Abstract
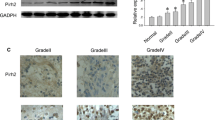
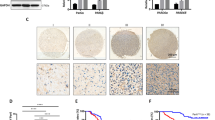
Glioma is the most common primary brain tumor in adults with an adverse prognosis and obscure pathogenesis. PICALM interacting mitotic regulator protein (PIMREG) functions as an oncogene in multiple types of cancer, but its function in glioma remains unknown. The Gene Expression Profiling Interactive Analysis 2 (GEPIA2, http://gepia2.cancer-pku.cn/#index) showed that PIMREG expression in the glioma tissues was higher than that in normal brain tissues. Herein, cell counting kit-8 assay and flow cytometry analysis exhibited that overexpression of PIMREG significantly promoted the proliferation of glioma cells and the transition from G1 phase of the cell cycle to S phase. Wound-healing and transwell assays showed that overexpression of PIMREG markedly enhanced the migration and invasion of glioma cells. Western blot analysis revealed that overexpression of PIMREG increased the expression of cyclin D1, cyclin E, Vimentin, matrix metalloproteinase (MMP)-2, and MMP-9, but reduced the expression of E-cadherin. In addition, overexpression of PIMREG activated the β-catenin signaling pathway, as evidenced by the increased total and nuclear expression of β-catenin and the up-regulated expression of its downstream target c-myc. Furthermore, immunofluorescence staining further indicated the increased nuclear translocation of β-catenin in PIMREG-overexpressing cells. However, knockdown of PIMREG exerted opposite effects on glioma cells. Blockade of the β-catenin signaling by ICG-001 markedly impeded the promoting effects of PIMREG on glioma cell proliferation and invasion. In conclusion, PIMREG acts as a tumor promoter in glioma at least partly via activating the β-catenin signaling pathway. This study provides new insights into the molecular mechanism for glioma pathogenesis and treatment.





Similar content being viewed by others
Availability of data and materials
The data analyzed in the present study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Arato-Ohshima T, Sawa H (1999) Over-expression of cyclin D1 induces glioma invasion by increasing matrix metalloproteinase activity and cell motility. Int J Cancer 83:387–392. https://doi.org/10.1002/(sici)1097-0215(19991029)83:3%3c387::aid-ijc15%3e3.0.co;2-o
Archangelo LF et al (2008) The CALM and CALM/AF10 interactor CATS is a marker for proliferation. Mole Oncol 2:356–367. https://doi.org/10.1016/j.molonc.2008.08.001
Carro MS et al (2010) The transcriptional network for mesenchymal transformation of brain tumours. Nature 463:318–325. https://doi.org/10.1038/nature08712
Chen R, Smith-Cohn M, Cohen AL, Colman H (2017) Glioma subclassifications and their clinical significance. Neurotherapeutics 14:284–297. https://doi.org/10.1007/s13311-017-0519-x
Chen Y, Yuan S, Ning T, Xu H, Guan B (2020) SNHG7 facilitates glioblastoma progression by functioning as a molecular sponge for MicroRNA-449b-5p and thereby increasing MYCN expression. Technol Cancer Res Treat 19:1533033820945802. https://doi.org/10.1177/1533033820945802
Dongre A, Weinberg RA (2019) New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol 20:69–84. https://doi.org/10.1038/s41580-018-0080-4
Edwards LA et al (2011) Effect of brain- and tumor-derived connective tissue growth factor on glioma invasion. J Natl Cancer Inst 103:1162–1178. https://doi.org/10.1093/jnci/djr224
Fraczek M, Wozniak Z, Ramsey D, Zatonski T, Krecicki T (2008) Clinicopathologic significance and prognostic role of cyclin E and cyclin A expression in laryngeal epithelial lesions. Acta Otolaryngol 128:329–334. https://doi.org/10.1080/00016480701487742
Gautschi O, Ratschiller D, Gugger M, Betticher DC, Heighway J (2007) Cyclin D1 in non-small cell lung cancer: a key driver of malignant transformation. Lung Cancer 55:1–14. https://doi.org/10.1016/j.lungcan.2006.09.024
