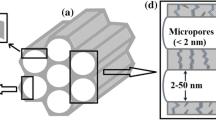
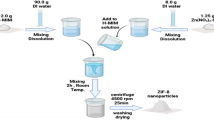
Abstract
In this work, an improved method of template synthesis including an additional post-carbonization stage has been developed. A sample of nanostructured carbonaceous material synthesized by this method was studied using low-temperature N2 ad(de)sorption and derivatography and compared with the conventionally synthesized sample and the activated anthracite sample. Also, the acidity of the materials by Boehm titration and the effect of Ni introduction on thermal characteristics of the samples were evaluated. In the post-carbonized sample, the development of the external surface and mesoporosity in the nanoscale range of 6–20 nm was observed as well as a reduction in the number of micropores. This had a positive effect on the thermal stability of the material, which was confirmed by the calculated values of the combustion activation energy. The post-carbonization stage promoted the formation of a phase whose thermal stability was close to that in activated anthracite. In terms of thermal stability, this phase was also less sensitive to the negative effect of the introduction of Ni compared to the phases of the conventionally synthesized sample. The existence of phases with different thermal stability in nanostructured carbonaceous materials was interpreted in terms of differences in the local concentration of activity centers in the material. An increased thermal stability of the additionally carbonized material was explained by a reduction of its microporosity and, ultimately, a decrease in the number of activity centers (surface defects, acid sites, etc.), which are the factors that initiate the carbon combustion.




Similar content being viewed by others
References
Asaula VM, Shvets OV, Pariiska OO, Buryanov VV, Ryabukhin SV, Volochnyuk DM, Kolotilov SV (2020) Composites based on nanodispersed nickel, graphene-like carbon, and aerosil for catalytic hydrogenation of furfural and quinoline. Theor Exp Chem 56:261–267. https://doi.org/10.1007/s11237-020-09657-z
Chuenchom L, Kraehnert R, Smarsly BM (2012) Recent progress in soft-templating of porous carbon materials. Soft Matter 8:10801–10812. https://doi.org/10.1039/C2SM07448F
Delgado JJ, Chen X, Tessonnier JP, Schuster ME, Del Rio E, Schlögl R, Su DS (2010) Influence of the microstructure of carbon nanotubes on the oxidative dehydrogenation of ethylbenzene to styrene. Catal Today 150(1–2):49–54. https://doi.org/10.1016/j.cattod.2009.07.103
Deng Y, Cai Y, Sun Z, Gu D, Wei J, Li W, Guo X, Yang J, Zhao D (2010) Controlled synthesis and functionalization of ordered large-pore mesoporous carbons. Adv Funct Mater 20(21):3658–3665. https://doi.org/10.1002/adfm.201001202
Dyachenko AG, Tsapyuk GG, Gaidai SV, Ischenko OV, Zakharova TM, Il’nitskaya GD, Loginova OB (2020) Synthetic nanodiamonds (SNDs) containing bimetallic Ni(Co)–Fe composites: preparation, characterization and catalytic performance in the reaction of CO2 methanation. Mol Cryst Liq Cryst 701:91–97. https://doi.org/10.1080/15421406.2020.1732566
Dyachenko AG, Ischenko OV, Prygunova OV, Diyuk VE, Tsapyuk GG, Gaidai SV, Yatsymyrskyi AV, Zakharova TM, Kostyrko EO (2021) Ni–Fe, Co–Fe, and Co–Ni nanocomposites based on carbon nanotubes in the reaction of CO2 methanation. Mol Cryst Liq Cryst 720:38–46. https://doi.org/10.1080/15421406.2021.1905281
Frank B, Morassutto M, Schomäcker R, Schlögl R, Su DS (2010) Oxidative dehydrogenation of ethane over multiwalled carbon nanotubes. ChemCatChem 2:644–648. https://doi.org/10.1002/cctc.201000035
Hoheisel TN, Schrettl S, Szilluweit R, Frauenrath H (2010) Nanostructured carbonaceous materials from molecular precursors. Angew Chem Int Ed 49(37):6496–6515. https://doi.org/10.1002/anie.200907180
Landers J, Gor GYu, Neimark AV (2013) Density functional theory methods for characterization of porous materials. Colloids Surf, A 437:3–32. https://doi.org/10.1016/j.colsurfa.2013.01.007
Lee J, Kim J, Hyeon T (2006) Recent progress in the synthesis of porous carbon materials. Adv Mater 18:2073–2094. https://doi.org/10.1002/adma.200501576
Li Z, Jaroniec M (2003) Synthesis and adsorption properties of colloid-imprinted carbons with surface and volume mesoporosity. Chem Mater 15(6):1327–1333. https://doi.org/10.1021/cm020617a
