Abstract
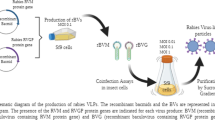
In the present study, we developed a complete process to produce in insect cells a high amount of the ectodomain of rabies virus glycoprotein G (GE) as suitable antigen for detecting anti-rabies antibodies. Using the baculovirus expression vector system in Sf9 insect cells combined with a novel chimeric promoter (polh-pSeL), the expression level reached a yield of 4.1 ± 0.3 mg/L culture, which was significantly higher than that achieved with the standard polh promoter alone. The protein was recovered from the cell lysates and easily purified in only one step by metal ion affinity chromatography, with a yield of 95% and a purity of 87%. Finally, GE was successfully used in an assay to detect specific antibodies in serum samples derived from rabies-vaccinated animals. The efficient strategy developed in this work is an interesting method to produce high amounts of this glycoprotein.






Similar content being viewed by others
References
Ashikawa Y, Ihara M, Matsuura N, Fukunaga Y, Kusakabe Y, Yamashita A (2011) GFP-based evaluation system of recombinant expression through the secretory pathway in insect cells and its application to the extracellular domains of class C GPCRs. Protein Sci 20:1720–1734. https://doi.org/10.1002/pro.707
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Carpenter EP, Beis K, Cameron AD, Iwata S (2008) Overcoming the challenges of membrane protein crystallography. Curr Opin Struct Biol 18:581–586. https://doi.org/10.1016/j.sbi.2008.07.001
Cliquet F, McElhinney LM, Servat A, Boucher JM, Lowings JP, Goddard T, Mansfield KL, Fooks AR (2004) Development of a qualitative indirect ELISA for the measurement of rabies virus-specific antibodies from vaccinated dogs and cats. J Virol Methods 117:1–8. https://doi.org/10.1016/j.jviromet.2003.12.001
Dalton AC, Barton WA (2014) Over-expression of secreted proteins from mammalian cell lines. Protein Sci 23:517–525. https://doi.org/10.1002/pro.2439
Feyssaguet M, Dacheux L, Audry L, Compoint A, Morize JL, Blanchard I, Bourhy H (2007) Multicenter comparative study of a new ELISA, PLATELIA™ RABIES II, for the detection and titration of anti-rabies glycoprotein antibodies and comparison with the rapid fluorescent focus inhibition test (RFFIT) on human samples from vaccinated and non-vaccinated people. Vaccine 25:2244–2251. https://doi.org/10.1016/j.vaccine.2006.12.012
Friedrich A, Ledesma M, Landone Ferrari A, Leoni J (2014) Production of a monoclonal antibody against serum immunoglobulin M of South American camelids and assessment of its suitability in two immunoassays. J Vet Diagn Invest 26:646–650. https://doi.org/10.1177/1040638714543675
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker JM (ed) The Proteomics Protocols Handbook. Humana Press, New York, pp 571–607
Gupta PK, Sharma S, Walunj SS, Chaturvedi VK, Raut AA, Patial S et al (2005) Immunogenic and antigenic properties of recombinant soluble glycoprotein of rabies virus. Vet Microbiol 108:207–214. https://doi.org/10.1016/j.vetmic.2005.04.007
Hampson K, Coudeville L, Lembo T, Sambo M, Kieffer A, Attlan M et al (2015) Estimating the global burden of endemic canine rabies. PLoS Negl Trop Dis 9:1–20. https://doi.org/10.1371/journal.pntd.0003709
Huang J, Zhang Y, Huang Y, Gnanadurai CW, Zhou M, Zhao L, Fu ZF (2017) The ectodomain of rabies virus glycoprotein determines dendritic cell activation. Antiviral Res 141:1–6. https://doi.org/10.1016/j.antiviral.2017.01.022
Ikonomou L, Shneider YJ, Agathos SN (2003) Insect cell culture for industrial production of recombinant proteins. Appl Microbiol Biotechnol 62:1–20. https://doi.org/10.1007/s00253-003-1223-9
