Abstract
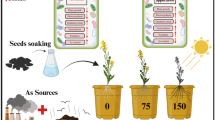
Depleting fossil fuels target plant weeds which have the potential to be converted into efficient biofuels. In this study, mimosa seeds were utilized as a substrate for bioethanol production. This investigation was divided into three parts: breaking dormancy of seeds, mimosa seeds germination, and bioethanol production from mimosa seeds. Seed dormancy breaking was initiated by seeds soaked in hot distilled water to analyze the sugar quantity. Sugar content was measured relevance with root length results. According to results, root length obtained revealed that at 0.5–1.0 cm for root size has the most sugar availability. It was revealed that the total sugar 548.21 g/L and reducing sugar has a concentration of 248.67 g/L. Therefore, the broken dormancy of seeds using hot water at 95 °C for 10 min with a root length of 0.5–1 cm was used for ethanol fermentation. Ethanol fermentation was done by free yeast cell and immobilized yeast by injecting yeast directly. The ethanol yield was measured on the 3rd day of every fermentation. Results showed that the free cell yeast during the 1st day of fermentation afforded an ethanol production of 57.574 g/L, while the yield for immobilized yeast was 60.714 g/L. Consequently, the ethanol yield on the 2nd day of fermentation from the directly injected immobilized yeast was 60.088 g/L. Results revealed that the immobilization of yeast cells in fermentation provided a higher probability for bioethanol yield and could be utilized as a baseline for future bioethanol production. Stimulation of natural enzymes by germination of seeds for enhanced bioethanol production will be a novel approach towards next-generation biofuels.






Similar content being viewed by others
References
Aggarangsi P, Tippayawong N, Moran JC, Rerkkriangkrai P (2013) Overview of livestock biogas technology development and implementation in Thailand Energy for Sustainable development. Energy Sustain Dev 17(4):371–377
Ashraf M, Foolad MR (2005) Pre-sowing seed treatment—A shotgun approach to improve germination, plant growth, and crop yield under saline and non-saline conditions. Adv Agron 88:223–271
Banos R, Manzano-Agugliaro F, Montoya FG, Gil C, Alcayde A, Gómez J (2011) Optimization methods applied to renewable and sustainable energy: A review. Renew Sustain Energy Rev 15(4):1753–1766
Bautista K, Unpaprom Y, Ramaraj R (2019) Bioethanol production from corn stalk juice using Saccharomyces cerevisiae TISTR 5020. Energy Sour Part A Recover, Util Environ Eff 41(13):1615–1621
Bhuyar P, Sathyavathi S, Math RK (2020a) Production of bioethanol from starchy tuber (Amorphophallus commutatus) and antimicrobial activity study of its extracts. Afr J Biol Sci 2(2):70–76
Bhuyar P, Yusoff MM, Rahim MHA, Sundararaju S, Maniam GP, Govindan N (2020b) Effect of plant hormones on the production of biomass and lipid extraction for biodiesel production from microalgae Chlorella sp. J Microbiol Biotechnol Food Sci 10(1):671–674
Brito AS, Pinto M, Araújo AV, Souza VN (2014) Superação de dormência em Mimosa ophthalmocentra Mart Ex Benth. Enciclopédia Biosfera 10(18):2792
Camargo-Ricalde SL, Dhillion SS, Grether R (2002) Community structure of endemic Mimosa species and environmental heterogeneity in a semi-arid Mexican valley. J Veg Sci 13(5):697–704
Chaiyapa W, Esteban M, Kameyama Y (2018) Why go green? Discourse analysis of motivations for Thailand’s oil and gas companies to invest in renewable energy. Energy Policy 120:448–459
Chau TL, Guillan A, Roca E, Nunez MJ, Lema JM (2000) Enhancement of plasmid stability and enzymatic expression by immobilizing recombinant Saccharomyces cerevisiae. Biotech Lett 22:1247–1250
Chen WM, Laevens S, Lee TM, Coenye T, De Vos P, Mergeay M, Vandamme P (2001) Ralstonia taiwanensis sp nov, isolated from root nodules of Mimosa species and sputum of a cystic fibrosis patient. Int J Syst Evol Microbiol 51(5):1729–1735
Darzins A, Pienkos P, Edye L (2010) Current status and potential for algal biofuels production Bioind Partnes NREL. In: Report prepared for the International Energy Agency, Bioenergy Task 39:131
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28(3):350–356
Fowler JAP, Bianchetti A (2000) Dormancy in forest seeds. Documentos-Embrapa Florestas 40:31
Grice AC (2006) The impacts of invasive plant species on the biodiversity of Australian rangelands. Rangel J 28(1):27–35
Herrero M, Laca A, Garcı́a LA, Dı́az M, (2001) Controlled malolactic fermentation in cider using Oenococcus oeni immobilized in alginate beads and comparison with free cell fermentation. Enzyme Microb Technol 28(1):35–41
Hsu CH, Chu YE, Argin-Soyal S, Hahm TS, Lo YM (2004) Effects of surface characteristics and xanthan polymers on the immobilization of Xanthomonas Campestris to fibrous matrices. J Food Sci 69(9):E441–E448
