Abstract
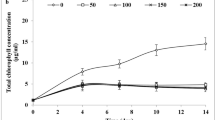
In this study, we aimed to investigate the taxonomy and various characteristics of Dunaliella salina IBSS-2 strain and describe its cultivation potential in mid-latitude climate during springtime. In addition, our analysis confirmed the essentiality of combining morphological, physiological, and other characteristics when identifying new species and strains of the genus Dunaliella, along with the molecular marker (internal transcribed spacer (ITS) of rDNA gene). The pilot cultivation of microalgae during the springtime in the south of Russia demonstrated that the climatic conditions of this region allow D. salina cultivation for biomass accumulation during this season, highlighting light and temperature conditions as the main factors determining the growth rate of D. salina. A two-fold increase in daily insolation and, consequently, in temperature in April resulted in a more than three-fold increase in productivity of D. salina culture. The maximum productivity of D. salina both in April and May was comparable and reached 2 g m−2 day−1, and the total yield for 8–10 days was about 14.5–16 g m−2. The additional CO2 supply into the D. salina culture did not show any significant effect on its growth rate; however, it contributed to maintaining the diversity of morphometric characteristics over a longer period of time. Changes in the morphological and morphometric characteristics of algal cells, including size reduction, were observed during the batch cultivation. Thus, the production potential of the green carotenogenic microalga D. salina was determined in the springtime, which allows expanding the seasonal interval of its cultivation in temperate latitudes.







Similar content being viewed by others
References
Absher M (1973) Hemocytometer counting. In: Kruse PF Jr, Patterson MK Jr (eds) Tissue culture: methods and applications. Academic Press, New York, pp 395–397. https://doi.org/10.1016/B978-0-12-427150-0.50098-X
Assunção P, Jaén-Molina R, Caujapé-Castells J, de la Jara A, Carmona L, Freijanes K, Mendoza H (2012) Molecular taxonomy of Dunaliella (Chlorophyceae), with a special focus on D. salina: ITS2 sequences revisited with an extensive geographical sampling. Aquat Biosyst 8:2. https://doi.org/10.1186/2046-9063-8-2
Avron M, Ben-Amotz A (eds) (1992) Dunaliella: physiology, biochemistry, and biotechnology. CRC Press, Boca Raton
Ben-Amotz A (1987) Effect of irradiance and nutrient deficiency on the chemical composition of Dunaliella bardawil Ben-Amotz and Avron (Volvocales, Chlorophyta). J Plant Physiol 131(5):479–487. https://doi.org/10.1016/S0176-1617(87)80290-0
Ben-Amotz A (1995) New mode of Dunaliella biotechnology: two-phase growth for β-carotene production. J Appl Phycol 7(1):65–68. https://doi.org/10.1007/BF00003552
Ben-Amotz A (2019) Bioactive compounds: glycerol production, carotenoid production, fatty acids production. In: Ben-Amotz A, Polle JEW, Subba Rao DW (eds) The Alga Dunaliella, biodiversity, physiology, genomics and biotechnology. CRC Press, Boca Raton, pp 189–207
Ben-Amotz A, Shaish A, Avron M (1991) The biotechnology of cultivating Dunaliella for production of β-carotene rich algae. Bioresour Technol 38(2–3):233–235. https://doi.org/10.1016/0960-8524(91)90160-L
Ben-Amotz A, Polle J, Rao DS (2009) The alga Dunaliella: biodiversity, physiology, genomics and biotechnology. Science Publishers, Enfield, p 555
Borovkov AB, Gudvilovich IN, Memetshaeva OA, Avsiyan AL, Lelekov AS, Novikova TM (2019) Morphological and morphometrical features in Dunaliella salina (Chlamydomonadales, Dunaliellaceae) during the two-phase cultivation mode. Ecologica Montenegrina 22:157–165. https://doi.org/10.37828/em.2019.22.12
Borovkov AB, Gudvilovich IN, Avsiyan AL (2020a) Scale-up of Dunaliella salina cultivation: from strain selection to open ponds. J Appl Phycol 32:1545–1558. https://doi.org/10.1007/s10811-020-02104-5
Borovkov AB, Gudvilovich IN, Avsiyan AL, Memetshaeva OA, Lelekov AS, Novikova TM (2020b) Production characteristics of Dunaliella salina at two-phase pilot cultivation (Crimea). Turk J Fish Aquat Sc 20(5):401–408. https://doi.org/10.4194/1303-2712-v20_5_08
Borowitzka MA, Siva CJ (2007) The taxonomy of the genus Dunaliella (Chlorophyta, Dunaliellales) with emphasis on the marine and halophilic species. J Appl Phycol 19(5):567–590. https://doi.org/10.1007/s10811-007-9171-x
