Abstract
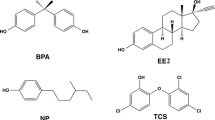
Thermophilic fungi are known to develop different metabolic and catabolic activities that enable them to function at elevated temperatures. Screening heat-resistant fungi, as promising resources for enzymatic activities, are still recommended. A total of eleven wood-decay thermophilic fungal strains were isolated from decaying organic materials (DOM) collected from arid areas of Khulais (Saudi Arabia). Six of these isolates are laccase-producing thermophilic strains growing at 50 °C. Among Laccase positive (Lac+) isolates, Chaetomium brasiliense (G3), Canariomyces notabilis (KW1), and Paecilomyces formosus (KW3) were exploited to treat single selected endocrine-disrupting chemicals (EDCs) that belonged to different classes (synthetic steroid hormone: 17α-ethinyl estradiol (EE2), and alkylphenols:4-tert-butylphenol (4-t-BP)). Chaetomium sp. was selected due to its potentialities against target EDCs, and then, their laccases were extracted and exploited for the biocatalytic degradation of treated municipal sewage wastewaters (TMWW) mixed with 4-t-BP and EE2. The results show that within 2 h of catalyzing at 50 °C, laccase could degrade 60 ± 4.8% of 4-t-BP; however, it oxidized EE2 less efficiently, reaching 35 ± 4.1%. The influence of some redox mediators on the laccase oxidation system was investigated. The 1-hydroxybenzotriazole (HBT) and syringaldehyde led to the highest transformation rates of EE2 (approximately 80 ± 2.4%). Near-total removal (90 ± 7.2%) of 4-t-BP was achieved with TEMPO in 2 h. With the metabolites identified through gas chromatography-tandem mass spectrometry (GC–MS), metabolic pathways of degradation were suggested. The results highlight the potential of Chaetomium sp. strains in the conversion of micropollutants.






Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Agrawal S, Nandeibam J, Sarangthem I (2021) Ultrastructural changes in methicillin-resistant Staphylococcus aureus (MRSA) induced by metabolites of thermophilous fungi Acrophialophora levis. PLoS ONE 16(10):e0258607. https://doi.org/10.1371/journal.pone.0258607
Ahirwar S, Soni H, Prajapati BP, Kango N (2017) Isolation and screening of thermophilic and thermotolerant fungi for production of hemicellulases from heated environments. Mycology 8:125–134. https://doi.org/10.1080/21501203.2017.1337657
Armenante A, Longobardi S, Rea I, De Stefano L, Giocondo M, Silipo A et al (2010) The Pleurotus ostreatus hydrophobin Vmh2 and its interaction with glucans. Glycobiology 20:594–602. https://doi.org/10.1093/glycob/cwq009
Ashe B, Nguyen LN, Hai FI, Lee D-J, van de Merwe JP, Leusch FDL, Price WE, Nghiem LD (2016) Impacts of redox-mediator type on trace organic contaminants degradation by laccase: degradation efficiency, laccase stability and effluent toxicity. Int Biodeterior Biodegrad 113:169–176
Barbosa MO, Moreira NFF, Ribeiro AR, Pereira MFR, Silva AMT (2016) Occurrence and removal of organic micropollutants: an overview of the watch list of EU decision 2015/495. Water Res 94:257–279. https://doi.org/10.1016/j.watres.2016.02.047
Ben Younes S, Karray F, Sayadi S (2011) Isolation of thermophilic fungal strains producing oxido-reductase and hydrolase enzymes from various Tunisian biotopes. Int Biodeter Biodegrad 65(7):1104–1109. https://doi.org/10.1016/j.ibiod.2011.08.003
