Abstract
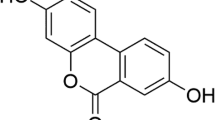
Umbelliferone (UB) is a phenylpropanoid-based pharmacologically active agent with promising anti-tumor activities. However, complete elucidation of its therapeutic efficacy remains challenging due to low solubility and bioavailability. The present study aimed to develop a liposomal delivery system for UB to enhance its therapeutic efficacy against Dalton’s ascites lymphoma tumor model. Umbelliferone-encapsulated nanoliposomes (nLUB) were prepared using the thin-film hydration method and performed a series of characterizations to confirm successful development. The nLUB showed a particle size of 116 ± 3.2 nm with a negative surface charge and encapsulation efficiency of 78%. In vitro study results showed that nLUB treatment significantly enhanced cellular uptake and apoptosis induction in lymphoma cells compared to free UB. nLUB treatment significantly stabilized body weight, reduced tumor growth, and improved the serum biochemical and hematological parameters of experimental animals, thereby improving their overall survivability compared to an free UB. Our result indicates that nanoencapsulation enhanced the therapeutic potential of UB, which may find its way to clinical application in the near future.






Similar content being viewed by others
Data availability
The data will be available on request.
References
Abeesh P, Vishnu WK, Guruvayoorappan C (2021) Preparation and characterization of withaferin A loaded pegylated nanoliposomal formulation with high loading efficacy: in vitro and in vivo anti-tumour study. Mater Sci Eng C 128:112335
Allen TM, Cullis PR (2013) Liposomal drug delivery systems: from concept to clinical applications. Adv Drug Deliv Rev 65(1):36–48
Cruz LF, de Figueiredo GF, Pedro LP, Amorin YM, Andrade JT, Passos TF, Rodrigues FF, Souza ILA, Gonçalves TPR, dos Santos Lima LAR (2020) Umbelliferone (7-hydroxycoumarin): a non-toxic antidiarrheal and antiulcerogenic coumarin. Biomed Pharmacother 129:110432
Damari SP, Shamrakov D, Varenik M, Koren E, Nativ-Roth E, Barenholz Y, Regev O (2018) Practical aspects in size and morphology characterization of drug-loaded nano-liposomes. Int J Pharm 547(1–2):648–655
Danaei M, Dehghankhold M, Ataei S, Hasanzadeh Davarani F, Javanmard R, Dokhani A, Khorasani S, Mozafari M (2018) Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 10(2):57
Dhamija I, Kumar N, Manjula S, Parihar V, Setty MM, Pai K (2013) Preliminary evaluation of in vitro cytotoxicity and in vivo antitumor activity of Premna herbacea Roxb. in Ehrlich ascites carcinoma model and Dalton’s lymphoma ascites model. Exp Toxicol Pathol 65(3):235–242
Dong H, Parekh HS, Xu ZP (2015) Particle size-and number-dependent delivery to cells by layered double hydroxide nanoparticles. J Colloid Interface Sci 437:10–16
Evans BC, Nelson CE, Shann SY, Beavers KR, Kim AJ, Li H, Nelson HM, Giorgio TD, Duvall CL (2013) Ex vivo red blood cell hemolysis assay for the evaluation of pH-responsive endosomolytic agents for cytosolic delivery of biomacromolecular drugs. JoVE 73:e50166
Guruvayoorappan C, Kuttan G (2007) Immunomodulatory and antitumor activity of Biophytum sensitivum extract. Asian Pac J Cancer Prev 8(1):27
He C, Hu Y, Yin L, Tang C, Yin C (2010) Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 31(13):3657–3666
Heurtault B, Saulnier P, Pech B, Proust J-E, Benoit J-P (2003) Physico-chemical stability of colloidal lipid particles. Biomaterials 24(23):4283–4300
Iglesias-Montes ML, Luzi F, Dominici F, Torre L, Manfredi LB, Cyras VP, Puglia D (2021) Migration and degradation in composting environment of active polylactic acid bilayer nanocomposites films: combined role of umbelliferone, lignin and cellulose nanostructures. Polymers 13(2):282