Borowitzka MA, Vonshak A (2017) Scaling up microalgal cultures to commercial scale. Eur J Phycol 52(4):407–418. https://doi.org/10.1080/09670262.2017.1365177
Chekushkin AA, Gudvilovich IN, Lelekov AS (2019) Production characteristics of Spirulina platensis and Dunaliella salina cultures in the Sevastopol region at the off-season. Issues Mod Algol 19:96–104. https://doi.org/10.33624/2311-0147-2019-1(19)-96-104(in Russian)
Chekushkin AA, Lelekov AS, Gevorgiz RG (2020) Seasonal dynamics of ultimate productivity in horizontal photobioreactor. Russ J Biol Phys 5(3):405–411
Chen Y, Wang C, Xu C (2020) Nutritional evaluation of two marine microalgae as feedstock for aquafeed. Aquac Res 51:946–956. https://doi.org/10.1111/are.14439
da Silva MROB, Moura YAS, Converti A, Porto ALF, Marques DDAV, Bezerra RP (2021) Assessment of the potential of Dunaliella microalgae for different biotechnological applications: a systematic review. Algal Res 58:102396. https://doi.org/10.1016/j.algal.2021.102396
Darriba D, Taboada GL, Doallo R, Posada D (2012) JModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
Del Campo JA, García-González M, Guerrero MG (2007) Outdoor cultivation of microalgae for carotenoid production: current state and perspectives. Appl Microbiol Biotechnol 74(6):1163–1174. https://doi.org/10.1007/s00253-007-0844-9
Díaz JP, Inostroza C, Acién FG (2021) Scale-up of a Fibonacci-type photobioreactor for the production of Dunaliella salina. Appl Biochem Biotech 193:88–204. https://doi.org/10.1007/s12010-020-03410-x
Edmundson SJ, Huesemann MH (2015) The dark side of algae cultivation: characterizing night biomass loss in three photosynthetic algae, Chlorella sorokiniana, Nannochloropsis salina and Picochlorum sp. Algal Res 12:470–476. https://doi.org/10.1016/j.algal.2015.10.012
Emami K, Hack E, Nelson A, Brain CM, Lyne FM, Mesbahi E, Day JG, Caldwell GS (2015) Proteomic-based biotyping reveals hidden diversity within a microalgae culture collection: an example using Dunaliella. Sci Rep. UK 5(1):1–15. https://doi.org/10.1038/srep10036
García-González M, Moreno J, Cañavate JP, Anguis V, Prieto A, Manzano C, Florencio FJ, Guerrero MG (2003) Conditions for open-air outdoor culture of Dunaliella salina in southern Spain. J Appl Phycol 15:177–184. https://doi.org/10.1023/A:1023892520443
Griffiths MJ, Garcin C, van Hille RP, Harrison ST (2011) Interference by pigment in the estimation of microalgal biomass concentration by optical density. J Microbiol Meth 85(2):119–123. https://doi.org/10.1016/j.mimet.2011.02.005
Gudvilovich IN, Borovkov AB (2019) Testing of two-stage cultivation of Dunaliella salina (Teodoresco, 1905) in the Sevastopol region. South Russia Ecol Dev. 14(2):211–220. https://doi.org/10.18470/1992-1098-2019-2-211-220 (in Russian)
Highfield A, Ward A, Pipe R, Schroeder DC (2021) Molecular and phylogenetic analysis reveals new diversity of Dunaliella salina from hypersaline environments. J Mar Biol Assoc U K. https://doi.org/10.1017/S0025315420001319
Khadim SR, Singh P, Singh AK, Tiwari A, Mohanta A, Asthana RK (2018) Mass cultivation of Dunaliella salina in a flat plate photobioreactor and its effective harvesting. Bioresour Technol 270:20–29. https://doi.org/10.1016/j.biortech.2018.08.071
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. https://doi.org/10.1093/molbev/msy096
Lamers PP, van de Laak CC, Kaasenbrood PS, Lorier J, Janssen M, De Vos RC, Wijffels RH et al (2010) Carotenoid and fatty acid metabolism in light-stressed Dunaliella salina. Biotechnol Bioeng 106(4):638–648. https://doi.org/10.1002/bit.22725
Lelekov AS, Trenkenshu RP (2007) Simplest models of microalgae growth 4. Exponential and linear growth phases of microalgae culture. Ekologiya Morya 74:47–49 (in Russian)
Leliaert F, Verbruggen H, Vanormelingen P, Steen F, López-Bautista JM, Zuccarello GC, De Clerck O (2014) DNA-based species delimitation in algae. Eur J Phycol 49:179–196. https://doi.org/10.1080/09670262.2014.904524
Massyuk NP (1973) Morphology, taxonomy, ecology, geographical distribution of the genus Dunaliella Teod. and perspective for its practical use, 1st edn. Nauk. Dumka Publ., Kiev, p 487 (in Russian)
Morgulis A, Coulouris G, Raytselis Y, Madden TL, Agarwala R, Schäffer AA (2008) Database indexing for production MegaBLAST searches. Bioinformatics 24(16):1757–1764. https://doi.org/10.1093/bioinformatics/btn322
NASA Langley Research Center (LaRC) POWER Project (2021) https://power.larc.nasa.gov/data-access-viewer/. Accessed 06 Mar 2021
