Abstract
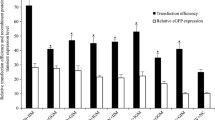
In this study, we systemically investigated the positions and orientations of matrix attachment regions (MARs) in expression vectors to fully explore the mechanism for improving transgene expression. We constructed 14 vectors that incorporated human β-globin MARs into pIRES-eGFP backbone vectors. The MARs flanked the eGFP expression cassette or promoter in a forward/reverse orientation. After stable transfection into CHO-K1 cells with these vectors, eGFP expression levels were increased significantly relative to that of the control vector (MAR-devoid) when two MARs flanking the expression cassette were incorporated, followed by those at the 5′ site (upstream of the promoter). Simultaneously, the percentage of the eGFP-expressing cells was elevated to some extent. The vector with both MARs in forward orientation flanking the expression cassette yielded the highest transgene expression levels (2.5-fold). The orientation (forward or reverse) of the MARs did not present a significant difference when added in the same site. In addition, transgene expression levels were not exclusively dependent on transgene copy numbers. Bioinformatic analysis indicated that some specific transcription factors may contribute to the transcriptional process. In conclusion, two MARs in a forward orientation and flanking the expression cassette comprised the optimal construct for improving the stable transgene expression in the CHO-K1 cells. The effects may be related to specific transcription factors, such as PRDM1 and REL.





Similar content being viewed by others
References
Arope S, Harraghy N, Pjanic M, Mermod N (2013) Molecular characterization of a human matrix attachment region epigenetic regulator. PLoS One 8:e79262
Bode J, Benham C, Knopp A, Mielke C (2000) Transcriptional augmentation: modulation of gene expression by scaffold/matrix-attached regions (S/MAR elements). Crit Rev Eukaryot Gene Expr 10:73–90
Buceta M, Galbete JL, Kostic C, Arsenijevic Y, Mermod N (2011) Use of human MAR elements to improve retroviral vector production. Gene Ther 18:7–13
Dolgova AS, Dolgov SV (2019) Matrix attachment regions as a tool to influence plant transgene expression. 3 Biotech 9:176
Galbete JL, Buceta M, Mermod N (2009) MAR elements regulate the probability of epigenetic switching between active and inactive gene expression. Mol Biosyst 5:143–150
Gaussin A, Modlich U, Bauche C, Niederländer NJ, Schambach A, Duros C, Artus A, Baum C, Cohen-Haguenauer O, Mermod N (2012) CTF/NF1 transcription factors act as potent genetic insulators for integrating gene transfer vectors. Gene Ther 19:15–24
Girod PA, Mermod N (2003) Use of scaffold/matrix attachment regions for protein production. Gene Transf Expr Mamm Cells 38:359–379
Girod PA, Zahn-Zabal M, Mermod N (2005) Use of the chicken lysozyme matrix attachment region to generate high producer CHO cell lines. Biotechnol Bioeng 91:1–11
Girod PA, Nguyen DQ, Calabrese D, Puttini S, Grandjean M, Martinet D, Regamey A, Saugy D, Beckmann JS, Bucher P, Mermod N (2007) Genome-wide prediction of matrix attachment regions that increase gene expression in mammalian cells. Nat Methods 4:747–753
Grandjean M, Girod PA, Calabrese D, Kostyrko K, Wicht M, Yerly F, Mazza C, Beckmann JS, Martinet D, Mermod N (2011) High-level transgene expression by homologous recombination-mediated gene transfer. Nucleic Acids Res 39:e104
Heng HH, Goetze S, Ye CJ, Liu G, Stevens JB, Bremer SW, Wykes SM, Bode J, Krawetz SA (2004) Chromatin loops are selectively anchored using scaffold/matrix-attachment regions. J Cell Sci 117:999–1008
Jia YL, Guo X, Ni TJ, Lu JT, Wang XY, Wang TY (2019a) Novel short synthetic matrix attachment region for enhancing transgenic expression in recombinant Chinese hamster ovary cells. J Cell Biochem 120:18478–18486
Jia YL, Guo X, Wang XC, Wang TY (2019b) Human genome-derived TOP1 matrix attachment region enhances transgene expression in the transfected CHO cells. Biotechnol Lett 41:701–709
