Abstract
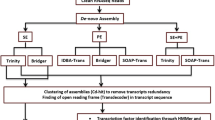
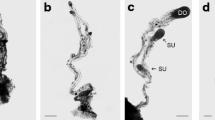
Chickpea (Cicer arietinum L.) seeds are valued for their nutritional scores and limited information on the molecular mechanisms of chickpea fertilization and seed development is available. In the current work, comparative transcriptome analysis was performed on two different stages of chickpea ovules (pre- and post-fertilization) to identify key regulatory transcripts. Two-staged transcriptome sequencing was generated and over 208 million reads were mapped to quantify transcript abundance during fertilization events. Mapping to the reference genome showed that the majority (92.88%) of high-quality Illumina reads were aligned to the chickpea genome. Reference-guided genome and transcriptome assembly yielded a total of 28,783 genes. Of these, 3399 genes were differentially expressed after the fertilization event. These involve upregulated genes including a protease-like secreted in CO(2) response (LOC101500970), amino acid permease 4-like (LOC101506539), and downregulated genes MYB-related protein 305-like (LOC101493897), receptor like protein 29 (LOC101491695). WGCNA analysis and pairwise comparison of datasets, successfully constructed four co-expression modules. Transcription factor families including bHLH, MYB, MYB-related, C2H2 zinc finger, ERF, WRKY and NAC transcription factor were also found to be activated after fertilization. Activation of these genes and transcription factors results in the accumulation of carbohydrates and proteins by enhancing their trafficking and biosynthesis. Total 17 differentially expressed genes, were randomly selected for qRT-PCR for validation of transcriptome analysis and showed statistically significant correlations with the transcriptome data. Our findings provide insights into the regulatory mechanisms underlying changes in fertilized chickpea ovules. This work may come closer to a comprehensive understanding of the mechanisms that initiate developmental events in chickpea seeds after fertilization.








Similar content being viewed by others
Data availability
The transcriptome data deposited into the Gene Expression Omnibus database under accession number GSE217186 and are available at the following URL: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE217186.
References
Adhikari PB, Liu X, Wu X et al (2020) Fertilization in flowering plants: an odyssey of sperm cell delivery. Plant Mol Biol 103:9–32. https://doi.org/10.1007/s11103-020-00987-z
Agarwal P, Kapoor S, Tyagi AK (2011) Transcription factors regulating the progression of monocot and dicot seed development. BioEssays News Rev Mol Cell Dev Biol 33:189–202. https://doi.org/10.1002/bies.201000107
Agarwal G, Jhanwar S, Priya P et al (2012) Comparative analysis of kabuli chickpea transcriptome with desi and wild chickpea provides a rich resource for development of functional markers. PLoS ONE 7:e52443. https://doi.org/10.1371/journal.pone.0052443
Alabadí D, Blázquez MA, Carbonell J et al (2009) Instructive roles for hormones in plant development. Int J Dev Biol 53:1597–1608. https://doi.org/10.1387/ijdb.072423da
Ambawat S, Sharma P, Yadav NR, Yadav RC (2013) MYB transcription factor genes as regulators for plant responses: an overview. Physiol Mol Biol Plants 19:307–321. https://doi.org/10.1007/s12298-013-0179-1
Ariel FD, Manavella PA, Dezar CA, Chan RL (2007) The true story of the HD-Zip family. Trend Plant Sci 12:419–426. https://doi.org/10.1016/j.tplants.2007.08.003
