Abstract
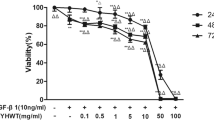
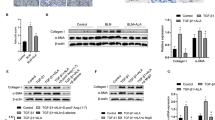
The purpose of this study was to confirm whether metformin can attenuate TGF-β1-induced pulmonary fibrosis through inhibition of transglutaminase 2 (TG2) and subsequent TGF-β pathways. In vitro, MTT assay and Annexin V-FITC/PI staining assay were performed to determine the effect of metformin on the proliferation and apoptosis of human fetal lung fibroblasts (HFL-1 cell). Protein expression of TG2, Collagen I (Col I) and α-smooth muscle actin (α-SMA) were determined by western blot. To further confirm the relationship between TG2 and the anti-fibrotic effect of metformin, TG2 siRNA and TG2 overexpression plasmid were used to interfere the expression of TG2. A bleomycin-induced pulmonary fibrosis model was employed to determine the in vivo inhibitory effect of metformin. The concentrations of TG2, both in supernatants of cells and serum of rats, were determined by ELISA assay. Our results showed that metformin concentration-dependently inhibited the proliferation and promoted the apoptosis of TGF-β1-stimulated HFL-1 cells. The protein expressions of TG2, Col I and α-SMA stimulated by TGF-β1 were decreased after metformin intervention, which was confirmed in both siRNAs and plasmids treatment conditions. In vivo, metformin attenuated bleomycin-induced pulmonary fibrosis as demonstrated by H&E and Masson staining, as well as the protein expressions of Col I and α-SMA. Besides, phosphorylated SMAD2, phosphorylated SMAD3, phosphorylated Akt and phosphorylated ERK1/2 were all significantly increased after bleomycin treatment and decreased to normal levels after metformin intervention. Taken together, our results demonstrated that metformin can attenuate TGF-β1-induced pulmonary fibrosis, at least partly, through inhibition of TG2 and subsequent TGF-β pathways.






Similar content being viewed by others
References
Agnihotri N, Mehta K (2017) Transglutaminase-2: evolution from pedestrian protein to a promising therapeutic target. Amino Acids 49(3):425–439. https://doi.org/10.1007/s00726-016-2320-2
Berschneider B, Ellwanger DC, Baarsma HA, Thiel C, Shimbori C, White ES, Kolb M, Neth P, Königshoff M (2014) miR-92a regulates TGF-β1-induced WISP1 expression in pulmonary fibrosis. Int J Biochem Cell B 53:432–441. https://doi.org/10.1016/j.biocel.2014.06.011
Bhedi CD, Nasirova S, Toksoz D, Warburton RR, Morine KJ, Kapur NK, Galper JB, Preston IR, Hill NS, Fanburg BL (2019) Glycolysis regulated transglutaminase 2 activation in cardiopulmonary fibrogenic remodeling. FASEB J. https://doi.org/10.1096/fj.201902155R
Chen H-H, Zhao P, Zhao W-X, Tian J, Guo W, Xu M, Zhang C, Lu R (2017) Stachydrine ameliorates pressure overload-induced diastolic heart failure by suppressing myocardial fibrosis. Am J Transl Res 9(9):4250
Heckman-Stoddard BM, DeCensi A, Sahasrabuddhe VV, Ford LG (2017) Repurposing metformin for the prevention of cancer and cancer recurrence. Diabetologia 60(9):1639–1647. https://doi.org/10.1007/s00125-017-4372-6
Hsu H-S, Liu C-C, Lin J-H, Hsu T-W, Hsu J-W, Su K, Hung S-C (2017) Involvement of ER stress, PI3K/AKT activation, and lung fibroblast proliferation in bleomycin-induced pulmonary fibrosis. Sci Rep 7(1):14272. https://doi.org/10.1038/s41598-017-14612-5
Jiang H-d, Guan H-s (2009) MS80, a novel sulfated oligosaccharide, inhibits pulmonary fibrosis by targeting TGF-β1 both in vitro and in vivo. Acta Pharmacol Sin 30(7):973. https://doi.org/10.1038/aps.2009.86
Kheirollahi V, Wasnick RM, Biasin V, Vazquez-Armendariz AI, Chu X, Moiseenko A, Weiss A, Wilhelm J, Zhang J-S, Kwapiszewska G (2019) Metformin induces lipogenic differentiation in myofibroblasts to reverse lung fibrosis. Nat Commun 10(1):1–16. https://doi.org/10.1038/s41467-019-10839-0
Lee S-S, Chen Y-J, Tsai C-H, Huang F-M, Chang Y-C (2016) Elevated transglutaminase-2 expression mediates fibrosis in areca quid chewing-associated oral submucocal fibrosis via reactive oxygen species generation. Clin Oral Investig 20(5):1029–1034. https://doi.org/10.1007/s00784-015-1579-0
