6374(02)00136-3
Hajati H, Rezaei M (2010) The application of prebiotics in poultry production. Int J Poult Sci 9:298–304
Huang XY, Ansari AR, Huang HB, Zhao X, Li NY, Sun ZJ, Peng KM, Zhong J, Liu HZ (2017) Lipopolysaccharide mediates immuno-pathological alterations in young chicken liver through TLR4 signaling. BMC Immunol 18:1–9. https://doi.org/10.1186/s12865-017-0199-7
Kulak E, Ognik K, Stpniowska A, Sembratowicz I (2018) The effect of administration of silver nanoparticles on silver accumulation in tissues and immune and antioxidant status of chickens. J Anim Feed Sci 27:44–54. https://doi.org/10.22358/jafs/84978/2018
Lara HH, Ayala-Núnez NV, Ixtepan Turrent LD, Rodríguez Padilla C (2010) Bactericidal effect of silver nanoparticles against multidrug-resistant bacteria. World J Microbiol Biotechnol 26:615–621. https://doi.org/10.1007/s11274-009-0211-3
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Llamas-Moya S, Girdler CP, Shalash SMM (2020) Effect of a multicarbohydrase containing α-galactosidase enzyme on the performance, carcass yield, and humoral immunity of broilers fed corn–soybean meal–based diets of varying energy density. J Appl Poult Res 29:142–151
Loghman A, Iraj SH, Naghi DA, Pejman M (2012) Histopathologic and apoptotic effect of nanosilver in liver of broiler chickens. African J Biotechnol 11:6207–6211. https://doi.org/10.5897/ajb11.1768
Lu FZ, Wang XX, Pan QX, Huang RH, Liu HL (2008) Expression of genes involved in the somatotropic, thyrotropic, and corticotropic axes during development of Langshan and Arbor Acres chickens. Poult Sci 87:2087–2097. https://doi.org/10.3382/ps.2007-00493
Mahmoud UT (2012) Silver nanoparticles in poultry production. J Adv Vet Res 2:303–306
McAuliffe ME, Perry MJ (2007) Are nanoparticles potential male reproductive toxicants? A literature review. Nanotoxicology 1:204–210. https://doi.org/10.1080/17435390701675914
McIntyre RA (2012) Common nano-materials and their use in real world applications. Sci Prog 95:1–22. https://doi.org/10.3184/003685012X13294715456431
National Research Council (1994) Nutrient Requirements of Poultry: ninth Revised Edition. The National Academic Press, Washington, DC
Panyala NR, Peña-Méndez EM, Havel J (2008) Silver or silver nanoparticles: a hazardous threat to the environment and human health? J Appl Biomed 6:117–129. https://doi.org/10.32725/jab.2008.015
Pineda L, Chwalibog A, Sawosz E, Hotowy A, Elnif J, Sawosz F (2012) Investigating the effect of in ovo injection of silver nanoparticles on fat uptake and development in broiler and layer hatchlings. J Nanotechnol. https://doi.org/10.1155/2012/212486
Pradeepta KR, Subrat KB, Manish M, Akshat G, Aswathi PB (2018) Effect of in ovo supplemented silver nanoparticles on the gut morphology and hemato-serological parameters in broiler chickens. Indian J Poul Sci 53:188–191. https://doi.org/10.5958/0974-8180.2018.00036.3
Saki AA, Salary J (2015) The impact of in ovo injection of silver nanoparticles, thyme and savory extracts in broiler breeder eggs on growth performance, lymphoid-organ weights, and blood and immune parameters of broiler chicks. Poult Sci J 3:165–172
Salama B, Alzahrani KJ, Alghamdi KS, Al-Amer O, Hassan KE, Elhefny MA, Albarakati AJ, Alharthi F, Althagafi HA, Al Sberi H, Amin HK (2022) Silver nanoparticles enhance oxidative stress, inflammation, and apoptosis in liver and kidney tissues: potential protective role of thymoquinone. Biol Trace Elem Res. https://doi.org/10.1007/s12011-022-03399-w
Saleh AA, El-Magd MA (2018) Beneficial effects of dietary silver nanoparticles and silver nitrate on broiler nutrition. Environ Sci Pollut Res 25:27031–27038. https://doi.org/10.1007/s11356-018-2730-7
Salehimanesh A, Mohammadi M, Roostaei-Ali Mehr M (2016) Effect of dietary probiotic, prebiotic and synbiotic supplementation on performance, immune responses, intestinal morphology and bacterial populations in broilers. J Anim Physiol Anim Nutr (berl) 100:694–700. https://doi.org/10.1111/jpn.12431
Sawosz E, Binek M, Grodzik M, Zielińska M, Sysa P, Szmidt M, Niemiec T, Chwalibog A (2007) Influence of hydrocolloidal silver nanoparticles on gastrointestinal microflora and morphology of enterocytes of quails. Arch Anim Nutr 61:444–451. https://doi.org/10.1080/17450390701664314
Sawosz E, Grodzik M, Zielińska M, Niemiec T, Olszańska B, Chwalibog A (2009) Nanoparticles of silver do not affect growth, development and DNA oxidative damage in chicken embryos. Arch Fur Geflugelkd 73:208–213
Vadalasetty KP, Lauridsen C, Engberg RM, Vadalasetty R, Kutwin M, Chwalibog A, Sawosz E (2018) Influence of silver nanoparticles on growth and health of broiler chickens after infection with Campylobacter jejuni. BMC Vet Res 14:1–11. https://doi.org/10.1186/s12917-017-1323-x
Vinus NS, Tewatia BS (2017) Organic acids as alternatives to antibiotic growth promoters in poultry. Pharm Innov 6:164–169
Acknowledgements
We are grateful to Gorgan University of Agricultural Sciences and Natural Resources for facility support.
Author information
Authors and Affiliations
Contributions
Each named author has substantially contributed to conducting the underlying research and drafting this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahini, M., Arabameri, S., Ashayerizadeh, O. et al. In ovo injection of silver nanoparticles modulates some productive traits and hepatic gene expression of broilers exposed to lipopolysaccharide challenge. 3 Biotech 13, 197 (2023). https://doi.org/10.1007/s13205-023-03627-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03627-7