Bajaj D, Upadhyaya HD, Khan Y et al (2015) A combinatorial approach of comprehensive QTL-based comparative genome mapping and transcript profiling identified a seed weight-regulating candidate gene in chickpea. Sci Rep 5:9264. https://doi.org/10.1038/srep09264
Baker SC, Robinson-Beers K, Villanueva JM et al (1997) Interactions among genes regulating ovule development in Arabidopsis thaliana. Genetics 145:1109–1124. https://doi.org/10.1093/genetics/145.4.1109
Becker MG, Hsu SW, Harada JJ, Belmonte MF (2014) Genomic dissection of the seed. Front Plant Sci 5:464. https://doi.org/10.3389/fpls.2014.00464
Belmonte MF, Kirkbride RC, Stone SL et al (2013) Comprehensive developmental profiles of gene activity in regions and subregions of the Arabidopsis seed. Proc Natl Acad Sci USA 110:E435–E444. https://doi.org/10.1073/pnas.1222061110
Chen Z, Lancon-Verdier V, Le Signor C et al (2021) Genome-wide association study identified candidate genes for seed size and seed composition improvement in M. truncatula. Sci Rep 11:4224. https://doi.org/10.1038/s41598-021-83581-7
Cheng ZJ, Zhao XY, Shao XX et al (2014) Abscisic acid regulates early seed development in Arabidopsis by ABI5-mediated transcription of SHORT HYPOCOTYL UNDER BLUE1. Plant Cell 26:1053–1068. https://doi.org/10.1105/tpc.113.121566
Cheng Y, Liu J, Zhang H et al (2015) Transcriptome analysis and gene expression profiling of abortive and developing ovules during fruit development in hazelnut. PLoS ONE 10:e0122072. https://doi.org/10.1371/journal.pone.0122072
Chettoor AM, Givan SA, Cole RA et al (2014) Discovery of novel transcripts and gametophytic functions via RNA-seq analysis of maize gametophytic transcriptomes. Genome Biol 15:414. https://doi.org/10.1186/s13059-014-0414-2
Cobos MJ, Rubio J, Fernández-Romero MD et al (2007) Genetic analysis of seed size, yield and days to flowering in a chickpea recombinant inbred line population derived from a Kabuli × Desi cross. Ann Appl Biol 151:33–42. https://doi.org/10.1111/j.1744-7348.2007.00152.x
Del Toro-De LG, García-Aguilar M, Gillmor CS (2014) Non-equivalent contributions of maternal and paternal genomes to early plant embryogenesis. Nature 514:624–627. https://doi.org/10.1038/nature13620
Deveshwar P, Bovill WD, Sharma R et al (2011) Analysis of anther transcriptomes to identify genes contributing to meiosis and male gametophyte development in rice. BMC Plant Biol 11:78. https://doi.org/10.1186/1471-2229-11-78
Dresselhaus T, Franklin-Tong N (2013) Male-female crosstalk during pollen germination, tube growth and guidance, and double fertilization. Mol Plant 6:1018–1036. https://doi.org/10.1093/mp/sst061
Endress PK (2011) Angiosperm ovules: diversity, development, evolution. Ann Bot 107:1465–1489. https://doi.org/10.1093/aob/mcr120
Engineer CB, Ghassemian M, Anderson JC, Peck SC, Hu H, Schroeder JI (2014) Carbonic anhydrases, EPF2 and a novel protease mediate CO2 control of stomatal development. Nature 513:246–250. https://doi.org/10.1023/nature13452
Frey A, Godin B, Bonnet M et al (2004) Maternal synthesis of abscisic acid controls seed development and yield in Nicotiana plumbaginifolia. Planta 218:958–964. https://doi.org/10.1007/s00425-003-1180-7
Galbiati F, Sinha Roy D, Simonini S et al (2013) An integrative model of the control of ovule primordia formation. Plant J Cell Mol Biol 76:446–455. https://doi.org/10.1111/tpj.12309
