Abstract
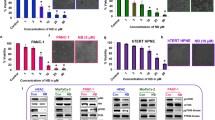
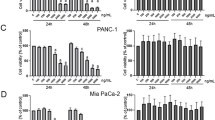
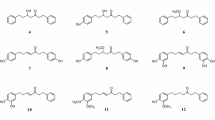
This study explored the effect of methyl-indole on pancreatic cancer cell viability and investigated the mechanism involved. The viability of pancreatic cells showed a significant suppression on treatment with methyl-indole in dose-based manner. Treatment with 5 µM methyl-indole suppressed Capan-1 cell viability to 23%. The viability of Aspc-1 cells was reduced to 20% and those of MIApaCa-2 cells to 18% by 5 µM methyl-indole. The apoptotic proportion of Capan-1 cells was 67%, while as those of Aspc-1 and MIApaCa-2 cells increased to 72 and 77%, respectively, on treatment with 5 µM methyl-indole. The level of P13K, p-Tyr, p-Crkl and p-Akt was inhibited in the cells by methyl-indole. Moreover, methyl-indole also suppressed zinc-finger protein, X-linked mRNA and protein expression in tested cells. In summary, methyl-indole exhibits anti-proliferative effect on pancreatic cancer cells and induces apoptosis. It targeted ZFX expression and down-regulated P13K/AKT pathway in pancreatic cancer cells. Therefore, methyl-indole acts as therapeutic agent for pancreatic cancer and may be studied further.








Similar content being viewed by others
References
Burchert A, Wang Y, Cai D, Von Bubnoff N, Paschka P, Müller-Brüsselbach S, Ottmann OG, Duyster J, Hochhaus A, Neubauer A (2005) compensatory Pi3-kinase/akt/mTor activation regulates imatinib resistance development. Leukemia 19:1774–1782
Carstens JL et al (2017) Spatial computation of intratumoral T cells correlates with survival of patients with pancreatic cancer. Nat Commun 8:15095
Dehn DL, Siegel D, Zafar KS, Reigan P, Swann E, Moody CJ, Ross D (2006) 5-Methoxy-1,2-dimethyl-3-[(4-nitrophenoxy)methyl]indole-4,7-dione, a mechanism-based inhibitor of NAD(P)H:quinone oxidoreductase 1, exhibits activity against human pancreatic cancer in vitro and in vivo. Mol Cancer Ther 5:1702–1709
Desplat V, Lagarde V, Belloc F, Chollet C, Leguay T, Pasquet JM, Praloran V, Mahon FX (2004) Rapid detection of phosphotyrosine proteins by flow cytometric analysis in Bcr-Abl-positive cells. Cytometry A 62:35–45
Fang Q, Fu WH, Yang J, Li X, Zhou ZS, Chen ZW, Pan JH (2014) Knockdown of ZFX suppresses renal carcinoma cell growth and induces apoptosis. Cancer Genet 207:461–466
Hamilton A, Alhashimi F, Myssina S, Jorgensen HG, Holyoake TL (2009) Optimization of methods for the detection of Bcr-aBl activity in Philadelphia-positive cells. Exp Hematol 37:395–401
Harel S, Tu EY, Weisberg S, Esquilin M, Chambers SM, Liu B, Carson CT, Studer L, Reizis B, Tomishima MJ (2012) ZFX controls the self-renewal of human embryonic stem cells. PLoS ONE 7:e42302
Höhne MW, Halatsch ME, Kahl GF, Weinel RJ (1992) Frequent loss of expression of the potential tumor suppressor gene DCC in ductal pancreatic adenocarcinoma. Cancer Res 52:2616–2619
La Rosée P, Holm-Eriksen S, Konig H, Härtel N, Ernst T, Debatin J, Mueller MC, Erben P, Binckebanck A, Wunderle L et al (2008) Phospho-crKl monitoring for the assessment of Bcr-aBl activity in imatinib-resistant chronic myeloid leukemia or Ph+ acute lymphoblastic leukemia patients treated with nilotinib. Haematologica 93:765–769
Le DT et al (2013) Evaluation of ipilimumab in combination with allogeneic pancreatic tumor cells transfected with a GM-CSF gene in previously treated pancreatic cancer. J Immunother Hagerstown Md 1997 36:382–389
Lewis A, Ough M, Li L, Hinkhouse MM, Ritchie JM, Spitz DR, Cullen JJ (2004) Treatment of pancreatic cancer cells with dicumarol induces cytotoxicity and oxidative stress. Clin Cancer Res 10:4550–4558
Li K, Zhu ZC, Liu YJ, Liu JW, Wang HT, Xiong ZQ, Shen X, Hu ZL, Zheng J (2013) ZFX knockdown inhibits growth and migration of non-small cell lung carcinoma cell line H1299. Int J Clin Exp Pathol 6:2460–2467
Liu L et al (2016) Low intratumoral regulatory T cells and high peritumoral CD8(+) T cells relate to long-term survival in patients with pancreatic ductal adenocarcinoma after pancreatectomy. Cancer Immunol Immunother CII 65:73–82
Meng H, Yang C, Jin J, Zhou Y, Qian W (2008) Homoharringtonine inhibits the aKT pathway and induces in vitro and in vivo cytotoxicity in human multiple myeloma cells. Leuk Lymphoma 49:1954–1962
Ryan DP, Hong TS, Bardeesy N (2014) Pancreatic adenocarcinoma. N Engl J Med 371:1039–1049
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69:7–34
Steelman LS, Pohnert SC, Shelton JG, Franklin RA, Bertrand FE, Mccubrey JA (2004) JAK/STAT, Raf/MeK/erK, PI3K/akt and BcCR-ABl in cell cycle progression and leukemogenesis. Leukemia 18:189–218
Wang L, You LS, Ni WM, Ma QL, Tong Y, Mao LP, Qian JJ, Jin J (2013) β-catenin and aKT are promising targets for combination therapy in acute myeloid leukemia. Leuk Res 37:1329–1340
Weisberg SP, Smith-raska MR, Esquilin JM, Zhang J, Arenzana TL, Lau CM, Churchill M, Pan H, Klinakis A, Dixon JE et al (2014) ZFX controls propagation and prevents differentiation of acute T-lymphoblastic and myeloid leukemia. Cell Rep 6:528–540
Winski SL, Faig M, Bianchet MA, Siegel D, Swann E, Fung K, Duncan MW, Moody CJ, Amzel LM, Ross D (2001) Characterization of a mechanism-based inhibitor of NAD(P)H:quinone oxidoreductase 1 by biochemical, X-ray crystallographic, and mass spectrometric approaches. Biochemistry 40:15135–15142
Wu J, Wei B, Wang Q, Ding Y, Deng Z, Lu X, Li Y (2016) ZFX facilitates cell proliferation and imatinib resistance in chronic myeloid leukemia cells. Cell Biochem Biophys 74:277–283
Yang H, Lu Y, Zheng Y, Yu X, Xia X, He X, Feng W, Xing L, Ling Z (2014) Shrna-mediated silencing of ZFX attenuated the proliferation of breast cancer cells. Cancer Chemother Pharmacol 73:569–576
Zhang S, Shu R, Yue M, Zhang S (2016) Effect of over-expression of Zinc-finger protein (ZFX) on self-renewal and drug-resistance of hepatocellular carcinoma. Med Sci Monit 22:3025–3034
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declared that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Qin, X., Cui, X. Methyl-indole inhibits pancreatic cancer cell viability by down-regulating ZFX expression. 3 Biotech 10, 187 (2020). https://doi.org/10.1007/s13205-020-02179-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02179-4