Jibat T, Mourits MC, Hogeveen H (2016) Incidence and economic impact of rabies in the cattle population of Ethiopia. Prev Vet Med 130:67–76. https://doi.org/10.1016/j.prevetmed.2016.06.005
Klaus T, Kulesza M, Bzowska M, Wyroba B, Kilarski WW, Bereta J (2015) Overcoming inefficient secretion of recombinant VEGF-C in baculovirus expression vector system by simple purification of the protein from cell lysate. Protein Expr Purif 110:151–158. https://doi.org/10.1016/j.pep.2015.03.001
Martínez-Solís M, Gómez-Sebastián S, Escribano JM, Jakubowska AK, Herrero S (2016) A novel baculovirus-derived promoter with high activity in the baculovirus expression system. PeerJ 4:e2183. https://doi.org/10.7717/peerj.2183
Martínez-Solís M, Herrero S, Targovnik AM (2019) Engineering of the baculovirus expression system for optimized protein production. Appl Microbiol Biotechnol 103:113–123. https://doi.org/10.1007/s00253-018-9474-7
Metz SW, Geertsema C, Martina BE, Andrade P, Heldens JG, van Oers MM, Goldbach RW, Vlak JM, Pijlman GP (2011) Functional processing and secretion of Chikungunya virus E1 and E2 glycoproteins in insect cells. Virol J 8:353. https://doi.org/10.1186/1743-422X-8-353
O´Reilly DR, Miller LK, Luckow VA (1994) Baculovirus expression vector:a laboratory manual. Oxford University Press, Oxford
Perrin P, Thibodeau L, Sureau P (1985) Rabies immunosome (subunit vaccine) structure and immunogenicity. Pre- and postexposure protection studies. Vaccine 3:325–332. https://doi.org/10.1016/S0264-410X(85)90224-5
Picotto LD, Sguazza GH, Tizzano MA, Galosi CM, Cavalitto SF, Pecoraro MR (2017) An effective and simplified DO-stat control strategy for production of rabies glycoprotein in Pichia pastoris. Protein Expr Purif 132:124–130. https://doi.org/10.1016/j.pep.2017.02.004
Qian W, Aguilar F, Wang T, Qiu B (2013) Secretion of truncated recombinant rabies virus glycoprotein with preserved antigenic properties using a co-expression system in Hansenula polymorpha. J Microbiol 51:234–240. https://doi.org/10.1007/s12275-013-2337-0
Qoronfleh MW, Benton B, Ignacio R, Kaboord B (2003) Selective enrichment of membrane proteins by partition phase separation for proteomic studies. J Biomed Biotechnol 2003:249–255. https://doi.org/10.1155/S1110724303209244
Ramya R, Mohana Subramanian B, Sivakumar V, Senthilkumar RL, Sambasiva Rao KRS, Srinivasan VA (2011) Expression and solubilization of insect cell-based rabies virus glycoprotein and assessment of its immunogenicity and protective efficacy in mice. Clin Vaccine Immunol 18:1673–1679. https://doi.org/10.1128/CVI.05258-11
Rendić D, Wilson IBH, Paschinger K (2008) The glycosylation capacity of insect cells. Croat Chem Acta 81:7–21
Sakamoto S, Ide T, Tokiyoshi S, Nakao J, Hamada F, Yamamoto M et al (1999) Studies on the structures and antigenic properties of rabies virus glycoprotein analogues produced in yeast cells. Vaccine 17:205–218. https://doi.org/10.1016/S0264-410X(98)00196-0
Shakin-Eshleman SH, Remaley AT, Eshleman JR, Wunner WH, Spitalnik SL (1992) N-linked glycosylation of rabies virus glycoprotein. Individual sequons differ in their glycosylation efficiencies and influence on cell surface expression. J Biol Chem 25:10690–10698
Sissoëff L, Mousli M, England P, Tuffereau C (2005) Stable trimerization of recombinant rabies virus glycoprotein ectodomain is required for interaction with the p75NTR receptor. J Gen Virol 86:2543–2552. https://doi.org/10.1099/vir.0.81063-0
Steele KH, Stone BJ, Franklin KM, Fath-Goodin A, Zhang X, Jiang H, Webb BA, Geisler C (2017) Improving the baculovirus expression vector system with vankyrin-enhanced technology. Biotechnol Prog 33:1496–1507. https://doi.org/10.1002/btpr.2516