Hwang JH, Church J, Lee SJ, Park J, Lee WH (2016) Use of microalgae for advanced wastewater treatment and sustainable bioenergy generation. Environ Eng Sci 33(11):882–897
Kaewdiew J, Ramaraj R, Koonaphapdeelert S, Dussadee N (2019) Assessment of the biogas potential from agricultural waste in northern Thailand. Maejo Int J Energy Environ Commun 1(1):40–47
Khammee P, Ramaraj R, Whangchai N, Bhuyar P, Unpaprom Y (2020) The immobilization of yeast for fermentation of macroalgae Rhizoclonium sp for efficient conversion into bioethanol. Biomass Convers Biorefinery 11:827–835
Kourkoutas A, Kourkoutas A, Bekatorou A, Banat IM, Marchant R, Koutinas AA (2004) Immobilization technologies and support materials suitable in alcohol beverages production: a review. Food Microbiol 21:377–397
Manmai N, Siriboon T, Tipnee S, Unpaprom Y, Ramaraj R (2017) Bioconversion of lignocellulosic biomass, Sorghum bicolor L into bioethanol. In: Proceedings of academics World 73 rd International Conference. Taipei, Taiwan, 41–45
Melo JS, D’Souza SF (1999) Simultaneous filtration and immobilization of cells from a flowing suspension using a bioreactor containing polyethylenimine coated cotton threads: application in the continuous inversion of concentrated sucrose syrups. World J Microbiol Biotechnol 15(1):23–27
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Nguyen T, Unpaprom Y, Ramaraj R (2020) Enhanced fermentable sugar production from low grade and damaged longan fruits using cellulase with algal enzymes for bioethanol production. ELSR 6(2):26–31
Nikolić S, Mojović L, Pejin D, Rakin M, Vukašinović M (2010) Production of bioethanol from corn meal hydrolyzates by free and immobilized cells of Saccharomyces cerevisiae var ellipsoideus. Biomass Bioenerg 34(10):1449–1456
Ramaraj R, Tsai DDW, Chen PH (2014) An exploration of the relationships between microalgae biomass growth and related environmental variables. J Photochem Photobiol, B 135:44–47
Saengsawang B, Bhuyar P, Manmai N, Ponnusamy VK, Ramaraj R, Upaprom Y (2020) The optimization of oil extraction from macroalgae, Rhizoclonium sp by chemical methods for efficient conversion into biodiesel. Fuel 274:117841
Sasujit K, Dussadee N, Homdoung N, Ramaraj R, Kiatsiriroat T (2017) Waste-to-Energy: Producer gas production from fuel briquette of energy crop in Thailand. Int Energy J 17(1):37–45
Silveira FAO, Fernandes GW (2006) Effect of light, temperature and scarification on the germination of Mimosa foliolosa (Leguminosae) seeds. Seed Sci Technol 34(3):585–592
Silveira FA, Negreiros D, Fernandes GW (2004) Influência da luz e da temperatura na germinação de sementes de Marcetia taxifolia (A St-Hil) DC (Melastomataceae). Acta Bot Bras 18(4):847–851
Singh D, Nigam P, Banat IM, Marchant R, McHale AP (1998) Ethanol production at elevated temperatures and alcohol concentrations: Part II–Use of Kluyveromyces marxianus IMB3. World J Microbiol Biotechnol 14(6):823–834
Sperandio HV, Lopes JC, Matheus MT (2013) Superação de dormência em sementes de Mimosa setosa Benth. Comunicata Scientiae 4(4):385–390
Suganya T, Varman M, Masjuki HH, Renganathan S (2016) Macroalgae and microalgae as a potential source for commercial applications along with biofuels production: a biorefinery approach. Renew Sustain Energy Rev 55:909–941
Tsai DDW, Chen PH, Chou CM, Hsu CF, Ramaraj R (2015) Carbon sequestration by alga ecosystems. Ecol Eng 84:386–389
Verboven P, Flick D, Nicolai BM, Alvarez G (2006) Modeling transport phenomena in refrigerated food bulks, packages and stacks: basics and advances. Int J Refrig 29(6):985–997
Vu PT, Unpaprom Y, Ramaraj R (2017) Evaluation of bioethanol production from rice field weed biomass. Emergent Life Sci Res 3:42–49
Whangchai K, Inta W, Unpaprom Y, Bhuyar P, Adoonsook D, Ramaraj R (2021) Comparative analysis of fresh and dry free-floating aquatic plant Pistia stratiotes via chemical pretreatment for second-generation (2G) bioethanol production. Biores Technol Rep 14:100651
Williams D, Munnecke DM (1981) The production of ethanol by immobilized yeast cells. Biotechnol Bioeng 23(8):1813–1825
Yang ST, Shu CH (1996) Kinetics and stability of GM-CSF production by recombinant yeast cells immobilized in a fibrousbed bioreactor. Biotechnol Prog 12:449–456
Yang ST, Huang Y, Hong G (1995) A novel recycle batch immobilized cell bioreactor for propionate production from whey lactose. Biotechnol Bioeng 45:379–386
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ramaraj, R., Bhuyar, P., Intarod, K. et al. Stimulation of natural enzymes for germination of mimosa weed seeds to enhanced bioethanol production. 3 Biotech 11, 307 (2021). https://doi.org/10.1007/s13205-021-02859-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02859-9