Cajthaml T (2015) Biodegradation of endocrine-disrupting compounds by ligninolytic fungi: mechanisms involved in the degradation. Environ Microbiol 17(12):4822–4834
Chang Y-C, Reddy MV, Umemoto H, Kondo S, Choi DB (2020) Biodegradation of alkylphenols by rhizosphere microorganisms isolated from the roots of Hosta undulata. J Environ Chem Eng 8(3):103771
Chen Y-L, Fu H-Y, Lee T-H, Shih C-J, Huang L, Wang Y-S, Ismail W, Chiang Y-R (2018) Estrogen degraders and estrogen degradation pathway identified in an activated sludge. Appl Environ Microbiol 84:e00001-18. https://doi.org/10.1128/AEM.00001-18
Combalbert S, Hernandez-Raquet G (2010) Occurrence, fate, and biodegradation of estrogens in sewage and manure. Appl Microbiol Biotechnol 86(6):1671–1692
Cooney DG, Emerson R (1964) Methods of isolation and culture. Thermophilic fungi, an account of their biology, activities and classification. W.H. Freeman & Company, San Francisco, pp 8–13
Daâssi D, Prieto A, Zouari-Mechichi H, Martínez MJ, Nasri M, Mechichi T (2016a) Degradation of bisphenol A by different fungal laccases and identification of its degradation products. Int Biodeter Biodegrad 110:181–188
Daâssi D, Zouari-Mechichi H, Belbahri L, Barriuso J, Martınez MJ, Nasri M, Mechichi T (2016b) Phylogenetic and metabolic diversity of Tunisian forest wood-degrading fungi: a wealth of novelties and opportunities for biotechnology. 3 Biotech 6:46. https://doi.org/10.1007/s13205-015-0356-8
de Nooy AEJ, Besemer AC, van Bekkum H (1996) On the use of stable organic nitroxyl radicals for the oxidation of primary and secondary alcohols. Synthesis 10:1153–1174
De Toni L, Tisato F, Seraglia R, Roverso M, Gandin V, Marzano C, Padrini R, Foresta C (2017) Phthalates and heavy metals as endocrine disruptors in food: a study on pre-packed coffee products. Toxicol Rep 4:234–239
Esplugas S et al (2007) Ozonation and advanced oxidation technologies to remove endocrine disrupting chemicals (EDCs) and pharmaceuticals and personal care products (PPCPs) in water effluents. J Hazard Mater 149(3):631–642. https://doi.org/10.1016/j.jhazmat.2007.07.073
Gao X, Kang S, Xiong R, Chen M (2020) Environment-friendly removal methods for endocrine disrupting chemicals. Sustainability 12(18):7615. https://doi.org/10.3390/su12187615
Guardiol ALP, Belleville MP, Alanis M, Saldivar RP, Sanchez-Marcano J (2019) Effect of redox mediators in pharmaceuticals degradation by laccase: a comparative study. Process Biochem 78:123–131
Hibbett D, Abarenkov K, Kõljalg U, Öpik M, Chai B, Cole J, Wang Q, Crous P, Robert V, Helgason T, Herr JR et al (2016) Sequence-based classification and identification of Fungi. Mycologia 108(6):1049–1068. https://doi.org/10.3852/16-130
Huang S, Hyde KD, Mapook A, Maharachchikumbura SSN, Bhat JD, McKenzie EHC, Jeewon R, Wen T-C (2021a) Taxonomic studies of some often over-looked Diaporthomycetidae and Sordariomycetidae. Fungal Divers 111:443–572. https://doi.org/10.1007/s13225-021-00488-4
Huang Y, Chen Q, Wang Z, Yan H, Chen C, Yan D, Ji X (2021b) Abatement technology of endocrine-disrupting chemicals (EDCs) by means of enhanced coagulation and ozonation for wastewater reuse. Chemosphere 285:131515
Hutchinson MI, Powell AJ, Herrera J, Natvig DO (2019) New perspectives on the distribution and roles of thermophilic fungi. Chapter 4. Ecological role and biotechnological significance. 59–80. https://doi.org/10.1007/978-3-030-19030-9_4.