Li L, Huang W, Li K, Zhang K, Lin C, Han R, Lu C, Wang Y, Chen H, Sun F (2015) Metformin attenuates gefitinib-induced exacerbation of pulmonary fibrosis by inhibition of TGF-β signaling pathway. Oncotarget 6(41):43605. https://doi.org/10.18632/oncotarget.6186
Liu Y-y, Shi Y, Liu Y, Pan X-h, Zhang K-x (2018) Telomere shortening activates TGF-β/Smads signaling in lungs and enhances both lipopolysaccharide and bleomycin-induced pulmonary fibrosis. Acta Pharmacol Sin 39(11):1735–1745. https://doi.org/10.1038/s41401-018-0007-9
Morales DR, Morris AD (2015) Metformin in cancer treatment and prevention. Annu Rev Med 66:17–29. https://doi.org/10.1146/annurev-med-062613-093128
Nesti L, Natali A (2017) Metformin effects on the heart and the cardiovascular system: a review of experimental and clinical data. Nutr Metab Cardiovas Dis 27(8):657–669. https://doi.org/10.1016/j.numecd.2017.04.009
Olsen KC, Sapinoro R, Kulkarni AA, Kottmann M, Ringo K, Strong E, Johnson GV, Phipps RP, Sime PJ (2010) Tissue transglutaminase promotes pulmonary fibrosis through multiple mechanisms including AKT activation. In: A58. Animal models of pulmonary fibrosis. American Thoracic Society, pp A1963–A1963. https://doi.org/10.1164/ajrccm-conference.2010.181.1_MeetingAbstracts.A1963
Olsen KC, Sapinoro RE, Kottmann R, Kulkarni AA, Iismaa SE, Johnson GV, Thatcher TH, Phipps RP, Sime PJ (2011) Transglutaminase 2 and its role in pulmonary fibrosis. Am J Resp Crit Care Med 184(6):699–707. https://doi.org/10.1164/rccm.201101-0013OC
Olsen KC, Epa AP, Kulkarni AA, Kottmann RM, McCarthy CE, Johnson GV, Thatcher TH, Phipps RP, Sime PJ (2014) Inhibition of transglutaminase 2, a novel target for pulmonary fibrosis, by two small electrophilic molecules. Am J Respir Cell Mol Biol 50(4):737–747. https://doi.org/10.1165/rcmb.2013-0092OC
Philp CJ, Siebeke I, Clements D, Miller S, Habgood A, John AE, Navaratnam V, Hubbard RB, Jenkins G, Johnson SR (2018) Extracellular matrix cross-linking enhances fibroblast growth and protects against matrix proteolysis in lung fibrosis. Am J Respir Cell Mol Biol 58(5):594–603. https://doi.org/10.1165/rcmb.2016-0379OC
Rangarajan S, Bone NB, Zmijewska AA, Jiang S, Park DW, Bernard K, Locy ML, Ravi S, Deshane J, Mannon RB (2018) Metformin reverses established lung fibrosis in a bleomycin model. Nat Med 24(8):1121. https://doi.org/10.1038/s41591-018-0087-6
Wang Z, Griffin M (2012) TG2, a novel extracellular protein with multiple functions. Amino Acids 42(2–3):939–949. https://doi.org/10.1007/s00726-011-1008-x
Wang M, Weng X, Guo J, Chen Z, Jiang G, Liu X (2016) Metformin alleviated EMT and fibrosis after renal ischemia–reperfusion injury in rats. Ren Fail 38(4):614–621. https://doi.org/10.3109/0886022X.2016.1149770
Wang K, Zu C, Zhang Y, Wang X, Huan X, Wang L (2020) Blocking TG2 attenuates bleomycin-induced pulmonary fibrosis in mice through inhibiting EMT. Respir Physiol Neurobiol 276:103402. https://doi.org/10.1016/j.resp.2020.103402
Xiao M, Li L, Li C, Zhang P, Hu Q, Ma L, Zhang H (2014) Role of autophagy and apoptosis in wound tissue of deep second-degree burn in rats. Acad Emerg Med 21(4):383–391. https://doi.org/10.1111/acem.12352
Zaghloul MS, Abdel-Salam RA, Said E, Suddek GM, Salem HA-R (2017) Attenuation of Bleomycin-induced pulmonary fibrosis in rats by flavocoxid treatment. Egyptian journal of basic and applied sciences 4(4):256–263. https://doi.org/10.1016/j.ejbas.2017.10.005
Zhang YE (2009) Non-Smad pathways in TGF-β signaling. Cell Res 19(1):128–139. https://doi.org/10.1038/cr.2008.328
Acknowledgements
This research was supported by the Chongqing General Research Project of Basic Science & Frontier Technology (No. cstc2016jcyjA0592).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, Y., Lin, C., Han, R. et al. Metformin attenuates TGF-β1-induced pulmonary fibrosis through inhibition of transglutaminase 2 and subsequent TGF-β pathways. 3 Biotech 10, 287 (2020). https://doi.org/10.1007/s13205-020-02278-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02278-2