Stiles B, Wood HA (1983) A study of glycoproteins of Autographa californica Nuclear polyhedrosis virus (AcNPV). Virology 131:230–241. https://doi.org/10.1016/0042-6822(83)90548-2
Targovnik AM, Romero LV, Wolman FJ, Cascone O, Miranda MV (2010) Horseradish peroxidase production from Spodoptera frugiperda larvae: a simple and inexpensive method. Process Biochem 45:835–840. https://doi.org/10.1016/j.procbio.2010.02.003
Toth AM, Geisler C, Aumiller JJ, Jarvis DL (2011) Factors affecting recombinant Western equine encephalitis virus glycoprotein production in the baculovirus system. Protein Expr Purif 80:274–282. https://doi.org/10.1016/j.pep.2011.08.002
Trabucchi A, Bombicino SS, Targovnik AM, Marfía JI, Sabljic AV, Faccinetti NI, Guerra LL, Iacono RF, Miranda MV, Valdez SN (2019) Expression of recombinant glutamic acid decarboxylase in insect larvae and its application ina an immunoassay for the diagnosis of autoimmune diabetes mellitus. Sci Rep 9:824. https://doi.org/10.1038/s41598-018-35744-2
Tuchiya K, Matsuura Y, Kawai A, Ishihama A, Ueda S (1992) Characterization of rabies virus glycoprotein expressed by recombinant baculovirus. Virus Res 25:1–13. https://doi.org/10.1016/0168-1702(92)90095-Q
Urtasun N, Baieli MF, Cascone O, Wolman FJ, Miranda MV (2015) High-level expression and purification of recombinant wheat germ agglutinin in Rachiplusia nu larvae. Process Biochem 50:40–47. https://doi.org/10.1016/j.procbio.2014.11.004
van Oers MM (2011) Opportunities and challenges for the baculovirus expression system. J Invertebr Pathol 107:S3–S15. https://doi.org/10.1016/j.jip.2011.05.001
Wasniewski M, Cliquet F (2012) Evaluation of ELISA for detection of rabies antibodies in domestic carnivores. J Virol Methods 179:166–175. https://doi.org/10.1016/j.jviromet.2011.10.019
Wunner WH, Larson JK, Dietzschold B, Smith CL (1988) The molecular biology of rabies viruses. Rev Infect Dis 10:S771–S784
Yelverton E, Norton S, Obijeski JF, Goeddel DV (1983) Rabies virus glycoprotein analogs: biosynthesis in Escherichia coli. Science 219:614–620. https://doi.org/10.1126/science.6297004
Zhang S, Liu Y, Zhang F, Hu R (2009) Competitive ELISA using a rabies glycoprotein-transformed cell line to semi-quantify rabies neutralizing-related antibodies in dogs. Vaccine 27:2108–2113. https://doi.org/10.1016/j.vaccine.2009.01.126
Acknowledgements
This work was supported by grants from Agencia Nacional de Promoción Científica y Tecnológica de Argentina (PICT 2015-1992; PICT 2014-3350), by Universidad de Buenos Aires (UBACyT 2016-20020150100145BA). AMT, VA, MGL, AF, and MVM are career researchers of the Consejo Nacional de Investigaciones Científicas y Técnicas de Argentina (CONICET). GJM, MBA, and IS are research fellows of CONICET.
Author information
Authors and Affiliations
Contributions
AMT, SH, AF, and MVM designed the experiments. AMT and MMS constructed the baculovirus vectors. AMT and GJM performed the expression experiments. AMT and LFB contributed to downstream processing. IS and MBA performed the characterization experiments. AF contributed to the development of the ELISA. VA and MGL performed the confocal analysis. AMT, AF, SH, and MVM analyzed the data. All authors discussed the results and agreed upon the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Targovnik, A.M., Ferrari, A., Mc Callum, G.J. et al. Highly efficient production of rabies virus glycoprotein G ectodomain in Sf9 insect cells. 3 Biotech 9, 385 (2019). https://doi.org/10.1007/s13205-019-1920-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1920-4