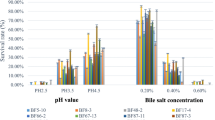
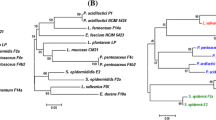
Abstract
This study was carried out to understand the probiotic features, ability to utilize non-digestible carbohydrates and comparative genomics of anti-inflammatory Bifidobacterium strains isolated from human infant stool samples. Bacterial strains were isolated from the stool samples using serial dilution on MRS agar plates supplemented with 0.05% l-cysteine hydrochloride and mupirocin. Molecular characterization of the strains was carried out by 16S rRNA gene sequencing. Anti-inflammatory activity was determined using TNF-α and lipopolysaccharide (LPS) induced inflammation in Caco2 cells. Probiotic attributes were determined as per the established protocols. Isomaltooligosaccharides (IMOS) utilization was determined in the broth cultures. Whole genome sequencing and analysis was carried out for three strains. Four obligate anaerobic, Gram positive Bifidobacterium strains were isolated from the infant stool samples. Strains were identified as Bifidobacterium longum Bif10, B. breve Bif11, B. longum Bif12 and B. longum Bif16. The strains were able to prevent inflammation in the Caco2 cells through lowering of IL8 production that was caused by TNF-α and LPS treatment. The strains exhibited desirable probiotic attributes such as acid and bile tolerance, mucin binding, antimicrobial activity, bile salt hydrolase activity, cholesterol lowering ability and could ferment non-digestible carbohydrates such as isomaltooligosaccharides and raffinose. Furthermore, Isomaltooligosaccharides supported the optimum growth of the strains in vitro, which was comparable to that on glucose. Strains could metabolize IMOS through cell associated α-glucosidase activity. Genomic features revealed the presence of genes responsible for the utilization of IMOS and for the probiotic attributes.





Similar content being viewed by others
References
Abe F, Muto M, Yaeshima T, Iwatsuki K, Aihara H, Ohashi Y, Fujisawa T (2010) Safety evaluation of probiotic bifidobacteria by analysis of mucin degradation activity and translocation ability. Anaerobe 16:131–136. https://doi.org/10.1016/j.anaerobe.2009.07.006
Achi SC, Talahalli RR, Halami PM (2019) Prophylactic effects of probiotic Bifidobacterium spp. in the resolution of inflammation in arthritic rats. Appl Microbiol Biotechnol 103:6287–6296. https://doi.org/10.1007/s00253-019-09864-2
Alnaseri H, Arsic B, Schneider JET, Kaiser JC, Scinocca ZC, Heinrichs DE, McGavin MJ (2015) Inducible expression of a resistance-nodulation-division-type efflux pump in Staphylococcus aureus provides resistance to linoleic and arachidonic acids. J Bacteriol 197:1893–1905. https://doi.org/10.1128/JB.02607-14
Andriantsoanirina V, Allano S, Butel MJ, Aires J (2013) Tolerance of Bifidobacterium human isolates to bile, acid and oxygen. Anaerobe 21:39–42. https://doi.org/10.1016/j.anaerobe.2013.04.005
Antikainen J, Kupannen V, Lahteenmaki K, Korhonen TK (2007) pH-dependent association of enolase and glyceraldehyde-3-phosphate dehydrogenase of Lactobacillus crispatus with the cell wall and lipoteichoic acids. J Bacteriol 189:4539–4543. https://doi.org/10.1128/JB.00378-07
Arboleya S, Watkins C, Stanton C, Ross RP (2016) Gut bifidobacteria populations in human health and aging. Front Microbiol 7:1204. https://doi.org/10.3389/fmicb.2016.01204
Aziz RK, Bartels D, Best A, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O (2008) The RAST server: rapid annotations using subsystems technology. BMC Genomics 9:75. https://doi.org/10.1186/1471-2164-9-75
Belkaid Y, Hand TW (2014) Role of the microbiota in immunity and inflammation. Cell 157:121–141. https://doi.org/10.1016/j.cell.2014.03.011