Hwang SS, Choi HT, Song HG (2008) Biodegradation of endocrine-disrupting phthalates by pleurotus ostreatus. J Microbial Biotechnol 18(4):767–772
Kiiskinen L-L, Ratto M, Kruus K (2004) Screening for novel laccase-producing microbes. J Appl Microbiol 97(3):640–646
Korniłłowicz-Kowalska T, Kitowski I (2012) Aspergillus fumigatus and other thermophilic fungi in nests of wetland birds. Mycopathologia 175(1–2):43–56. https://doi.org/10.1007/s11046-012-9582-3
Křesinová Z, Moeder M, Ezechiáš M, Svobodová K, Cajthaml T (2012) Mechanistic study of 17α-ethinylestradiol biodegradation by Pleurotus ostreatus: tracking of extracelullar and intracelullar degradation mechanisms. Environ Sci Technol 46(24):13377–13385. https://doi.org/10.1021/es3029507
Křesinová Z, Linhartová L, Filipová A, Ezechiáš M, Mašín P, Cajthaml T (2018) Biodegradation of endocrine disruptors in urban wastewater using Pleurotus ostreatus bioreactor. New Biotechnol 25(43):53–61
Kumar M, Sarma DK, Shubham S, Kumawat M, Verma V, Prakash A, Tiwari R (2020) Environmental endocrine-disrupting chemical exposure: role in non-communicable diseases. Front Public Health 8:553850. https://doi.org/10.3389/fpubh.2020.553850
Lange IG, Daxenberger A, Schiffer B, Witters H, Ibarreta D, Meyer HHD (2002) Sex hormones originating from different livestock production systems: fate and potential disrupting activity in the environment. Anal Chim Acta 473:27–37
Lloret L, Eibes G, Moreira MT, Feijoo G, Lema JM (2013) On the use of a high-redox potential laccase as an alternative for the transformation of non-steroidal anti-inflammatory drugs (NSAIDs). J Mol Catal B Enzym 97:233–242
Longe LF, Couvreur J, Grandchamp ML, Garnier G, Allais F, Saito K (2018) Importance of mediators for lignin degradation by fungal laccase. ACS Sustain Chem Eng 6(8):10097–10107. https://doi.org/10.1021/acssuschemeng.8b01426
Morozova OV, Shumakovich GP, Shleev SV, Yaropolov YI (2007) Laccase-mediator systems and their applications: a review. Appl Biochem Microbiol 43:523–535
Mtibaà R, de Eugenio L, Ghariani B, Louati I, Belbahri L, Nasri M, Mechichi T (2017) A halotolerant laccase from Chaetomium strain isolated from desert soil and its ability for dye decolourization. 3 Biotech 7:329
Mtibaà R, Ezzanad A, Aranda E, Pozo C, Ghariani B, Moraga J, Nasri M, Garrido CJM (2020) Biodegradation and toxicity reduction of nonylphenol, 4-tert-octylphenol and 2,4-dichlorophenol by the ascomycetous fungus Thielavia sp HJ22: Identification of fungal metabolites and proposal of a putative pathway. Sci Total Environ 708:135129
Palma TL, Shylova A, Costa MC (2021) Isolation and characterization of bacteria from activated sludge capable of degrading 17α-ethinylestradiol, a contaminant of high environmental concern. Microbiology. https://doi.org/10.1099/mic.0.001038
Papaevangelou VA, Gikas GD, Tsihrintzis VA, Antonopoulou M, Konstantinou IK (2016) Removal of endocrine disrupting chemicals in HSF and VF pilot-scale constructed wetlands. Chem Eng J 294:146–156
Pezzella C, Macellaro G, Sannia G, Raganati F, Olivieri G, Marzocchella A, Schlosser D, Piscitelli A (2017) Exploitation of Trametes versicolor for bioremediation of endocrine-disrupting chemicals in bioreactors. PLoS ONE 12(6):e0178758
Rodríguez E, Ruiz-Duenas FJ, Kooistra R, Ram A, Martínez AT, Martínez MJ (2008) Isolation of two laccase genes from the white-rot fungus Pleurotus eryngii and heterologous expression of the pel3 encoded protein. J Biotechnol 134:9–19
Rossello Mora R, Amman R (2001) The species concept for prokaryotes. Microbiol Res 25:39–67
Samson RA, Houbraken J, Varga J, Frisvad JC (2009) Polyphasic taxonomy of the heat resistant ascomycete genus Byssochlamys and its Paecilomyces anamorphs. Persoonia Mol Phylogeny Evol Fungi 22:14–27
Sarangapani C, Danaher M, Tiwari B, Lu P, Bourke P, Cullen PJ (2017) Efficacy and mechanistic insights into endocrine disruptor degradation using atmospheric air plasma. Chem Eng J 326:700–714
Saroj P, Manasa P, Narasimhulu K (2018) Characterization of thermophilic fungi producing extracellular lignocellulolytic enzymes for lignocellulosic hydrolysis under solid-state fermentation. Bioresour Bioprocess 5:31. https://doi.org/10.1186/s40643-018-0216-6
