Abstract
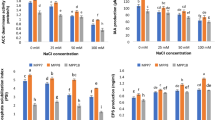
Salinity stress is one of the most serious environmental stresses which limit plant growth, development and productivity. In this study, we screened 25 bacterial isolates based on the biochemical activity of ACC deaminase. Two potent PGPR namely Bacillus marisflavi (CHR JH 203) and Bacillus cereus (BST YS1_42) having the highest ACC deaminase (ACCD) activity were selected for further analyses such as polymerase chain reaction (PCR), salt tolerance assay, expression analysis, antioxidant assay, etc. The structural gene for ACCD activity was further confirmed by PCR showing the amplicon size ̴ 800 bp. The acdS positive isolates exhibited optimum growth at 3% w/v (NaCl), indicating its ability to survive and thrive in induced saline soil. Inoculation of acdS+ strain on pea plants was found to be efficient and ameliorated the induced NaCl-stress by enhancing the various parameters like plant-biomass, carbohydrates, reducing sugars, protein, chlorophylls, phenol, flavonoids content and increasing antioxidants enzymes levels in plants. Moreover, the expression of ROS scavenging genes (PsSOD, PsCAT, PsPOX, PsNOS, PsAPX, PsChla/bBP), defense genes and cell rescue genes (PsPRP, PsMAPK, PsFDH) were analyzed. Inoculated plants exhibited a higher gene expression level and salt tolerance under 1%NaCl concentration. Thus, our results indicate that CHR JH 203 and BST YS1_42 strain showed the highest plant growth-promoting attributes could be used as bio-inoculants for crops under saline stress in the field towards sustainable crop development.








Similar content being viewed by others
References
Abdel Latef AAH, Chaoxing H (2014) Does Inoculation with Glomus mosseae improve salt tolerance in pepper plants? J Plant Growth Regul 33:644–653. https://doi.org/10.1007/s00344-014-9414-4
Abeles WF (1992) Roles and physiological effects of ethylene in plant physiology : dormancy, growth, and development. Ethyl Plant Biol
Aebi H (1984) [13] Catalase invitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahanger MA, Agarwal RM (2017) Salinity stress induced alterations in antioxidant metabolism and nitrogen assimilation in wheat (Triticum aestivum L.) as influenced by potassium supplementation. Plant Physiol Biochem 115:449–460. https://doi.org/10.1016/J.PLAPHY.2017.04.017
Ahmad P (2012) Salt-induced changes in photosynthetic activity and oxidative defense system of three cultivars of mustard (Brassica juncea L.). Afr J Biotechnol 11:2694–2703. https://doi.org/10.5897/ajb11.3203
Ali S, Charles TC, Glick BR (2014a) Amelioration of high salinity stress damage by plant growth-promoting bacterial endophytes that contain ACC deaminase. Plant Physiol Biochem 80:160–167. https://doi.org/10.1016/j.plaphy.2014.04.003
Ali S, Charles TC, Glick BR (2014b) Plant physiology and biochemistry amelioration of high salinity stress damage by plant growth- promoting bacterial endophytes that contain ACC deaminase. Plant Physiol Biochem 80:160–167. https://doi.org/10.1016/j.plaphy.2014.04.003
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Arnon DI (1949) Copper enzymes in isolated chloroplasts. polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Bal HB, Nayak L, Das S, Adhya TK (2013) Isolation of ACC deaminase producing PGPR from rice rhizosphere and evaluating their plant growth promoting activity under salt stress. Plant Soil. https://doi.org/10.1007/s11104-012-1402-5
Barnawal D, Bharti N, Maji D et al (2014) ACC deaminase-containing Arthrobacter protophormiae induces NaCl stress tolerance through reduced ACC oxidase activity and ethylene production resulting in improved nodulation and mycorrhization in Pisum sativum. J Plant Physiol 171:884–894. https://doi.org/10.1016/j.jplph.2014.03.007
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Belimov AA, Safronova VI, Sergeyeva TA et al (2001) Characterization of plant growth promoting rhizobacteria isolated from polluted soils and containing 1-aminocyclopropane-1-carboxylate deaminase. Can J Microbiol 47:642–652. https://doi.org/10.1139/w01-062