Bermudez-Brito M, Plaza-Díaz J, Muñoz-Quezada S, Gómez-Llorente C, Gil A (2012) Probiotic mechanisms of action. Ann Nutr Metab 61:160–174. https://doi.org/10.1159/000342079
Blin K, Shaw S, Steinke K, Villebro R, Ziemert N, Lee SY, Medema MH, Weber T (2019) AntiSMASH 5.0: updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res 47:W81–W87. https://doi.org/10.1093/nar/gkz310
Carevic M, Banjanac K, Corovic M, Jakovetic S, Milivojevic A, Vukasinovic-Sekulic M, Bezbradica D (2016) Selection of lactic acid bacteria strain for simultaneous production of α- and β-galactosidases. Zast Mater 57:265–273. https://doi.org/10.5937/zasmat1602265c
Cazorla SI, Maldonado-Galdeano C, Weill R, De Paula J, Perdigón GDV (2018) Oral administration of probiotics increases paneth cells and intestinal antimicrobial activity. Front Microbiol 9:736. https://doi.org/10.3389/fmicb.2018.00736
Chander AM, Singh S, Sharma S, Chaudhry V, Rajarammohan S, Mantri SS, Bishnoi M, Bhadada SK, Kondepudi KK (2020) Draft Genome Sequence of Bifidobacterium pseudocatenulatum Bif 4, Isolated from Healthy Infant Feces. Microbiol Resour Announc 9(26):e00561-e620. https://doi.org/10.1128/mra.00561-20
Corcoran BM, Stanton C, Fitzgerald GF, Ross RP (2005) Survival of probiotic lactobacilli in acidic environments is enhanced in the presence of metabolizable sugars. Appl Environ Microbiol 71:3060–3067. https://doi.org/10.1128/AEM.71.6.3060-3067.2005
Couvin D, Bernheim A, Toffano-Nioche C, Touchon M, Michalik J, Néron B, Rocha EPC, Vergnaud G, Gautheret D, Pourcel C (2018) CRISPRCasFinder, an update of CRISRFinder, includes a portable version, enhanced performance and integrates search for Cas proteins. Nucleic Acids Res 46:W246–W251. https://doi.org/10.1093/nar/gky425
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Fu X, Liu Z, Zhu C, Mou H, Kong Q (2019) Nondigestible carbohydrates, butyrate, and butyrate-producing bacteria. Crit Rev Food Sci Nutr 59:S130–S152. https://doi.org/10.1080/10408398.2018.1542587
Gagliardi A, Totino V, Cacciotti F, Iebba V, Neroni B, Bonfiglio G, Trancassini M, Passariello C, Pantanella F, Schippa S (2018) Rebuilding the gut microbiota ecosystem. Int J Environ Res Public Health 15:1679. https://doi.org/10.3390/ijerph15081679
Ganguly NK, Bhattacharya SK, Sesikeran B, Nair GB, Ramakrishna BS, Sachdev HPS, Batish VK, Kanagasabapathy AS, Muthuswamy V, Kathuria SC, Katoch VM, Satyanarayana K, Toteja GS, Rahi M, Rao S, Bhan MK, Kapur R, Hemalatha R (2011) ICMR-DBT Guidelines for evaluation of probiotics in food. Indian J Med Res 134:22–25. https://www.ijmr.org.in/text.asp?2011/134/1/22/83322
Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, Scott K, Stanton C, Swanson KS, Cani PD, Verbeke K, Reid G (2017) Expert consensus document: the international scientific association for probiotics and prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol 14:491–502. https://doi.org/10.1038/nrgastro.2017.75
Guarino MPL, Altomare A, Emerenziani S, Di Rosa C, Ribolsi M, Balestrieri P, Iovino P, Rocchi G, Cicala M (2020) Mechanisms of action of prebiotics and their effects on gastro-intestinal disorders in adults. Nutrients 12:1037. https://doi.org/10.3390/nu12041037
Islam ST, Taylor VL, Qi M, Lam JS (2010) Membrane topology mapping of the O-antigen flippase (Wzx), polymerase (Wzy), and ligase (WaaL) from Pseudomonas aeruginosa PAO1 reveals novel domain architectures. Mbio 1(3):e00189-10. https://doi.org/10.1128/mBio.00189-10
Kajander K, Myllyluoma E, Rajilić-Stojanović M, Kyronpalo S, Rasmussen M, Jarvenpaa S, Zoetendal EG, De Vos WM, Vapaatalo H, Korpela R (2008) Clinical trial: multispecies probiotic supplementation alleviates the symptoms of irritable bowel syndrome and stabilizes intestinal microbiota. Aliment Pharmacol Ther 27:48–57. https://doi.org/10.1111/j.1365-2036.2007.03542.x