Siegrist H, Joss A, Ternes T, Oehlmann J (2005) Fate of EDCs in wastewater treatment and EU perspective on EDC regulation. Proc Water Envir 13:3142–3165. https://doi.org/10.2175/193864705783865640
Suzuki K, Hirai H, Murata H, Nishida T (2003) Removal of estrogenic activities of 17β-estradiol and ethinylestradiol by ligninolytic enzymes from white rot fungi. Water Res 37(8):1972–1975. https://doi.org/10.1016/S0043-1354(02)00533-X
Syafrudin M, Ayu Kristanti R, Yuniarto A, Hadibarata T, Rhee J, Al-onazi WA, Saad Algarni T, Almarri AH, Al-Mohaimeed AM (2021) Pesticides in drinking water—A review. Int J Environ Res Public Health 18(2):468. https://doi.org/10.3390/ijerph18020468
Taboada-Puig R, Moreira MT, Agathos SN, Lema JM (2017) The impact of endocrine disrupting chemicals on the environmental and their potential biotransformation by White-rot Fungi and their oxidative enzymes. Rev Bionatura 2(1):263–271. https://doi.org/10.21931/RB/2017.02.01.9
Tamura K, Nei M, Kumar S (2004) Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc Natl Acad Sci U S A 101(30):11030–11035. https://doi.org/10.1073/pnas.0404206101
Tamura K, Stecher G, Kumar S (2021) MEGA 11: Molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027. https://doi.org/10.1093/molbev/msab120
Teles M, Gravato C, Pacheco M, Santos MA (2004) Juvenile sea bass biotransformation, genotoxic and endocrine responses to β-naphthoflavone, 4-nonylphenol and 17β-estradiol individual and combined exposures. Chemosphere 57:147–158. https://doi.org/10.1016/j.chemosphere.2004.02.023
Toyama T, Momotani N, Ogata Y, Miyamori Y, Inoue D, Sei K, Mori K, Kikuchi S, Ike M (2010) Isolation and characterization of 4-tert-butylphenol-utilizing Sphingobium fuliginis strains from Phragmites australis rhizosphere sediment. Appl Environ Microbiol 76:6733–6740. https://doi.org/10.1128/AEM.00258-10
Vandenberg LN, Maffini MV, Sonnenschein C, Rubin BS, Soto AM (2009) Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption. Endocr Rev 30:75–95
Wang R, Ma X, Liu T, Li Y, Song L, Tjong SC, Cao L, Wang W, Yua Q, Wang Z (2020) Degradation aspects of endocrine-disrupting chemicals: a review on photocatalytic processes and photocatalysts. Appl Catal A Gen 597:117547. https://doi.org/10.1016/j.apcata.2020.117547
Wang F, Zhang Q, Sha W, Wang X, Miao M, Wang Z-L, Li Y (2021) The adsorption of endocrine-disrupting compounds (EDCs) on carbon nanotubes: effects of algal extracellular organic matter (EOM). J Nanopart Res 23(4):109
Whitman WB (2017) Bacteria and the fate of estrogen in the environment. Cell Chem Biol 24:652–653
Xu F, Kulys JJ, Duke K, Li K, Krikstopaitis K, Deussen HJW, Abbate E, Galinyte V, Schneider P (2000) Redox chemistry in laccase-catalyzed oxidation of N-hydroxy compounds. Appl Environ Microb 66:2052–2056
Zhang C, Li Y, Wang C, Niu L, Cai W (2016) Occurrence of endocrine disrupting compounds in aqueous environment and their bacterial degradation: a review. Crit Rev Environ Sci Technol 46:1–5
Acknowledgements
All the authors acknowledge and thank the Chemical Department of the College of Science and Arts of Khulais, for allowing the use of spectrophotometer. Also, we thank the students Lama Jamal Hussain Alssulime and Fatimah Qabil Abdulrahman Almaghrabi for helping in wood and wastewaters sampling.
Funding
This project was funded by the Deanship of Scientific Research (DSR) of the University of Jeddah, Jeddah, Saudi Arabia, under Grant No. (UJ-20-109-DR). Therefore, the authors acknowledge and thank the DSR for its technical and financial support.
Author information
Authors and Affiliations
Contributions
DD proposed the research topic, provided necessary tools for the experiments, conceived, planned, and conducted all experiments, collected the data, contributed to the analysis and interpretation of the results, and contributed substantially to the writing and revision of the manuscript. DD was the academic supervisor of the student SRA and the principal investigator. SRA provided some necessary tools for the experiments and performed the GC–MS analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Daâssi, D., Alharbi, S.R. Degradation of endocrine-disrupting chemicals in wastewater by new thermophilic fungal isolates and their laccases. 3 Biotech 13, 26 (2023). https://doi.org/10.1007/s13205-022-03439-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03439-1