Blaha D, Prigent-Combaret C, Mirza MS, Moënne-Loccoz Y (2006) Phylogeny of the 1-aminocyclopropane-1-carboxylic acid deaminase-encoding gene acdS in phytobeneficial and pathogenic Proteobacteria and relation with strain biogeography. FEMS Microbiol Ecol 56:455–470. https://doi.org/10.1111/j.1574-6941.2006.00082.x
Bouffaud ML, Renoud S, Dubost A et al (2018) 1-Aminocyclopropane-1-carboxylate deaminase producers associated to maize and other Poaceae species. Microbiome. https://doi.org/10.1186/s40168-018-0503-7
Burd GI, Dixon DG, Glick BR (2000) Plant growth-promoting bacteria that decrease heavy metal toxicity in plants. Can J Microbiol 46:237–245. https://doi.org/10.1139/w99-143
Chance B, Maehly AC (1955) [136] Assay of catalases and peroxidases. {black small square}. Methods Enzymol 2:764–775. https://doi.org/10.1016/S0076-6879(55)02300-8
Chang CC, Yang MH, Wen HM, Chern JC (2002) Estimation of total flavonoid content in propolis by two complementary colometric methods. J Food Drug Anal 10:178–182. https://doi.org/10.38212/2224-6614.2748
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560
Cheng Z, Park E, Glick BR (2007) 1-Aminocyclopropane-1-carboxylate deaminase from Pseudomonas putida UW4 facilitates the growth of canola in the presence of salt. Can J Microbiol 53:912–918. https://doi.org/10.1139/W07-050
Cristina M, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649. https://doi.org/10.1146/annurev-arplant-042809-112252
Du J, Huang YP, Xi J et al (2008) Functional gene-mining for salt-tolerance genes with the power of Arabidopsis. Plant J 56:653–664. https://doi.org/10.1111/j.1365-313X.2008.03602.x
Dubois M, Gilles K, Hamilton JK et al (1951) A colorimetric method for the determination of sugars. Nature 168:167. https://doi.org/10.1038/168167a0
Dworkin M, Foster JW (1958) Experiments with some microorganisms which utilize ethane and hydrogen. J Bacteriol 75:592–603
Efremova N, Schreiber L, Bär S et al (2004) Functional conservation and maintenance of expression pattern of FIDDLEHEAD-like genes in Arabidopsis and Antirrhinum. Plant Mol Biol 56:821–837. https://doi.org/10.1007/s11103-004-5576-y
Farooq M, Hussain M, Wahid A, Siddique KHM (2012) Drought stress in plants: an overview. Plant responses to drought stress: from morphological to molecular features. Springer, Berlin, Heidelberg, pp 1–33
Flowers TJ, Galal HK, Bromham L (2010) Evolution of halophytes: multiple origins of salt tolerance in land plants. Funct Plant Biol 37:604. https://doi.org/10.1071/FP09269
Freitas VS, Miranda RS, Freitas VS et al (2018) Ethylene triggers salt tolerance in maize genotypes by modulating polyamine catabolism enzymes associated with H2O2 production. Environ Exp Bot 145:75–86. https://doi.org/10.1016/J.ENVEXPBOT.2017.10.022
Ghosh PK, De TK, Maiti TK (2018) Role of ACC deaminase as a stress ameliorating enzyme of plant growth-promoting rhizobacteria useful in stress agriculture: a review. Role Rhizospheric Microbes Soil Stress Manag Agric Sustain 1:57–106. https://doi.org/10.1007/978-981-10-8402-7_3
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Glick BR, Glick BR (2020) Modulating phytohormone levels. Beneficial plant-bacterial interactions. Springer International Publishing, New York, pp 139–180
Glick BR, Karaturovic DM, Newell PC (1995) A novel procedure for rapid isolation of plant growth promoting pseudomonads. Can J Microbiol 41:533–536
Gontia-Mishra I, Sapre S, Sharma A, Tiwari S (2016) Amelioration of drought tolerance in wheat by the interaction of plant growth-promoting rhizobacteria. Plant Biol 18:992–1000. https://doi.org/10.1111/PLB.12505
Gupta A, Bano A, Rai S et al (2021a) Plant growth promoting rhizobacteria (PGPR): a sustainable agriculture to rescue the vegetation from the effect of biotic stress: a review 10:2459–2465. https://doi.org/10.33263/LIANBS103.24592465