Garg R, Singh VK, Rajkumar MS, Kumar V, Jain M (2017) Global transcriptome and coexpression network analyses reveal cultivar-specific molecular signatures associated with seed development and seed size/weight determination in chickpea. Plant J 91:1088–1107. https://doi.org/10.1111/tpj.13621
Gehring M (2013) Genomic imprinting: insights from plants. Annu Rev Genet 47:187–208. https://doi.org/10.1146/annurev-genet-110711-155527
Groszmann M, Paicu T, Smyth DR (2008) Functional domains of SPATULA, a bHLH transcription factor involved in carpel and fruit development in Arabidopsis. Plant J 55:40–52
Gutierrez-Gonzalez JJ, Tu ZJ, Garvin DF (2013) Analysis and annotation of the hexaploid oat seed transcriptome. BMC Genomics 14:471. https://doi.org/10.1186/1471-2164-14-471
Harris JC, Hrmova M, Lopato S, Langridge P (2011) Modulation of plant growth by HD-Zip class I and II transcription factors in response to environmental stimuli. New Phytol 190:823–837. https://doi.org/10.1111/j.1469-8137.2011.03722.x
Hiremath PJ, Farmer A, Cannon SB et al (2011) Large-scale transcriptome analysis in chickpea (Cicer arietinum L.), an orphan legume crop of the semi-arid tropics of Asia and Africa. Plant Biotechnol J 9:922–931. https://doi.org/10.1111/j.1467-7652.2011.00625.x
Hirner B, Fischer WN, Rentsch D, Kwart M, Frommer WB (1998) Developmental control of H+/amino acid permease gene expression during seed development of Arabidopsis. Plant J 14:535–544. https://doi.org/10.1046/j.1365-313x.1998.00151.x
Honys D, Twell D (2003) Comparative analysis of the Arabidopsis pollen transcriptome. Plant Physiol 132:640–652. https://doi.org/10.1104/pp.103.020925
Jhanwar S, Priya P, Garg R et al (2012) Transcriptome sequencing of wild chickpea as a rich resource for marker development. Plant Biotechnol J 10:690–702. https://doi.org/10.1111/j.1467-7652.2012.00712.x
Jin Y, Pan W, Zheng X, Cheng X, Liu M, Ma H, Ge X (2018) OsERF101, an ERF family transcription factor, regulates drought stress response in reproductive tissues. Plant Mol Biol 98:51–65. https://doi.org/10.1007/s11103-018-0762-5
Jones SI, Vodkin LO (2013) Using RNA-Seq to profile soybean seed development from fertilization to maturity. PLoS ONE 8:e59270. https://doi.org/10.1371/journal.pone.0059270
Kalve S, Tadege M (2017) A comprehensive technique for artificial hybridization in Chickpea (Cicer arietinum). Plant Methods 13:1–9. https://doi.org/10.1186/s13007-017-0202-6
Kang X, Li W, Zhou Y, Ni M (2013) A WRKY transcription factor recruits the SYG1-like protein SHB1 to activate gene expression and seed cavity enlargement. PLoS Genet 9:e1003347. https://doi.org/10.1371/journal.pgen.1003347
Kanno Y, Jikumaru Y, Hanada A et al (2010) Comprehensive hormone profiling in developing Arabidopsis seeds: examination of the site of ABA biosynthesis, ABA transport and hormone interactions. Plant Cell Physiol 51:1988–2001. https://doi.org/10.1093/pcp/pcq158
Kelley DR, Gasser CS (2009) Ovule development: genetic trends and evolutionary considerations. Sex Plant Reprod 22:229–234. https://doi.org/10.1007/s00497-009-0107-2
Kim Y-S, Kim S-G, Park J-E et al (2006) A membrane-bound NAC transcription factor regulates cell division in Arabidopsis. Plant Cell 18:3132–3144. https://doi.org/10.1105/tpc.106.043018
Kubo T, Fujita M, Takahashi H et al (2013) Transcriptome analysis of developing ovules in rice isolated by laser microdissection. Plant Cell Physiol 54:750–765. https://doi.org/10.1093/pcp/pct029
Kudapa H, Azam S, Sharpe AG et al (2014) Comprehensive transcriptome assembly of Chickpea (Cicer arietinum L.) using sanger and next generation sequencing platforms: development and applications. PLoS ONE 9:e86039. https://doi.org/10.1371/journal.pone.0086039