Gusyatiner O, Hegi ME (2018) Glioma epigenetics: From subclassification to novel treatment options. Semin Cancer Biol 51:50–58. https://doi.org/10.1016/j.semcancer.2017.11.010
Hashimoto K et al (2017) Fam64a is a novel cell cycle promoter of hypoxic fetal cardiomyocytes in mice. Sci Rep 7:4486. https://doi.org/10.1038/s41598-017-04823-1
He L, Zhou H, Zeng Z, Yao H, Jiang W, Qu H (2019) Wnt/β-catenin signaling cascade: a promising target for glioma therapy. J Cell Physiol 234:2217–2228. https://doi.org/10.1002/jcp.27186
Hirakawa T, Nasu K, Miyabe S, Kouji H, Katoh A, Uemura N, Narahara H (2019) β-catenin signaling inhibitors ICG-001 and C-82 improve fibrosis in preclinical models of endometriosis. Sci Rep 9:20056. https://doi.org/10.1038/s41598-019-56302-4
Hui W, Yuntao L, Lun L, WenSheng L, ChaoFeng L, HaiYong H, Yueyang B (2013) MicroRNA-195 inhibits the proliferation of human glioma cells by directly targeting cyclin D1 and cyclin E1. PLoS ONE 8:e54932. https://doi.org/10.1371/journal.pone.0054932
Huo XY, Zhang XY, Yuan F, Zhao XY, You BA (2019) HOXB7 promotes proliferation and metastasis of glioma by regulating the Wnt/β-catenin pathway. Eur Rev Med Pharmacol Sci 23:2476–2485. https://doi.org/10.26355/eurrev_201903_17395
Jiang L et al (2019) Overexpression of PIMREG promotes breast cancer aggressiveness via constitutive activation of NF-κB signaling. EBioMedicine 43:188–200. https://doi.org/10.1016/j.ebiom.2019.04.001
Jiang Y, Zhou C, Gao Q, Yin ZQ, Wang J, Mu H, Yan J (2020a) FAM64A promotes osteosarcoma cell growth and metastasis and is mediated by miR-493. J Oncol 2020:2518297. https://doi.org/10.1155/2020/2518297
Jiang ZM, Li HB, Chen SG (2020b) PIMREG, a marker of proliferation, facilitates aggressive development of cholangiocarcinoma cells partly through regulating cell cycle-related markers. Technol Cancer Res Treat 19:1533033820979681. https://doi.org/10.1177/1533033820979681
Jiao Y, Fu Z, Li Y, Zhang W, Liu Y (2019) Aberrant FAM64A mRNA expression is an independent predictor of poor survival in pancreatic cancer. PLoS ONE 14:e0211291. https://doi.org/10.1371/journal.pone.0211291
Johnson DG, Walker CL (1999) Cyclins and cell cycle checkpoints. Annu Rev Pharmacol Toxicol 39:295–312. https://doi.org/10.1146/annurev.pharmtox.39.1.295
Kahlert UD, Nikkhah G, Maciaczyk J (2013) Epithelial-to-mesenchymal(-like) transition as a relevant molecular event in malignant gliomas. Cancer Lett 331:131–138. https://doi.org/10.1016/j.canlet.2012.12.010
Lapointe S, Perry A, Butowski NA (2018) Primary brain tumours in adults. Lancet 392:432–446. https://doi.org/10.1016/s0140-6736(18)30990-5
Lee W et al (2017) Epimedium koreanum Nakai inhibits PMA-induced cancer cell migration and invasion by modulating NF-κB/MMP-9 signaling in monomorphic malignant human glioma cells. Oncol Rep 38:3619–3631. https://doi.org/10.3892/or.2017.6043
Ma H, Nguyen C, Lee KS, Kahn M (2005) Differential roles for the coactivators CBP and p300 on TCF/beta-catenin-mediated survivin gene expression. Oncogene 24:3619–3631. https://doi.org/10.1038/sj.onc.1208433
Meng Y, Shang FR, Zhu YL (2019) MiR-491 functions as a tumor suppressor through Wnt3a/β-catenin signaling in the development of glioma. Eur Rev Med Pharmacol Sci 23:10899–10907. https://doi.org/10.26355/eurrev_201912_19793
Nieto MA, Huang RY, Jackson RA, Thiery JP (2016) EMT: 2016. Cell 166:21–45. https://doi.org/10.1016/j.cell.2016.06.028
Noh MG et al (2017) Prognostic significance of E-cadherin and N-cadherin expression in gliomas. BMC Cancer 17:583. https://doi.org/10.1186/s12885-017-3591-z
Ostrom QT et al (2014) The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol 16:896–913. https://doi.org/10.1093/neuonc/nou087
Pang W, Li Y, Guo W, Shen H (2020) Cyclin E: a potential treatment target to reverse cancer chemoresistance by regulating the cell cycle. Am J Transl Res 12:5170–5187
Scharpenseel H et al (2019) EGFR and HER3 expression in circulating tumor cells and tumor tissue from non-small cell lung cancer patients. Sci Rep 9:7406. https://doi.org/10.1038/s41598-019-43678-6