Kataria A, Achi SC, Halami PM (2018) Effect of Encapsulation on viability of Bifidobacterium longum CFR815j and physiochemical properties of ice cream. Indian J Microbiol 58:248–251. https://doi.org/10.1007/s12088-018-0720-6
Kondepudi KK, Ambalam P, Nilsson I, Wadström T, Ljungh Å (2012) Prebiotic-non-digestible oligosaccharides preference of probiotic bifidobacteria and antimicrobial activity against Clostridium difficile. Anaerobe 18:489–497. https://doi.org/10.1016/j.anaerobe.2012.08.005
Kumar H, Collado MC, Wopereis H, Salminen S, Knol J, Roeselers G (2020) The bifidogenic effect revisited—ecology and health perspectives of bifidobacterial colonization in early life. Microorganisms 8:1–20. https://doi.org/10.3390/microorganisms8121855
Lee J-H, O’Sullivan DJ (2010) Genomic Insights into Bifidobacteria. Microbiol Mol Biol Rev 74:378–416. https://doi.org/10.1128/mmbr.00004-10
Li SC, Hsu WF, Chang JS, Shih CK (2019) Combination of Lactobacillus acidophilus and Bifidobacterium animalis subsp. Lactis shows a stronger anti-inflammatory effect than individual strains in HT-29 cells. Nutrients 11(5):969. https://doi.org/10.3390/nu11050969
Lye HS, Rusul G, Liong MT (2010) Removal of cholesterol by lactobacilli via incorporation and conversion to coprostanol. J Dairy Sci 93:1383–1392. https://doi.org/10.3168/jds.2009-2574
Mandlik A, Swierczynski A, Das A, Ton-That H (2008) Pili in Gram-positive bacteria: assembly, involvement in colonization and biofilm development. Trends Microbiol 16:33–40. https://doi.org/10.1016/j.tim.2007.10.010
Marianelli C, Cifani N, Pasquali P (2010) Evaluation of antimicrobial activity of probiotic bacteria against Salmonella enterica subsp. enterica serovar typhimurium 1344 in a common medium under different environmental conditions. Res Microbiol 161:673–680. https://doi.org/10.1016/j.resmic.2010.06.007
Meyer D, Stasse-Wolthuis M (2009) The bifidogenic effect of inulin and oligofructose and its consequences for gut health. Eur J Clin Nutr 63:1277–1289. https://doi.org/10.1038/ejcn.2009
Pokusaeva K, Fitzgerald GF, Van Sinderen D (2011) Carbohydrate metabolism in Bifidobacteria. Genes Nutr 6:285–306. https://doi.org/10.1007/s12263-010-0206-6
Rohith HS, Halami PM (2021) The combined effect of potential probiotic Bacillus licheniformis MCC 2514 and Bifidobacterium breve NCIM 5671 towards anti-inflammatory activity on HT-29 cell lines. Probiotics Antimicro Prot. https://doi.org/10.1007/s12602-021-09851-y
Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, Tuohy K (2018) Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr 57:1–24. https://doi.org/10.1007/s00394-017-1445-8
Ruiz L, Delgado S, Ruas-Madiedo P, Sánchez B, Margolles A (2017) Bifidobacteria and their molecular communication with the immune system. Front Microbiol 8:2345. https://doi.org/10.3389/fmicb.2017.02345
Sarkar A, Lehto SM, Harty S, Dinan TG, Cryan JF, Burnet PWJ (2016) Psychobiotics and the manipulation of bacteria–gut–brain signals. Trends Neurosci 39:763–781. https://doi.org/10.1016/j.tins.2016.09.002
Schindler BD, Kaatz GW (2016) Multidrug efflux pumps of Gram-positive bacteria. Drug Resist Updat 27:1–13. https://doi.org/10.1016/j.drup.2016.04.003
Singh D, Khare P, Zhu J, Kondepudi KK, Singh J, Baboota RK, Boparai RK, Khardori R, Chopra K, Bishnoi M (2016) A novel cobiotic-based preventive approach against high-fat diet-induced adiposity, nonalcoholic fatty liver and gut derangement in mice. Int J Obes 40:487–496. https://doi.org/10.1038/ijo.2015.197
Singh S, Bhatia R, Singh A, Singh P, Kaur R, Khare P, Purama RK, Boparai R, Rishi P, Ambalam P, Bhadada S, Bishnoi M, Kaur J, Kondepudi KK (2018) Probiotic attributes and prevention of LPS-induced pro-inflammatory stress in RAW264.7 macrophages and human intestinal epithelial cell line (Caco2) by newly isolated Weissella cibaria strains. Food Funct 9:1254–1264. https://doi.org/10.1039/C7FO00469A