Gupta A, Rai S, Bano A et al (2021b) Comparative evaluation of different salt-tolerant plant growth-promoting bacterial isolates in mitigating the induced adverse effect of salinity in Pisum sativum 11:13141–13154
Habib SH, Kausar H, Saud HM (2016) Plant growth-promoting rhizobacteria enhance salinity stress tolerance in okra through ros-scavenging enzymes. Biomed Res Int. https://doi.org/10.1155/2016/6284547
Hontzeas N, Richardson AO, Belimov A et al (2005) Evidence for horizontal transfer of 1-aminocyclopropane-1-carboxylate deaminase genes. Appl Environ Microbiol 71:7556–7558. https://doi.org/10.1128/AEM.71.11.7556-7558.2005
Hossein H, Rezvani MP (2006) Effect of water and salinity stress in seed germination on isabgol (PLANTAGO OVATA) 4:15–22
Islam F, Yasmeen T, Arif MS et al (2015) (2015) Plant growth promoting bacteria confer salt tolerance in Vigna radiata by up-regulating antioxidant defense and biological soil fertility. Plant Growth Regul 801(80):23–36. https://doi.org/10.1007/S10725-015-0142-Y
Jha B, Gontia I, Hartmann A (2012) The roots of the halophyte Salicornia brachiata are a source of new halotolerant diazotrophic bacteria with plant growth-promoting potential. Plant Soil 356:265–277. https://doi.org/10.1007/s11104-011-0877-9
Kang SM, Khan AL, Waqas M et al (2014) Plant growth-promoting rhizobacteria reduce adverse effects of salinity and osmotic stress by regulating phytohormones and antioxidants in Cucumis sativus. J Plant Interact 9:673–682. https://doi.org/10.1080/17429145.2014.894587
Kaur G, Asthir B (2015) Proline: a key player in plant abiotic stress tolerance. Biol Plant 59:609–619
Kloepper JW (1981) Relationship of in vitro antibiosis of plant growth-promoting rhizobacteria to plant growth and the displacement of root Microflora. Phytopathology 71:1020. https://doi.org/10.1094/PHYTO-71-1020
Kohler J, Hernández JA, Caravaca F, Roldán A (2008) Plant-growth-promoting rhizobacteria and arbuscular mycorrhizal fungi modify alleviation biochemical mechanisms in water-stressed plants. Funct Plant Biol 35:141. https://doi.org/10.1071/FP07218
Kohler J, Hernández JA, Caravaca F, Roldán A (2009) Induction of antioxidant enzymes is involved in the greater effectiveness of a PGPR versus AM fungi with respect to increasing the tolerance of lettuce to severe salt stress. Environ Exp Bot 65:245–252. https://doi.org/10.1016/j.envexpbot.2008.09.008
Kumar M, Mishra S, Dixit V et al (2016a) Synergistic effect of Pseudomonas putida and Bacillus amyloliquefaciens ameliorates drought stress in chickpea (Cicer arietinum L.). Plant Signal Behav. https://doi.org/10.1080/15592324.2015.1071004
Kumar S, Stecher G, Tamura K (2016b) MEGA7: Molecular evolutionary genetics analysis Version 7.0 for Bigger Datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kumari S, Vaishnav A, Jain S et al (2015) Bacterial-mediated induction of systemic tolerance to salinity with expression of stress alleviating enzymes in soybean (Glycine max L. Merrill). J Plant Growth Regul 34:558–573. https://doi.org/10.1007/s00344-015-9490-0
Li T, Zhang J, Shen C et al (2019) 1-Aminocyclopropane-1-carboxylate: a novel and strong chemoattractant for the plant beneficial Rhizobacterium Pseudomonas putida UW4. Mol Plant Microbe Interact 32:750–759. https://doi.org/10.1094/MPMI-11-18-0317-R
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/METH.2001.1262
Lowry O, Schagger H, Cramer WA, Vonjagow G (1994) Protein measurement with the folin phenol reagent. Anal Biochem 217:220–230
Lugtenberg BJJ, Chin-A-Woeng TFC, Bloemberg GV (2002) Microbe-plant interactions: principles and mechanisms. Antonie Van Leeuwenhoek Int J Gen Mol Microbiol 81:373–383. https://doi.org/10.1023/A:1020596903142
Manchanda G, Garg N (2008) Salinity and its effects on the functional biology of legumes. Acta Physiol Plant 305(30):595–618. https://doi.org/10.1007/S11738-008-0173-3
