Abstract
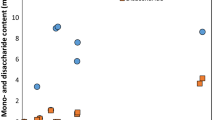
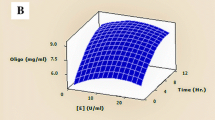
Copra meal is a by-product of coconut milk extraction, contained 4.60 ± 0.01 g/100 g DM and 62.19 ± 0.53% of protein and fiber, respectively. The optimal condition for quality improvement of copra meal was investigated using Box–Behnken design combined with response surface methodology (RSM). The simultaneous saccharification and fermentation (SSF) of Copra meal was performed by mannanase enzyme and yeast, Saccharomyces cerevisiae. The concentration of mannanase was determined as the most important factor to increase protein content in copra meal. The protein content was increased by 64% when 0.7% of enzyme per copra meal dry weight, the ratio of copra meal to water at 1:4.56 and fermentation time of 90.25 h at 30 °C were used. The program predicted an increase of 3.06 g of protein/100 g dry matter; however, the experimental result showed an increase of 3.35 g/100 g DM of protein in copra meal. The 10 kg of copra meal SSF in Koji reactor, the protein content increased to 4.18 g/100 g DM, while fiber content decreased 49%. Moreover, amino acids were increased by 64.05% and oligosaccharides, especially mannohexaose, were increased to 0.708 g/g DM. Results showed that fermentation of copra meal with mannanase and yeast offers a potential method to improve the nutrition of copra meal as animal feed.




Similar content being viewed by others
References
AOAC (2000) Official methods of analysis of AOAC International, 17th edn. AOAC International, Gaithersburg
Asano I, Nakamura Y, Hoshino H, Aoki K, Fujii S, Imura N, Iino H (2001) Use of mannooligosaccharides from coffee mannan by intestinal bacteria. Nippon Nogeikagaku Kaishi 75:1077–1083 (in Japanese)
Balasubramaniam K (1976) Polysaccharides of the kernel of maturing and matured coconuts. J Food Sci 41:1371–1373
Chandrakant P, Bisaria VS (1998) Simultaneous bioconversion of cellulose and hemicellulose to ethanol. Crit Rev Biotechnol 18(4):295–331
Creswell DC, Brooks CC (1971) Effect of coconut meal on Coturnix quail and of coconut meal and coconut oil on performance, carcass measurements and fat composition in swine. J Anim Sci 33(2):370–375
Dairo FAS, Ogunmodede BK (2004) Performance of laying hens fed coconut meal as replacement for groundnut cake. Trop J Anim Sci 2:87–90
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Graham H, Balnave D (1995) Dietary enzyme for increasing energy availability. In: Wallace RJ, Chesson A (eds) Biotechnology in animal feeds and animal feeding. VCH Verlagsgesellschaft, Weinheim, p 302
Hassaan MS, Soltan MA, Abdel-Moez AM (2015) Nutritive value of soybean meal after solid state fermentation with Saccharomyces cerevisiae for Nile tilapia, Oreochromis niloticus. Anim Fd Sci Technol 201:89–98
Herbert P, Barros P, Ratola N, Alves A (2000) HPLC determination of amino acids in musts and port wine using OPA/FMOC derivatives. J Food Sci 65(7):1130–1133
Index Mundi (2018) IndexMundi country reports, Massachusetts. http://www.indexmundi.com. Accessed 15 Aug 2019
Khanongnuch C, Sa-nguansook C, Lumyong S (2006) Nutritive quality of β-mannanase treated copra meal in broiler diets and effectiveness on some fecalbacteria. Int J Poult Sci 5:1087–1091
Khuwijitjaru P, Watsanit K, Adachi S (2012) Carbohydrate content and composition of product from subcritical water treatment of coconut meal. J Ind Eng Chem 18:225–229
Khuwijitjaru P, Pokpong A, Klinchongkon K, Adachi S (2014) Production of oligosaccharides from coconut meal by subcritical water treatment. Int J Food Sci Technol 49(8):1946–1952
Kim BG, Lee JH, Jung HJ, Han YK, Park KM, Han IK (2001) Effect of partial replacement of soybean meal with palm kernel meal and copra meal on growth performance, nutrient digestibility and carcass characteristics of finishing pigs. Asian Aust J Anim Sci 14(6):821–830
