Abstract
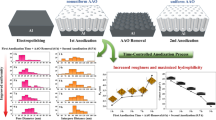
Surface modification of dental implants or orthopedic implants is important for biocompatibility, and Ti-6Al-4 V alloy is mainly used as implant material for clinical use. In particular, research on the formation of nanotubes for drug doping to improve biocompatibility is in progress. For the control of nanotube shape, in this study, nanotube shape changes on Ti-6Al-4 V alloy via various applied potential for bio-implants were researched using various instruments. The formation of nanotubes on the surface of the Ti-6Al-4 V alloy was performed using a DC power source as a two-electrode system, and platinum as a counter electrode and a working electrode as an anode. The electrolyte was 0.8 wt. % NaF solution and 1.0 M H3PO4 were used and mixed using a magnetic bar during anodization. At this time, the voltage applied when forming the nanotubes was varied as (10 → 20 → 30 V), (30 → 10 → 30 V), and (10 → 30 → 30 V), respectively, and these voltages were applied for 30 min for each step. After the formation of nanotubes, the surface morphology and structural properties of the samples were observed using field-emission scanning electron microscope, X-ray diffractometer, Fourier transform infrared spectroscopy, and nanoindentation tester. The higher the voltage applied during the formation of nanotubes, the larger is the diameter. In the case of the nanotube (10 → 20 → 30 V), the top of the nanotube was narrow and the diameter increased toward the bottom, forming a cone shape. In the case of the nanotube (30 → 10 → 30 V), a narrow waist shape was formed, and in the case of the nanotube (10 → 30 → 30 V), a jar shape was formed with anatase and rutile phases. From the FT-IR results, when nanotubes are formed, TiO2 anatase was detected and showed the tendency to change from crystallization to amorphous state. The indentation hardness and elastic modulus decreased for the nanotube surface compared with those of the bulk Ti alloy.







Similar content being viewed by others
Availability of data and material
(The data presented in this study are available on request from the corresponding author. The data are not publicly available as the data also forms part of an ongoing study.)
References
Anna M, Agata R, Kamil M, Marcin P, Marcin H, Piotr B, Malgorzata L (2021) Influence of microstructural features on the growth of nanotubular oxide layers on β-phase Ti-24Nb-4Zr-8Sn and α + β-phase Ti-13Nb-13Zr alloys. Surf Coat Technol 425:127695. https://doi.org/10.1016/j.surfcoat.2021.127695
Awad NK, Edwards SL, Morsi YS (2017) A review of TiO2 NTs on Ti metal: Electrochemical synthesis, functionalization and potential use as bone implants. Mater Sci Eng C Mater Biol Appl 76:1401–1412. https://doi.org/10.1016/j.msec.2017.02.150
Barbora V, Vojtech H, Veronika B, Josef S, Vera L, Karolina V, Vera S, Roman M, Jaroslav F, Ludek J, Matej D, Eva F (2019) Different diameters of titanium dioxide nanotubes modulate Saos-2 osteoblast-like cell adhesion and osteogenic differentiation and nanomechanical properties of the surface. RSC ADV 9(20):11341–11355. https://doi.org/10.1039/c9ra00761j
Cho HR, Choe HC (2021) Morphology of hydroxyapatite and Sr coatings deposited using radio frequency-magnetron sputtering method on nanotube formed Ti-6Al-4V alloy. Thin Solid Films 735:138893. https://doi.org/10.1016/j.tsf.2021.138893
Çırak BB, Karadeniz SM, Kılınç T, Caglar B, Ekinci AE, Yelgin H, Kürekçi M, Çırak Ç (2017) Synthesis, surface properties, crystal structure and dye sensitized solar cell performance of TiO2 nanotube arrays anodized under different voltages. Vacuum 144:183–189. https://doi.org/10.1016/j.rinp.2019.102609
Hanawa T (2019) Titanium–tissue interface reaction and its control with surface treatment. Front Bioeng Biotech. https://doi.org/10.3389/fbioe.2019.00170