Kumar S, Thakur M, Mitra R et al (2022) Sugar metabolism during pre- and post-fertilization events in plants under high temperature stress. Plant Cell Rep 41:655–673. https://doi.org/10.1007/s00299-021-02795-1
Lagacé M, Matton DP (2004) Characterization of a WRKY transcription factor expressed in late torpedo-stage embryos of Solanum chacoense. Planta 219:185–189. https://doi.org/10.1007/s00425-004-1253-2
Le BH, Cheng C, Bui AQ et al (2010) Global analysis of gene activity during Arabidopsis seed development and identification of seed-specific transcription factors. Proc Natl Acad Sci 107:8063–8070. https://doi.org/10.1073/pnas.1003530107
Li X, Chen L, Hong M, Zhang Y et al (2012) A large insertion in bHLH transcription factor BrTT8 resulting in yellow seed coat in Brassica rapa. PLoS ONE 7:e44145. https://doi.org/10.1371/journal.pone.0044145
Li J, Guang Y, Yang Y, Zhou Y (2019) Identification and expression analysis of two allene oxide cyclase (AOC) genes in watermelon. Agriculture 9:225. https://doi.org/10.3390/agriculture9100225
Li D, Li S, Li W et al (2020) Comparative transcriptome analysis provides insights into the molecular mechanism underlying double fertilization between self-crossed Solanum melongena and that hybridized with Solanum aethiopicum. PLoS ONE 15:e0235962. https://doi.org/10.1371/journal.pone.0235962
Liu G, Ren G, Guirgis A, Thronburg RW (2009) The MYB305 transcription factor regulates expression of nectarin genes in the ornamental tobacco floral nectary. Plant Cell 21:2672–2687. https://doi.org/10.1105/tpc.108.060079
Locascio A, Roig-Villanova I, Bernardi J, Varotto S (2014) Current perspectives on the hormonal control of seed development in Arabidopsis and maize: a focus on auxin. Front Plant Sci 5:412. https://doi.org/10.3389/fpls.2014.00412
Ma L, Zhang D, Miao Q et al (2017) Essential role of sugar transporter OsSWEET11 during the early stage of rice grain filling. Plant Cell Physiol 58:863–873. https://doi.org/10.1093/pcp/pcx040
Ma Q, Chen C, Zeng Z et al (2018) Transcriptomic analysis between self- and cross-pollinated pistils of tea plants (Camellia sinensis). BMC Genomics 19:289. https://doi.org/10.1186/s12864-018-4674-1
Matilla AJ (2020) Auxin: hormonal signal required for seed development and dormancy. Plants 9:705. https://doi.org/10.3390/plants9060705
Miranda M, Borisjuk L, Tewes A, Heim U, Sauer N, Wobus U, Weber H (2001) Amino acid permeases in developing seeds of Vicia faba L.: expression precedes storage protein synthesis and is regulated by amino acid supply. Plant J 28:61–71. https://doi.org/10.1046/j.1365-313x.2001.01129.x
Pillitteri LJ, Peterson KM, Horst RJ, Torii KU (2011) Molecular profiling of stomatal meristemoids reveals new component of asymmetric cell division and commonalities among stem cell populations in Arabidopsis. Plant Cell 23:3260–3275. https://doi.org/10.1105/tpc.111.088583
Pradhan S, Bandhiwal N, Shah N et al (2014) Global transcriptome analysis of developing chickpea (Cicer arietinum L.) seeds. Front Plant Sci 5:698. https://doi.org/10.3389/fpls.2014.00698
Ribalta FM, Pazos-Navarro M, Edwards K et al (2019) Expression patterns of key hormones related to pea (Pisum sativum L.) embryo physiological maturity shift in response to accelerated growth conditions. Front Plant Sci 10:1154. https://doi.org/10.3389/fpls.2019.01154