Knudsen KEB (1997) Carbohydrate and lignin contents of plant materials used in animal feeding. Anim Fd Sci. Technol 67:319–338
Limmanee M, Rumpagaporn P (2017) Preparation of coconut residue for chemical modification and addition in food products. In: The 55th Kasetsart University Annual Conference, pp 809–816
Marini AM, Yatim AM, Babji AS, Annuar BO, Noraini S (2006) Evaluation of nutrient contents and amino acid profiling of various types of PKC. Anal Chem 2:135–141
McCleary BV (1988) b-D-Mannanase. Methods Enzymol 160:596–610
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Moorthy M, Viswanathan K (2009) Nutritive value of extracted coconut (Cocos nucifera) meal. Res J Agric Biol Sci 5:515–517
Naughton PJ, Mikkelsen LL, Jensen BB (2001) Effects of nondigestible oligosaccharides on Salmonella enterica serovar Typhimurium and nonpathogenic Escherichia coli in the pig small intestine in vitro. Appl Environ Microbiol 67(8):3391–3395
Oboh G (2006a) Nutrient enrichment of Cassava peels using a mixed culture of Saccharomyces cerevisiae and Lactobacillus spp. solid media fermentation techniques. Electron. J Biotechnol 9:4649
Oboh G (2006b) Nutrient enrichment of Cassava peels using a mixed culture of Saccharomyces cerevisiae and Lactobacillus spp solid media fermentation techniques. Electron J Biotechnol 9:4649
Oboh G, Akindahunsi AA (2003) Biochemical changes in Cassava products (flour & gari) subjected to Saccharomyces cerevisiae solid media fermentation. Food Chem 82(4):599–602
Pangsri P (2014) Characterization of mannanase from Bacillus circulans NT 6.7 and its application in mannooligosaccharides preparation. Ph.D. thesis, Kasetsart University
Pangsri P, Pangsri P (2017) Mannanase enzyme from Bacillus subtilis P2-5 with waste management. Energy Proc 138:343–347
Pangsri P, Piwpankaew Y, Ingkakul A, Nitisinprasert S, Keawsompong S (2015) Characterization of mannanase from Bacillus circulans NT 6.7 and its application in mannooligosaccharides preparation as prebiotic. http://www.springerplus.com/content/4/1/77
Pham TA, Berrin JG, Record E, To KA, Sigoillot JC (2010) Hydrolysis of softwood by Aspergillus mannanase: role of a carbohydrate-binding module. J Biotechnol 148:163–170
Rungruangsaphakun J, Keawsompong S (2018) Optimization of hydrolysis conditions for the mannooligosaccharides copra meal hydrolysate production. 3 Biotech 8:169
Swick RA (1999) Considerations in using protein meals for poultry and swine. American Soybean Association, Technical Bulletin, Washington, p 19
Titapoka S, Keawsompong S, Haltrich D, Nitisinprasert S (2008) Selection and characterization of mannanase-producing bacteria useful for the formation of prebiotic manno-oligosaccharides from copra meal. World J Microbiol Biotechnol 24:1425–1433
Yalegama LLWC, Nedra Karunaratne D, Sivakanesan R, Jayasekara C (2013) Chemical and functional properties of fibre concentrates obtained from by-products of coconut kernel. Food Chem 141:124–130
Acknowledgements
This research was supported by Research and Researchers for Industries (RRI), the Thailand Research Fund under the Office of the Prime Minister, Royal Thai Government (code: MSD5810009) and Ampol Food Processing Limited, Nakhon Pathom Province, Thailand.
Author information
Authors and Affiliations
Contributions
Conceptualization: [SM, SK]; Methodology: [JK, SM and SK]; Formal analysis and investigation: [JK]; Writing—original draft preparation: [JK, SM and SK]; Writing—review and editing: [SM and SK]; Funding acquisition: [SM and SK]; Resources: [SM and SK]; Supervision: [SM and SK].
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kraikaew, J., Morakul, S. & Keawsompong, S. Nutritional improvement of copra meal using mannanase and Saccharomyces cerevisiae. 3 Biotech 10, 274 (2020). https://doi.org/10.1007/s13205-020-02271-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02271-9