Jo CI, Jeong YH, Choe HC, Brantley WA (2013) Hydroxyapatite precipitation on nanotubular films formed on Ti-6Al-4V alloy for biomedical applications. Thin Solid Films 549:135–140. https://doi.org/10.1016/j.tsf.2013.09.095
Kim SH, Choe HC (2014) Surface morphology of nanotube formed Ti alloy by electrochemical methods. J Nanosci Nanotechno 14:8372–8376. https://doi.org/10.1166/jnn.2014.9936
Kim HJ, Choe HC (2020) Magnesium, silicon, and hydroxyapatite deposition on the Ti-xNb-2Ag-2Pt alloy by co- sputtering after nanotube formation. Surf Coat Technol 404:126489. https://doi.org/10.1016/j.surfcoat.2020.126487
Kim SP, Cho HR, Choe HC (2021) Bioactive element coatings on nano-mesh formed Ti-6Al-4V alloy surface using plasma electrolytic oxidation. Surf Coat Technol 406:126649. https://doi.org/10.1016/j.surfcoat.2020.126649
Kunrath MF, Hubler R, Shinkai RSA, Teixeira ER (2018) Application of TiO2 nanotubes as a drug delivery system for biomedical implants: A Critical Overview. Chemistry Select 3(40):11180–11189. https://doi.org/10.1002/slct.201801459
Mosab K, Choe HC (2021) Simultaneous improvement of corrosion resistance and bioactivity of a titanium alloy via wet and dry plasma treatments. J Alloys Compd 851:156840. https://doi.org/10.1016/j.jallcom.2020.156840
Park SY, Choe HC (2016) Variations of nanotubes on the Ti–Nb–Hf alloys with applied voltages. Thin Solid Films 620:119–125. https://doi.org/10.1016/j.tsf.2016.09.061
Pravenn P, Viruthagiri G, Mugundan S, Shanmugam N (2014) Structural, optical, and morphological analyses of pristine titanium di-oxide nanoparticles – Synthesized via sol–gel route. Spectrochim Acta A 117:622–629. https://doi.org/10.1016/j.saa.2013.09.037
Rajakumar G, Rahuman AA, Roopan SM, Khanna VG, Elango G, Kamaraj C, Zahir AA, Velayutham K (2012) Fungus-mediated biosynthesis and characterization of TiO2 nanoparticles and their activity against pathogenic bacteria. Spectrochim Acta A 91:23–29. https://doi.org/10.1016/j.saa.2012.01.011
Yeo ISL (2020) Modifications of dental implant surfaces at the micro-and nano-level for enhanced osseointegration. Materials 13:89. https://doi.org/10.3390/ma13010089
Zhang W, Liu Y, Guo F, Liu J, Yang F (2019) Kinetic analysis of the anodic growth of TiO2 nanotubes: effects of voltage and temperature. J Mater Chem C 7(45):14098–14108. https://doi.org/10.1039/C9TC04532E
Zhang E, Zhao X, Hu J, Wang R, Fu S, Qin G (2021) Antibacterial metals and alloys for potential biomedical implants. Bioact Mater 6(8):2569–2612. https://doi.org/10.1016/j.bioactmat.2021.01.030
Zuncen Z, Frank MA, Yuyun Y, Sannakaisa V, Aldo RB (2018) A novel local drug delivery system: Superhydrophobic titanium oxide nanotube arrays serve as the drug reservoir and ultrasonication functions as the drug release trigger. Mater Sci Eng C Mater 82:277–283. https://doi.org/10.1016/j.msec.2017.08.066
Funding
(This research was supported by NRF: 2021R1A4A1030243).
Author information
Authors and Affiliations
Contributions
BSL: Formal analysis, writing, data collection, data analysis, data interpretation, HRC: data analysis, data interpretation, HCC: writing, review and editing, funding acquisition, study design, data collection, data analysis, data interpretation, resources, supervision.
Corresponding author
Ethics declarations
Conflicts of interest
(The authors declare no conflict of interest).
Ethics approval
The authors declare that they have no known compet ing fnancial interests, ethical interests, or personal relationships that could have appeared to infuence work reported in this paper.
Informed consent statement
Informed consent was obtained from all subjects involved in the study. The data presented in this study are available on request from the corresponding author.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lim, BS., Cho, HR. & Choe, HC. Nanotube shape changes on Ti-6Al-4 V alloy via various applied potential for bio-implants. Appl Nanosci 12, 3329–3336 (2022). https://doi.org/10.1007/s13204-022-02541-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-022-02541-3