Olmos J, Paniagua J, Contreras R (2000) Molecular identification of Dunaliella sp. utilizing the 18S rDNA gene. Lett Appl Microbiol 30:80–84. https://doi.org/10.1046/j.1472-765x.2000.00672.x
Olmos J, Ochoa L, Paniagua-Michel J, Contreras R (2009) DNA fingerprinting differentiation between -carotene hyperproducer strains of Dunaliella from around the world. Saline Syst. https://doi.org/10.1186/1746-1448-5-5
Oren A (2005) A hundred years of Dunaliella research: 1905–2005. Saline Syst 1(1):2. https://doi.org/10.1186/1746-1448-1-2
Polle JEW, Jin ES, Ben-Amotz A (2020) The alga Dunaliella revisited: Looking back and moving forward with model and production organisms. Algal Res 49:01948. https://doi.org/10.1016/j.algal.2020.101948
Pomroy AJ (1989) Scanning electron microscopy of Heterocapsa minima sp. nov. (Dinophyceae) and its seasonal distribution in the Celtic Sea. Br Phycol J 24(2):131–135. https://doi.org/10.1080/00071618900650121
Preetha K, John L, Subin CS, Vijayan KK (2012) Phenotypic and genetic characterization of Dunaliella (Chlorophyta) from Indian salinas and their diversity. Aquat Biosyst 8:27. https://doi.org/10.1186/2046-9063-8-27
Science4all (2009) Microscopy and photography. http://science4all.nl/?Microscopy_and_Photography. Accessed 05 Dec 2021
Shaish A, Avron M, Ben-Amotz A (1990) Effect of inhibitors on the formation of stereoisomers in the biosynthesis of β-carotene in Dunaliella bardawil. Plant Cell Physiol 31(5):689–696. https://doi.org/10.1093/oxfordjournals.pcp.a077964
Sui Y, Muys M, Vermeir P, D’Adamo S, Vlaeminck SE (2019) Light regime and growth phase affect the microalgal production of protein quantity and quality with Dunaliella salina. Bioresour Technol 275:145–152. https://doi.org/10.1016/j.biortech.2018.12.046
Sun J, Liu D (2003) Geometric models for calculating cell biovolume and surface area for phytoplankton. J Plankton Res 25(11):1331–1346. https://doi.org/10.1093/plankt/fbg096
Supamattaya K, Kiriratnikom S, Boonyaratpalin M, Borowitzka L (2005) Effect of a Dunaliella extract on growth performance, health condition, immune response and disease resistance in black tiger shrimp (Penaeus monodon). Aquaculture 248(1–4):207–216. https://doi.org/10.1016/j.aquaculture.2020.735562
Terez EI, Terez GA, Kozak AV, Kuzmin SV, Dolgii SO (2012) The study of atmospheric optical parameters according to multiyear photometric observations of the sun in Crimea. Bull Crime Astrophys Obs 108(1):146–157. https://doi.org/10.3103/S0190271712010214
Wolf L, Cummings T, Müller K, Reppke M, Volkmar M, Weuster-Botz D (2021) Production of β-carotene with Dunaliella salina CCAP19/18 at physically simulated outdoor conditions. Eng Life Sci 21(3–4):115–125. https://doi.org/10.1002/elsc.202000044
Wu Z, Dejtisakdi W, Kermanee P, Ma C, Arirob W, Sathasivam R, Juntawong N (2017) Outdoor cultivation of Dunaliella salina KU 11 using brine and saline lake water with raceway ponds in northeastern Thailand. Biotechnol Appl Biochem 64(6):938–943. https://doi.org/10.1002/bab.1537
Xu Y, Ibrahim IM, Harvey PJ (2016) The influence of photoperiod and light intensity on the growth and photosynthesis of Dunaliella salina (Chlorophyta) CCAP 19/30. Plant Physiol Biochem 106:305–315. https://doi.org/10.1016/j.plaphy.2016.05.021
Zhang Z, Schwartz S, Wagner L, Miller W (2000) A greedy algorithm for aligning DNA sequences. J Comput Biol 7(1–2):203–214. https://doi.org/10.1089/10665270050081478
Acknowledgements
This work was supported by A.O. Kovalevsky Institute of Biology of the Southern Seas of RAS, governmental research assignment # 121030300149-0 (AAAA-A18-118021350003-6) and an internal grant of Sevastopol State University for 2021 No. 30/06-31.
Author information
Authors and Affiliations
Contributions
ABB conceptualization, writing—review and editing, supervision. ING methodology, formal analysis, writing—original draft. ALA investigation, formal analysis, writing—review and editing. AOL methodology, formal analysis, writing—original draft. OAR methodology, investigation. OAM formal analysis, investigation. IVD investigation. AAC investigation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Rights and permissions
About this article
Cite this article
Borovkov, A.B., Gudvilovich, I.N., Avsiyan, A.L. et al. Productivity and morphometric parameters of the microalga Dunaliella salina IBSS-2 under pilot cultivation in continental mid-latitude climate in spring. 3 Biotech 11, 438 (2021). https://doi.org/10.1007/s13205-021-02982-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02982-7