Robert HS, Park C, Gutièrrez CL et al (2018) Maternal auxin supply contributes to early embryo patterning in Arabidopsis. Nat Plants 4:548–553. https://doi.org/10.1038/s41477-018-0204-z
Rodrigo J, Herrero M (1998) Influence of intraovular reserves on ovule fate in apricot (Prunus armeniaca L.). Sex Plant Reprod 11:86–93. https://doi.org/10.1007/s004970050124
Roodbarkelari F, Groot EP (2017) Regulatory function of homeodomain-leucine zipper (HD-ZIP) family proteins during embryogenesis. New Phytol 213:95–104. https://doi.org/10.1111/nph.14132
Ruan Y-L, Patrick JW, Bouzayen M et al (2012) Molecular regulation of seed and fruit set. Trends Plant Sci 17:656–665. https://doi.org/10.1016/j.tplants.2012.06.005
Rushton PJ, Somssich IE, Ringler P, Shen QJ (2010) WRKY transcription factors. Trends Plant Sci 15:247–258. https://doi.org/10.1016/j.tplants.2010.02.006
Schmidt A, Wuest SE, Vijverberg K et al (2011) Transcriptome analysis of the Arabidopsis megaspore mother cell uncovers the importance of RNA helicases for plant germline development. PLoS Biol 9:e1001155. https://doi.org/10.1371/journal.pbio.1001155
Schneitz K, Hülskamp M, Pruitt RE (1995) Wild-type ovule development in Arabidopsis thaliana: a light microscope study of cleared whole-mount tissue. Plant J 7:731–749. https://doi.org/10.1046/j.1365-313X.1995.07050731.x
Schulz KN, Harrison MM (2019) Mechanisms regulating zygotic genome activation. Nat Rev Genet 20:221–234. https://doi.org/10.1038/s41576-018-0087-x
Seo M, Marion-Poll A (2019) Chapter One - Abscisic acid metabolism and transport. In: Seo M, Marion-Poll A (eds) Advances in botanical research. Academic Press, pp 1–49
Severin AJ, Woody JL, Bolon Y-T et al (2010) RNA-Seq Atlas of Glycine max: a guide to the soybean transcriptome. BMC Plant Biol 10:160. https://doi.org/10.1186/1471-2229-10-160
Shen S, Ma S, Liu Y et al (2019) Cell wall invertase and sugar transporters are differentially activated in tomato styles and ovaries during pollination and fertilization. Front Plant Sci 10:506. https://doi.org/10.3389/fpls.2019.00506
Singh VK, Garg R, Jain M (2013) A global view of transcriptome dynamics during flower development in chickpea by deep sequencing. Plant Biotechnol J 11:691–701. https://doi.org/10.1111/pbi.12059
Souer E, van Houwelingen A, Kloos D et al (1996) The no apical meristem gene of Petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell 85:159–170. https://doi.org/10.1016/s0092-8674(00)81093-4
Sun H, Hu M, Li J et al (2018) Comprehensive analysis of NAC transcription factors uncovers their roles during fiber development and stress response in cotton. BMC Plant Biol 18:150. https://doi.org/10.1186/s12870-018-1367-5
Takahashi T, Mori T, Ueda K et al (2018) The male gamete membrane protein DMP9/DAU2 is required for double fertilization in flowering plants. Dev Camb Engl 145:dev170076. https://doi.org/10.1242/dev.170076
Tebbji F, Nantel A, Matton DP (2010) Transcription profiling of fertilization and early seed development events in a solanaceous species using a 7.7 K cDNA microarray from Solanum chacoenseovules. BMC Plant Biol 10:174. https://doi.org/10.1186/1471-2229-10-174
Tegeder M, Offler CE, Frommer WB, Patrick JW (2000) Amino acid transporters are localized to transfer cells of developing pea seeds. Plant Physiol 122:319–326. https://doi.org/10.1104/pp.122.2.319
Tolosa LN, Zhang Z (2020) The Role of major transcription factors in solanaceous food crops under different stress conditions: current and future perspectives. Plants Basel Switz 9:E56. https://doi.org/10.3390/plants9010056