Kim JM, Kim JS, Park DH, Kang HS, Yoon J, Baek K, Yoon Y (2004) Improved recombinant gene expression in CHO cells using matrix attachment regions. J Biotechnol 107:95–105
Kwaks TH, Otte AP (2006) Employing epigenetics to augment the expression of therapeutic proteins in mammalian cells. Trends Biotechnol 24:137–142
Li Q, Dong W, Wang T, Liu Z, Wang F, Wang X, Zhao C, Zhang J, Wang L (2013) Effect of β-globin MAR characteristic elements on transgene expression. Mol Med Rep 7:1871–1874
Li Q, Zhao CP, Lin Y, Song C, Wang F, Wang TY (2019) Two human MARs effectively increase transgene expression in transfected CHO cells. J Cell Mol Med 23:1613–1616
Linnemann AK, Krawetz SA (2009) Silencing by nuclear matrix attachment distinguishes cell-type specificity: association with increased proliferation capacity. Nucleic Acids Res 37:2779–2788
Linnemann AK, Platts AE, Krawetz SA (2009) Differential nuclear scaffold/matrix attachment marks expressed genes. Hum Mol Genet 18:645–654
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Majocchi S, Aritonovska E, Mermod N (2014) Epigenetic regulatory elements associate with specific histone modifications to prevent silencing of telomeric genes. Nucleic Acids Res 42:193–204
Mathelier A, Fornes O, Arenillas DJ, Chen CY, Denay G, Lee J, Shi W, Shyr C, Tan G, Worsley-Hunt R, Zhang AW, Parcy F, Lenhard B, Sandelin A, Wasserman WW (2016) JASPAR 2016: a major expansion and update of the open-access database of transcription factor binding profiles. Nucleic Acids Res 44:D110–D115
Sjeklocha L, Chen Y, Daly MC, Steer CJ, Kren BT (2011) β-globin matrix attachment region improves stable genomic expression of the sleeping beauty transposon. J Cell Biochem 112:2361–2375
Tian ZW, Xu DH, Wang TY, Wang XY, Xu HY, Zhao CP, Xu GH (2018) Identification of a potent MAR element from the human genome and assessment of its activity in stably transfected CHO cells. J Cell Mol Med 22:1095–1102
Varghese J, Alves W, Brill B, Wallace M, Calabrese D, Regamey A, Girod P (2008) Rapid development of high-performance, stable mammalian cell lines for improved clinical development. Bioprocess J 7:30–36
Wang TY, Yang R, Qin C, Wang L, Yang XJ (2008) Enhanced expression of transgene in CHO cells using matrix attachment region. Cell Biol Int 32:1279–1283
Wang TY, Zhang JH, Jing CQ, Yang XJ, Lin JT (2010) Positional effects of the matrix attachment region on transgene expression in stably transfected CHO cells. Cell Biol Int 34:141–145
Wang F, Wang TY, Tang YY, Zhang JH (2012) Different matrix attachment regions flanking a transgene effectively enhance gene expression in stably transfected Chinese hamster ovary cells. Gene 500:59–62
Wang XJ, Wang J, Wang YY, Guo YJ, Chu BB, Yang GY (2016) Sus scrofa matrix attachment region enhances expression of the PiggyBac system transfected into HEK293T cells. Biotechnol Lett 38:949–958
Wang W, Guo X, Li YM, Wang XY, Yang XJ, Wang YF, Wang TY (2018) Enhanced transgene expression using cis-acting elements combined with the EF1 promoter in a mammalian expression system. Eur J Pharm Sci 123:539–545
Yu J, Bock JH, Slightom JL, Villeponteau B (1994) A 5′ β-globin matrix attachment region and the polyoma enhancer together confer position-independent transcription. Gene 139:139–145
Acknowledgements
This work was supported by the Grant from the National Natural Science Foundation of China (no. U1604193).
Author information
Authors and Affiliations
Contributions
JZ designed, analyzed the experiments and wrote the manuscript. JZ performed the experiments and co-wrote the manuscript. SC and WY analyzed some experiment data and co-wrote the manuscript. SL performed the experiment for vector construction and cultured cells.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, J., Zhang, J., Cheng, S. et al. Enhanced transgene expression using two β-globin MARs flanking expression cassettes in stably transfected CHO-K1 cells. 3 Biotech 9, 435 (2019). https://doi.org/10.1007/s13205-019-1971-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1971-6