Immordino ML, Dosio F, Cattel L (2006) Stealth liposomes: review of the basic science, rationale, and clinical applications, existing and potential. Int J Nanomed 1(3):297
Kang TH, Mao C-P, He L, Tsai Y-C, Liu K, La V, Wu T-C, Hung C-F (2012) Tumor-targeted delivery of IL-2 by NKG2D leads to accumulation of antigen-specific CD8+ T cells in the tumor loci and enhanced anti-tumor effects. PLoS ONE 7(4):e35141
Kang C, Sun Y, Zhu J, Li W, Zhang A, Kuang T, Xie J, Yang ZJCDM (2016) Delivery of nanoparticles for treatment of brain tumor. Curr Drug Metab 17(8):745–754
Kumar V, Bhatt PC, Rahman M, Kaithwas G, Choudhry H, Al-Abbasi FA, Anwar F, Verma A (2017) Fabrication, optimization, and characterization of umbelliferone β-d-galactopyranoside-loaded PLGA nanoparticles in treatment of hepatocellular carcinoma: in vitro and in vivo studies. Int J Nanomed 12:6747
Kundu M, Chatterjee S, Ghosh N, Manna P, Das J, Sil PC (2020) Tumor targeted delivery of umbelliferone via a smart mesoporous silica nanoparticles controlled-release drug delivery system for increased anticancer efficiency. Mater Sci Eng C 116:111239
Lopez-Gonzalez JS, Prado-Garcia H, Aguilar-Cazares D, Molina-Guarneros JA, Morales-Fuentes J, Mandoki JJ (2004) Apoptosis and cell cycle disturbances induced by coumarin and 7-hydroxycoumarin on human lung carcinoma cell lines. Lung Cancer 43(3):275–283
Lu T, Haemmerich D, Liu H, Seynhaeve AL, van Rhoon GC, Houtsmuller AB, Ten Hagen TL (2021) Externally triggered smart drug delivery system encapsulating idarubicin shows superior kinetics and enhances tumoral drug uptake and response. Theranostics 11(12):5700
Muthu R, Thangavel P, Selvaraj N, Ramalingam R, Vaiyapuri M (2013) Synergistic and individual effects of umbelliferone with 5-flurouracil on the status of lipid peroxidation and antioxidant defense against 1, 2-dimethylhydrazine induced rat colon carcinogenesis. Biomed Prev Nutr 3(1):74–82
Priya R, Ilavenil S, Kaleeswaran B, Srigopalram S, Ravikumar S (2011) Effect of Lawsonia inermis on tumor expression induced by Dalton’s lymphoma ascites in Swiss albino mice. Saudi J Biol Sci 18(4):353–359
Ramesh B, Pugalendi K (2006) Antihyperglycemic effect of umbelliferone in streptozotocin-diabetic rats. J Med Food 9(4):562–566
Ramu R, S. Shirahatti P, Zameer F, Lakkappa Dhananjaya B, MN NP, (2016) Assessment of in vivo antidiabetic properties of umbelliferone and lupeol constituents of banana (Musa sp. Var. Nanjangud Rasa Bale) flower in hyperglycaemic rodent model. PLoS ONE 11(3):e0151135
Rauf A, Khan R, Khan H, Pervez S, Pirzada AS (2014) In vivo antinociceptive and anti-inflammatory activities of umbelliferone isolated from Potentilla evestita. Nat Prod Res 28(17):1371–1374
Sakthivel K, Kannan N, Angeline A, Guruvayoorappan C (2012) Anticancer activity of Acacia nilotica (L.) Wild. Ex. Delile subsp. indica against Dalton’s ascitic lymphoma induced solid and ascitic tumor model. Asian Pac J Cancer Prev 13(8):3989–3995
Sarabandi K, Jafari SM, Mohammadi M, Akbarbaglu Z, Pezeshki A, Heshmati MK (2019) Production of reconstitutable nanoliposomes loaded with flaxseed protein hydrolysates: stability and characterization. Food Hydrocolloids 96:442–450
Shi Y, Van der Meel R, Chen X, Lammers T (2020) The EPR effect and beyond: strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 10(17):7921
Singh R, Lillard JWJE Jr (2009) Nanoparticle-based targeted drug delivery. Exp Mol Pathol 86(3):215–223
Singh D, Rahman M (2020) Umbelliferone loaded nanocarriers for healthcare applications. Curr Biochem Eng 6(1):25–33
Suk JS, Xu Q, Kim N, Hanes J, Ensign LM (2016) PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev 99:28–51