Weber H, Borisjuk L, Heim U et al (1995) Seed coat-associated invertases of fava bean control both unloading and storage functions: cloning of cDNAs and cell type-specific expression. Plant Cell 7:1835–1846. https://doi.org/10.1105/tpc.7.11.1835
Whittle CA, Malik MR, Li R, Krochko JE (2010) Comparative transcript analyses of the ovule, microspore, and mature pollen in Brassica napus. Plant Mol Biol 72:279–299. https://doi.org/10.1007/s11103-009-9567-x
Wu X, Chory J, Weigel D (2007) Combinations of WOX activities regulate tissue proliferation during Arabidopsis embryonic development. Dev Biol 309:306–316. https://doi.org/10.1016/j.ydbio.2007.07.019
Wu C-C, Diggle PK, Friedman WE (2011) Female gametophyte development and double fertilization in Balsas teosinte, Zea mays subsp. parviglumis (Poaceae). Sex Plant Reprod 24:219–229. https://doi.org/10.1007/s00497-011-0164-1
Xu G, Huang J, Lei S-K et al (2019) Comparative gene expression profile analysis of ovules provides insights into Jatropha curcas L. ovule development. Sci Rep 9:15973. https://doi.org/10.1038/s41598-019-52421-0
Yang J, Zhang J, Huang Z et al (2002) Correlation of cytokinin levels in the endosperms and roots with cell number and cell division activity during endosperm development in rice. Ann Bot 90:369–377. https://doi.org/10.1093/aob/mcf198
You L, Yu L, Liang R et al (2021) Identification and analysis of genes involved in double fertilization in Rice. Int J Mol Sci 22:12850. https://doi.org/10.3390/ijms222312850
Yu H-J, Hogan P, Sundaresan V (2005) Analysis of the female gametophyte transcriptome of Arabidopsis by comparative expression profiling. Plant Physiol 139:1853–1869. https://doi.org/10.1104/pp.105.067314
Yuan Z, Zhang D (2015) Roles of jasmonate signalling in plant inflorescence and flower development. Curr Opin Plant Biol 27:44–51. https://doi.org/10.1016/j.pbi.2015.05.024
Zhang Y, Liang W, Shi J, Xu J, Zhang D (2013) MYB 56 encoding a R2 R 3 MYB transcription factor regulates seed size in Arabidopsis thaliana. J Integ Plant Biol 55:1166–1178. https://doi.org/10.1111/jipb.12094
Zhou Q, Cai Q (2021) Role of ethylene in the regulatory mechanism underlying the abortion of ovules after fertilization in Xanthoceras sorbifolium. Plant Mol Biol 106:67–84. https://doi.org/10.1007/s11103-021-01130-2
Zhou L-Z, Dresselhaus T (2019) Friend or foe: signaling mechanisms during double fertilization in flowering seed plants. Curr Top Dev Biol 131:453–496. https://doi.org/10.1016/bs.ctdb.2018.11.013
Acknowledgements
Authors would like to express gratitude to the Vice chancellor of Central University of Punjab, India for unconditional support during this course of work. RS acknowledged UGC for JRF fellowship for her doctoral work.
Funding
VK acknowledged the UGC-BSR research Start-Up-Grant for sponsoring this work.
Author information
Authors and Affiliations
Contributions
VK and SKY conceived and designed present work. VK, RS and RS, analyzed the data, RS and VK wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethical standard
Authors declared that the whole manuscript is strictly complied with ethical standards of 3Biotech.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, R., Shankar, R., Yadav, S.K. et al. Transcriptome analysis of ovules offers early developmental clues after fertilization in Cicer arietinum L.. 3 Biotech 13, 177 (2023). https://doi.org/10.1007/s13205-023-03599-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03599-8