Lippens BC, de Boer JH (1965) Studies on pore systems in catalysts: V. The t Method J Catal 4(3):319–323. https://doi.org/10.1016/0021-9517(65)90307-6
Liu Y, Goebl J, Yin Y (2013) Templated synthesis of nanostructured materials. Chem Soc Rev 42(7):2610–2653. https://doi.org/10.1039/c2cs35369e
Louis B, Bégin D, Ledoux M-J, Pham-Huu C (2009) Advances in the use of carbon nanomaterials in catalysis. In: Valtchev V, Mintova S, Tsapatsis M (eds) Ordered porous solids. Recent advances and prospects. Elsevier, Amsterdam, pp 621–649. https://doi.org/10.1016/B978-0-444-53189-6.00023-8
Mazumder B (2012) Coal structure and classification. In: Mazumder B (ed) Coal science and engineering. Woodhead Publishing India, New Delhi, pp 76–451. https://doi.org/10.1533/9780857098269.76
Pérez-Mayoral E, Calvino-Casilda V, Soriano E (2016) Metal-supported carbon-based materials: opportunities and challenges in the synthesis of valuable products. Catal Sci Technol 6:1265–1291. https://doi.org/10.1039/C5CY01437A
Pertko OP, Voloshyna YuG, Konovalov SV, Patrylak LK (2017) Deactivation of zeolite X based catalysts with active and ammonium hexafluorosilicate modified external surface in toluene methylation (in Ukrainian). Catal Petrochem 26:30–35
Pertko OP, Voloshyna YuG, Kontsevoi AL, Trachevsky VV (2021) Ethylbenzene formation and its conversion towards coke in the side-chain methylation of toluene on a basic X zeolite. J Porous Mater 28:1713–1723. https://doi.org/10.1007/s10934-021-01119-8
Rouquerol J, Llewellyn PL, Rouquerol F (2007) Is the BET equation applicable to microporous adsorbents? Stud Surf Sci Catal 160:49–56. https://doi.org/10.1016/S0167-2991%2807%2980008-5
Ryoo R, Joo SH, Kruk M, Jaroniec M (2001) Ordered mesoporous carbons. Adv Mater 13(9):677–681. https://doi.org/10.1002/1521-4095(200105)13:9%3C677::AID-ADMA677%3E3.0.CO;2-C
Schlögl R (2013) Carbon in catalysis. In: Gates BC, Jentoft FC (eds) Advances in catalysis. Academic Press, Amsterdam, pp 103–185. https://doi.org/10.1016/B978-0-12-420173-6.00002-4
Serp P, Corrias M, Kalck P (2003) Carbon nanotubes and nanofibers in catalysis. Appl Catal, A 253:337–358. https://doi.org/10.1016/S0926-860X(03)00549-0
Shcherban ND, Ilyin VG (2015) Matrix and bulk synthesis, activation and functionalization of nanoporous carbon adsorbents (in Ukrainian). Him Fiz Tehnol Poverhni 6(1):97–121. https://doi.org/10.15407/hftp06.01.097
Suárez-Ruiz I, Ward CR (2008) Coal combustion. In: Suárez-Ruiz I, Crelling JC (eds) Applied coal petrology. The role of petrology in coal utilization. Elsevier, Amsterdam, pp 85–117. https://doi.org/10.1016/B978-0-08-045051-3.00004-X
Tang W, Cao J-P, Wang Zh-H, He Z-M, Liu T-L, Wang Z-Y, Yang F-L, Ren J, Zhao X-Y, Feng X-B, Bai H-C (2021) Comparative evaluation of tar steam reforming over graphitic carbon supported Ni and Co catalysts at low temperature. Energy Convers Manage 244:114454. https://doi.org/10.1016/j.enconman.2021.114454
Thommes M, Kaneko K, Neimark AV, Olivier JP, Rodriguez-Reinoso F, Rouquerol J, Sing KSW (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Węgrzyniak A, Jarczewski S, Wach A, Hędrzak E, Kuśtrowski P, Michorczyk P (2015) Catalytic behaviour of chromium oxide supported on CMK-3 carbon replica in the dehydrogenation propane to propene. Appl Catal, A 508:1–9. https://doi.org/10.1016/j.apcata.2015.10.002
Zhang S, Yin H, Wang J, Zhu S, Xiong Y (2021) Catalytic cracking of biomass tar using Ni nanoparticles embedded carbon nanofiber/porous carbon catalysts. Energy 216:119285. https://doi.org/10.1016/j.energy.2020.119285
Zheng Ch, Chen C, Chen L, Wei M (2017) A CMK-5-encapsulated MoSe2 composite for rechargeable lithium-ion batteries with improved electrochemical performance. J Mater Chem, A 5(37):19632–19638. https://doi.org/10.1039/C7TA06286A
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial or non-financial interests or personal relationships that could impart bias on the work submitted for publication. The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Povazhnyi, V.A., Voloshyna, Y.G., Pertko, O.P. et al. Enhancing the thermal stability of nanostructured carbonaceous materials using an improved method of template synthesis. Appl Nanosci 13, 7491–7499 (2023). https://doi.org/10.1007/s13204-023-02908-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-023-02908-0