Sun Y, Kang C, Wang M, Zhu J, Jin L, Cheng X (2016) Nanosized camptothecin conjugates for single and combined drug delivery. Eur J BioMed Res 2:8–16
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clinicians 71(3):209–249
Telange DR, Nirgulkar SB, Umekar MJ, Patil AT, Pethe AM, Bali NR (2019) Enhanced transdermal permeation and anti-inflammatory potential of phospholipids complex-loaded matrix film of umbelliferone: formulation development, physico-chemical and functional characterization. Eur J Pharm Sci 131:23–38
Ullah S, Shah MR, Shoaib M, Imran M, Elhissi AM, Ahmad F, Ali I, Shah SWA (2016) Development of a biocompatible creatinine-based niosomal delivery system for enhanced oral bioavailability of clarithromycin. Drug Deliv 23(9):3480–3491
Vasconcelos JF, Teixeira MM, Barbosa-Filho JM, Agra MF, Nunes XP, Giulietti AM, Ribeiro-dos-Santos R, Soares MB (2009) Effects of umbelliferone in a murine model of allergic airway inflammation. Eur J Pharmacol 609(1–3):126–131
Vijayalakshmi A, Sindhu G (2017) Umbelliferone arrest cell cycle at G0/G1 phase and induces apoptosis in human oral carcinoma (KB) cells possibly via oxidative DNA damage. Biomed Pharmacother 92:661–671
Vishnu WK, Abeesh P, Guruvayoorappan C (2021) Pyrazole (1, 2-diazole) induce apoptosis in lymphoma cells by targeting BCL-2 and BCL-XL genes and mitigate murine solid tumour development by regulating cyclin-D1 and Ki-67 expression. Toxicol Appl Pharmacol 418:115491
Wei Y, Liang J, Zheng X, Pi C, Liu H, Yang H, Zou Y, Ye Y, Zhao L (2017) Lung-targeting drug delivery system of baicalin-loaded nanoliposomes: development, biodistribution in rabbits, and pharmacodynamics in nude mice bearing orthotopic human lung cancer. Int J Nanomed 12:251
Wu D, Gao Y, Qi Y, Chen L, Ma Y, Li Y (2014) Peptide-based cancer therapy: opportunity and challenge. Cancer Lett 351(1):13–22
Yu SM, Hu DH, Zhang JJ (2015) Umbelliferone exhibits anticancer activity via the induction of apoptosis and cell cycle arrest in HepG2 hepatocellular carcinoma cells. Mol Med Rep 12(3):3869–3873
Zocchi MR, Tosetti F, Benelli R, Poggi AJC (2020) Cancer nanomedicine special issue review anticancer drug delivery with nanoparticles: extracellular vesicles or synthetic nanobeads as therapeutic tools for conventional treatment or immunotherapy. Cancers 12(7):1886
Zucker D, Andriyanov AV, Steiner A, Raviv U, Barenholz Y (2012) Characterization of PEGylated nanoliposomes co-remotely loaded with topotecan and vincristine: relating structure and pharmacokinetics to therapeutic efficacy. J Control Release 160(2):281–289
Acknowledgements
The authors acknowledge the Kerala Biotechnology Commission, Kerala State Council for Science Technology and Environment (KSCSTE) (no. 454/2020/KSCSTE)., and Department of Biotechnology (DBT) (DBT/JRF/BET-16/1/2016/Al/37-458) for providing financial assistance to the study. The authors thank Dr. S. Lakshmi, Head, Division of Cancer Research and Dr. Rekha A. Nair, Director, Regional Cancer Centre, for their valuable support.
Author information
Authors and Affiliations
Contributions
PA: investigation, methodology, analysis, writing—original draft, editing, visualization, funding acquisition. CG: conceptualization, review—editing, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethical approval
Animal experiments were approved by the Institutional Animal Ethics Committee, Regional Cancer Centre (Kerala, India).
Informed consent
Not applicable.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abeesh, P., Guruvayoorappan, C. Umbelliferone loaded PEGylated liposomes: preparation, characterization and its mitigatory effects on Dalton’s ascites lymphoma development. 3 Biotech 13, 216 (2023). https://doi.org/10.1007/s13205-023-03615-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03615-x