McDonald S, Prenzler PD, Antolovich M, Robards K (2001) Phenolic content and antioxidant activity of olive extracts. Food Chem 73:73–84. https://doi.org/10.1016/S0308-8146(00)00288-0
Merchante C, Alonso JM, Stepanova AN (2013) Ethylene signaling: simple ligand, complex regulation. Curr Opin Plant Biol 16:554–560
Morgan PW, Drew MC (1997) Ethylene and plant responses to stress. Physiol Plant 100:620–630. https://doi.org/10.1111/J.1399-3054.1997.TB03068.X
Mukhtar T, Rehman S, Smith D et al (2020) Mitigation of heat stress in Solanum lycopersicum L. by ACC-deaminase and exopolysaccharide producing Bacillus cereus: effects on biochemical profiling. Sustain 12:2159. https://doi.org/10.3390/SU12062159
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Mustafa G, Akhtar MS, Abdullah R (2019) Global concern for salinity on various agro-ecosystems. Salt Stress Microbes Plant Interact Causes Solut 1:1–19. https://doi.org/10.1007/978-981-13-8801-9_1
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in Spinach Chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nascimento FX, Rossi MJ, Soares CRFS et al (2014) New insights into 1-Aminocyclopropane-1-Carboxylate (ACC) deaminase phylogeny, evolution and ecological significance. PLoS ONE. https://doi.org/10.1371/journal.pone.0099168
Nautiyal CS (1997) Rhizosphere competence of Pseudomonas sp. NBRI9926 and Rhizobium sp. NBRI9513 involved in the suppression of chickpea (Cicer arietinum L.) pathogenic fungi. FEMS Microbiol Ecol 23:145–158. https://doi.org/10.1111/J.1574-6941.1997.TB00398.X
Nelson N (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Niranjan Raj S, Deepak SA, Basavaraju P et al (2003) Comparative performance of formulations of plant growth promoting rhizobacteria in growth promotion and suppression of downy mildew in pearl millet. Crop Prot 22:579–588. https://doi.org/10.1016/S0261-2194(02)00222-3
Noumavo PA, Kochoni E, Didagbé YO et al (2013) Effect of different plant growth promoting rhizobacteria on maize seed germination and seedling development. Am J Plant Sci 04:1013–1021. https://doi.org/10.4236/ajps.2013.45125
Numan M, Bashir S, Khan Y et al (2018) Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: a review. Microbiol Res 209:21–32. https://doi.org/10.1016/J.MICRES.2018.02.003
Osman AS, Rady MM (2015) Ameliorative effects of sulphur and humic acid on the growth, anti-oxidant levels, and yields of pea (Pisum sativum L.) plants grown in reclaimed saline soil. J Hortic Sci Biotechnol 87:626–632. https://doi.org/10.1080/14620316.2012.11512922
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Penrose DM, Glick BR (2003) Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol Plant 118:10–15
Pitman MG, Läuchli A (2002) Global impact of salinity and agricultural ecosystems. Salin Environ Plants Mol. https://doi.org/10.1007/0-306-48155-3_1
Porcel R, Ruiz-Lozano JM (2004) Arbuscular mycorrhizal influence on leaf water potential, solute accumulation, and oxidative stress in soybean plants subjected to drought stress. J Exp Bot 55:1743–1750. https://doi.org/10.1093/jxb/erh188
Pourbabaee AA, Bahmani E, Alikhani HA, Emami S (2016) Promotion of wheat growth under salt stress by halotolerant bacteria containing ACC deaminase. J Agric Sci Technol 18:855–864
Ravanbakhsh M, Sasidharan R, Voesenek LACJ et al (2017) ACC deaminase-producing rhizosphere bacteria modulate plant responses to flooding. J Ecol 105:979–986. https://doi.org/10.1111/1365-2745.12721
Roy DG (2019) Scholarship @ Western A new algorithm for primer design
Saikia J, Sarma RK, Dhandia R et al (2018) (2018) Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci Rep 81(8):1–16. https://doi.org/10.1038/s41598-018-21921-w
Sapre S, Gontia-Mishra I, Tiwari S (2019) ACC deaminase-producing bacteria: a key player in alleviating abiotic stresses in plants. Plant Growth Promot Rhizobacteria Agric Sustain. https://doi.org/10.1007/978-981-13-7553-8_14