Shi F, Shi Z, Zhao Y, Tian J (2019) CircRNA hsa-circ-0014359 promotes glioma progression by regulating miR-153/PI3K signaling. Biochem Biophys Res Commun 510:614–620. https://doi.org/10.1016/j.bbrc.2019.02.019
Sun Y, Peng X, Li Y, Ma H, Li D, Shi X (2019) The effects of histamine H1 type receptor (H1R) in regulating osteoblastic cell differentiation and mineralization. Artif Cell Nanomed B 47:1281–1287. https://doi.org/10.1080/21691401.2019.1596924
Tang Z, Kang B, Li C, Chen T, Zhang Z (2019) GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res 47:W556–W560. https://doi.org/10.1093/nar/gkz430
Wang Z, Zhang S, Siu TL, Huang S (2015) Glioblastoma multiforme formation and EMT: role of FoxM1 transcription factor. Curr Pharm Des 21:1268–1271. https://doi.org/10.2174/1381612821666141211115949
Wang Y et al (2017) β-catenin-mediated YAP signaling promotes human glioma growth. J Exp Clin Cancer Res 36:136. https://doi.org/10.1186/s13046-017-0606-1
Wesseling P, Capper D (2018) WHO 2016 classification of gliomas. Neuropathol Appl Neurobiol 44:139–150. https://doi.org/10.1111/nan.12432
Xu H et al (2019a) Mesenchymal stem cell-derived exosomal microRNA-133b suppresses glioma progression via Wnt/β-catenin signaling pathway by targeting EZH2. Stem Cell Res Ther 10:381. https://doi.org/10.1186/s13287-019-1446-z
Xu ZS et al (2019b) FAM64A positively regulates STAT3 activity to promote Th17 differentiation and colitis-associated carcinogenesis. Proc Natl Acad Sci USA 116:10447–10452. https://doi.org/10.1073/pnas.1814336116
Yao Z et al (2019) Knockdown of FAM64A suppresses proliferation and migration of breast cancer cells. Breast Cancer 26:835–845. https://doi.org/10.1007/s12282-019-00991-2
Zhang J et al (2018) LGR5, a novel functional glioma stem cell marker, promotes EMT by activating the Wnt/β-catenin pathway and predicts poor survival of glioma patients. J Exp Clin Cancer Res 37:225. https://doi.org/10.1186/s13046-018-0864-6
Zhang J et al (2019) Up-regulation of FAM64A promotes epithelial-to-mesenchymal transition and enhances stemness features in breast cancer cells. Biochem Biophys Res Commun 513:472–478. https://doi.org/10.1016/j.bbrc.2019.03.207
Zhao WM, Coppinger JA, Seki A, Cheng XL, Yates JR 3rd, Fang G (2008) RCS1, a substrate of APC/C, controls the metaphase to anaphase transition. Proc Natl Acad Sci USA 105:13415–13420. https://doi.org/10.1073/pnas.0709227105
Zhao XF, Yang YS, Park YK (2020) HOXC9 overexpression is associated with gastric cancer progression and a prognostic marker for poor survival in gastric cancer patients. Int J Clin Oncol 25:2044–2054. https://doi.org/10.1007/s10147-020-01772-0
Zhao W, Zhao J, Guo X, Feng Y, Zhang B, Tian L (2021) LncRNA MT1JP plays a protective role in intrahepatic cholangiocarcinoma by regulating miR-18a-5p/FBP1 axis. BMC Cancer 21:142. https://doi.org/10.1186/s12885-021-07838-0
Zhong D et al (2015) LRG1 modulates invasion and migration of glioma cell lines through TGF-beta signaling pathway. Acta Histochem 117:551–558. https://doi.org/10.1016/j.acthis.2015.05.001
Zhou Y, An H, Wu G (2020) MicroRNA-6071 suppresses glioblastoma progression through the inhibition of PI3K/AKT/mTOR pathway by binding to ULBP2. Onco Targets Ther 13:9429–9441. https://doi.org/10.2147/OTT.S265791
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: LZ; methodology: LZ and DW; formal analysis and investigation: DW, AH, and HP; writing—original draft preparation: DW; writing—review and editing: DW; supervision: DL.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Rights and permissions
About this article
Cite this article
Wang, D., Hu, A., Peng, H. et al. Tumor-promoting function of PIMREG in glioma by activating the β-catenin pathway. 3 Biotech 11, 380 (2021). https://doi.org/10.1007/s13205-021-02922-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02922-5