Singh S, Bhatia R, Khare P, Sharma S, Rajarammohan S, Bishnoi M, Bhadada SK, Sharma SS, Kaur J, Kondepudi KK (2020) Anti-inflammatory Bifidobacterium strains prevent dextran sodium sulfate induced colitis and associated gut microbial dysbiosis in mice. Sci Rep 10:18597. https://doi.org/10.1038/s41598-020-75702-5
Sundararaman A, Bansal K, Sidhic J, Patil P, Prakash PM (2021) Genome of Bifidobacterium longum NCIM 5672 provides insights into its acid-tolerance mechanism and probiotic properties. Arch Microbiol 203:6109–6118. https://doi.org/10.1007/s00203-021-02573-3
Turroni F, Milani C, Duranti S, Mancabelli L, Mangifesta M, Viappiani A, Lugli GA, Ferrario C, Gioiosa L, Ferrarini A, Li J, Palanza P, Delledonne M, Van Sinderen D, Ventura M (2016) Deciphering bifidobacterial-mediated metabolic interactions and their impact on gut microbiota by a multi-omics approach. ISME J 10:1656–1668. https://doi.org/10.1038/ismej.2015.236
Usta-Gorgun B, Yilmaz-Ersan L (2020) Short-chain fatty acids production by Bifidobacterium species in the presence of salep. Electron J Biotechnol 47:29–35. https://doi.org/10.1016/j.ejbt.2020.06.004
Valdes AM, Walter J, Segal E, Spector TD (2018) Role of the gut microbiota in nutrition and health. BMJ 361:36–44. https://doi.org/10.1136/bmj.k2179
Van Heel AJ, De Jong A, Song C, Viel JH, Kok J, Kuipers OP (2018) BAGEL4: A user-friendly web server to thoroughly mine RiPPs and bacteriocins. Nucleic Acids Res 46:W278–W281. https://doi.org/10.1093/nar/gky383
Woodmansey EJ (2007) Intestinal bacteria and ageing. J Appl Microbiol 102:1178–1186. https://doi.org/10.1111/j.1365-2672.2007.03400.x
Zhou JS, Gopal PK, Gill HS (2001) Potential probiotic lactic acid bacteria Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019) do not degrade gastric mucin in vitro. Int J Food Microbiol 63:81–90. https://doi.org/10.1016/S0168-1605(00)00398-6
Acknowledgements
Authors would like to acknowledge National Agri-Food Biotechnology Institute (NABI); Department of Biotechnology, Government of India and Indian Council of Medical Research for providing financial support for this research. DBT-e-Library Consortium (DeLCON) is gratefully acknowledged for providing access to electronic journals.
Funding
This study was funded by Core Grant from NABI, DBT, Government of India and Indian council of Medical Research: File No: 5/9/1312/2020-Nut.
Author information
Authors and Affiliations
Contributions
SS1, SS2, KK and MB conceived the research and designed the experiments. SS2, KK and SKB isolated the Bifidobacterium strains; SS1, SS2, RM and PR conducted the experiments. VC, SM, AC, SR and RS performed the bacterial genomic characterization and in-silico data analysis. SS1, SS2, MB, SKB and KK analyzed the data. SS1, SS2, VC, SM, AC, MB and KK wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Data availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Whole genome sequencing data presented in this study are openly available in The National Centre for Biotechnology Information (NCBI).
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Consent for publication
Authors undersigned and give the consent for the publication of the given research material in the above journal.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, S., Singh, S., Chaudhary, V. et al. Isomaltooligosaccharides utilization and genomic characterization of human infant anti-inflammatory Bifidobacterium longum and Bifidobacterium breve strains. 3 Biotech 12, 89 (2022). https://doi.org/10.1007/s13205-022-03141-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03141-2