Saravanakumar D, Samiyappan R (2007) ACC deaminase from Pseudomonas fluorescens mediated saline resistance in groundnut (Arachis hypogea) plants. J Appl Microbiol 102:1283–1292. https://doi.org/10.1111/j.1365-2672.2006.03179.x
Shailendra Singh GG (2015) Plant growth promoting rhizobacteria (PGPR): current and future prospects for development of sustainable agriculture. J Microb Biochem Technol 07:96–102. https://doi.org/10.4172/1948-5948.1000188
Siddikee MA, Chauhan PS (2011) Sa T (2011) Regulation of ethylene biosynthesis under salt stress in red pepper (Capsicum annuum L.) by 1-Aminocyclopropane-1-Carboxylic Acid (ACC) Deaminase-producing Halotolerant Bacteria. J Plant Growth Regul 312(31):265–272. https://doi.org/10.1007/S00344-011-9236-6
Singh M, Srivastava PK, Verma PC et al (2015) Soil fungi for mycoremediation of arsenic pollution in agriculture soils. J Appl Microbiol 119:1278–1290. https://doi.org/10.1111/jam.12948
Sinha AK, Jaggi M, Raghuram B, Tuteja N (2011) Mitogen-activated protein kinase signaling in plants under abiotic stress. Plant Signal Behav 6:196–203. https://doi.org/10.4161/psb.6.2.14701
Stearns JC, Glick BR (2003) Transgenic plants with altered ethylene biosynthesis or perception. Biotechnol Adv 21:193–210. https://doi.org/10.1016/S0734-9750(03)00024-7
Sun Y, Cheng Z, Glick BR (2009) The presence of a 1-aminocyclopropane-1-carboxylate (ACC) deaminase deletion mutation alters the physiology of the endophytic plant growth-promoting bacterium Burkholderia phytofirmans PsJN. FEMS Microbiol Lett 296:131–136. https://doi.org/10.1111/j.1574-6968.2009.01625.x
Talaat NB (2019) Role of reactive oxygen species signaling in plant growth and development. Reactive oxygen, Nitrogen and sulfur species in plants. Wiley, New York, pp 225–266
Tiwari G, Duraivadivel P, Sharma S, Hariprasad P (2018) 1-Aminocyclopropane-1-carboxylic acid deaminase producing beneficial rhizobacteria ameliorate the biomass characters of Panicum maximum Jacq. by mitigating drought and salt stress. Sci Rep. https://doi.org/10.1038/s41598-018-35565-3
Tuteja N (2007) Mechanisms of high salinity tolerance in plants. Methods in Enzymology. Academic Press Inc, New York, pp 419–438
Vardharajula S, Zulfikar Ali S, Grover M et al (2011) Drought-tolerant plant growth promoting Bacillus spp.: effect on growth, osmolytes, and antioxidant status of maize under drought stress. J Plant Interact 6:1–14. https://doi.org/10.1080/17429145.2010.535178
Wang Y, Mopper S, Hasenstein KH (2001) Effects of salinity on endogenous ABA, IAA, JA, and SA in Iris hexagona. J Chem Ecol 27:327–342. https://doi.org/10.1023/A:1005632506230
Wang Q, Dodd IC, Belimov AA et al (2016) Rhizosphere bacteria containing 1-aminocyclopropane-1-carboxylate deaminase increase growth and photosynthesis of pea plants under salt stress by limiting Na+ accumulation. Funct Plant Biol 43:161–172. https://doi.org/10.1071/FP15200
Yang J, Kloepper JW, Ryu CM (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14:1–4
Acknowledgements
The authors are grateful to Integral University for providing MCN number IU/R&D/2020-MCN001003 and DST-FIST for providing Financial Assistance to the Department of Bioscience, Integral University, Lucknow, Uttar Pradesh, India.
Funding
This work was supported by Uttar Pradesh Council of Science and Technology, Lucknow [UPCST/D-514]; Department of Science and Technology, New Delhi [DST INSPIRE-IF160803].
Author information
Authors and Affiliations
Contributions
NP and SS conceived and coordinated the research. AG conducted the experiments and analyzed the data. AG and MK wrote the manuscript. AB, SR, JA, read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gupta, A., Bano, A., Rai, S. et al. ACC deaminase producing plant growth promoting rhizobacteria enhance salinity stress tolerance in Pisum sativum. 3 Biotech 11, 514 (2021). https://doi.org/10.1007/s13205-021-03047-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-03047-5