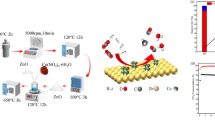
Abstract
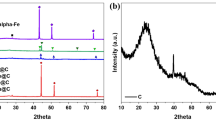
Pristine Co3O4 and Co3O4–halloysite nanotubes (Co3O4@HNTs) composite were synthesized and characterized using XRD, HR-TEM, Raman, and ATR–FTIR. Then, the catalytic activity of these materials towards dry methane reforming (DMR) either, the direct or indirect pathways were analyzed, through either direct or indirect pathways. The analysis was focused on the influence of different parameters, such as temperature, solid-phase mixture, and feeding gas stream composition. Results showed that both materials have bifunctional catalytic behavior. First, they showed high selectivity towards CO oxidation instead of methane oxidation, and subsequently, these materials also participated in the reaction between CH4 and CO2 to produce H2. The presence of oxygen in the gas stream, as well as the use of the halloysite as support, proved to be determining factors to prevent the formation of coke or metallic cobalt on the catalyst, which increases the H2 production. Finally, the highest H2 production was obtained using the Co3O4@HNTs composite under a gas stream composed of CH4/CO/O2 at 650 °C.









Similar content being viewed by others
References
Abdelkader A, Daly H, Saih Y et al (2013) Steam reforming of ethanol over Co3O4–Fe2O3 mixed oxides. Int J Hydrogen Energy 38:8263–8275. https://doi.org/10.1016/j.ijhydene.2013.04.009
Adach W, Błaszczyk M, Olas B (2020) Carbon monoxide and its donors—chemical and biological properties. Chem Biol Interact 318:108973. https://doi.org/10.1016/j.cbi.2020.108973
Ali F, Khalid NR (2020) Facile synthesis and properties of chromium-doped cobalt oxide (Cr-doped Co3O4) nanostructures for supercapacitor applications. Appl Nanosci 10:1481–1488. https://doi.org/10.1007/s13204-020-01266-5
Alsaffar MA, Ayodele BV, Mustapa SI (2020) Scavenging carbon deposition on alumina supported cobalt catalyst during renewable hydrogen-rich syngas production by methane dry reforming using artificial intelligence modeling technique. J Clean Prod 247:119168. https://doi.org/10.1016/j.jclepro.2019.119168
Ballarini AD, de Miguel SR, Jablonski EL et al (2005) Reforming of CH4 with CO2 on Pt-supported catalysts: effect of the support on the catalytic behaviour. Catal Today 107–108:481–486. https://doi.org/10.1016/j.cattod.2005.07.058
Bahari MB, Nguyen TD, Singh S et al (2019) Catalytic performance of yttrium-doped co/mesoporous alumina catalysts for methane dry reforming. AIP Conf Proc 2124:20018. https://doi.org/10.1063/1.5117078
Bergay PYATF (ed) (2016) Nanosized tubular clay minerals, 7th edn. Elsevier
Carrero A, Calles JA, Vizcaíno AJ (2010) Effect of Mg and Ca addition on coke deposition over Cu–Ni/SiO2 catalysts for ethanol steam reforming. Chem Eng J 163:395–402. https://doi.org/10.1016/j.cej.2010.07.029
Carrillo AM, Urruchurto CM, Carriazo JG et al (2014) Caracterización estructural y textural de una haloisita colombiana. Revista Mex Ing Química 13:563–571
Cavallaro G, Lazzara G, Massaro M et al (2015) Biocompatible Poly(N-isopropylacrylamide)-halloysite Nanotubes for Thermoresponsive Curcumin Release. J Phys Chem C 119:8944–8951. https://doi.org/10.1021/acs.jpcc.5b00991
Chen W, Zhao G, Xue Q et al (2013) High carbon-resistance Ni/CeAlO3-Al2O3 catalyst for CH4/CO2 reforming. Appl Catal B Environ 136–137:260–268. https://doi.org/10.1016/j.apcatb.2013.01.044
Chen J, Xu X, Feng L et al (2020a) One-step MOF assisted synthesis of SmVO4 nanorods for photocatalytic degradation of tetracycline under visible light. Mater Lett 276:128213. https://doi.org/10.1016/j.matlet.2020.128213
Chen Y, Chen T, Liu H et al (2020b) High catalytic performance of the Al-promoted Ni/Palygorskite catalysts for dry reforming of methane. Appl Clay Sci 188:105498. https://doi.org/10.1016/j.clay.2020b.105498
Chong CC, Bukhari SN, Cheng YW et al (2019) Robust Ni/Dendritic fibrous SBA-15 (Ni/DFSBA-15) for methane dry reforming: effect of Ni loadings. Appl Catal A Gen 584:117174. https://doi.org/10.1016/j.apcata.2019.117174
Ciambelli P, Cimino S, De Rossi S et al (2001) AFeO3 (A=La, Nd, Sm) and LaFe1−xMgxO3 perovskites as methane combustion and CO oxidation catalysts: structural, redox and catalytic properties. Appl Catal B Environ 29:239–250. https://doi.org/10.1016/S0926-3373(00)00215-0
Ciambelli P, Cimino S, Lasorella G et al (2002) CO oxidation and methane combustion on LaAl1−xFexO3 perovskite solid solutions. Appl Catal B Environ 37:231–241. https://doi.org/10.1016/S0926-3373(02)00004-8
Compaan A (1975) Surface damage effects on allowed and forbidden phonon Raman scattering in cuprous oxide. Solid State Commun 16:293–296. https://doi.org/10.1016/0038-1098(75)90171-4
Compaan A, Cummins HZ (1972) Raman scattering, luminescence, and exciton-phonon coupling in Cu2O. Phys Rev B 6:4753–4757. https://doi.org/10.1103/PhysRevB.6.4753
Dai H, Yu P, Liu H et al (2020) Ni-Based catalysts supported on natural clay of attapulgite applied in the dry reforming of methane reaction. New J Chem 44:16101–16109. https://doi.org/10.1039/D0NJ03069D
Das S, Shah M, Gupta RK, Bordoloi A (2019) Enhanced dry methane reforming over Ru decorated mesoporous silica and its kinetic study. J CO2 Util 29:240–253. https://doi.org/10.1016/j.jcou.2018.12.016
Daza-Gómez L-C, Ruiz-Ruiz V-F, Mendoza-Nieto JA et al (2020) Co3O4 nanostructures and Co3O4 supported on halloysite nanotubes: new highly active and thermally stable feasible catalysts for CO oxidation. Appl Clay Sci 190:105590. https://doi.org/10.1016/j.clay.2020.105590
Dey S, Dhal GC (2019) Highly active palladium nanocatalysts for low-temperature carbon monoxide oxidation. Polytechnica. https://doi.org/10.1007/s41050-019-00018-x
Dey S, Dhal GC (2020) Property and structure of various platinum catalysts for low-temperature carbon monoxide oxidations. Mater Today Chem 16:100228. https://doi.org/10.1016/j.mtchem.2019.100228
Djinović P, Črnivec IGO, Batista J et al (2011) Catalytic syngas production from greenhouse gasses: performance comparison of Ru-Al2O3 and Rh-CeO2 catalysts. Chem Eng Process Process Intensif 50:1054–1062. https://doi.org/10.1016/j.cep.2011.05.018
Elvidge CD, Bazilian MD, Zhizhin M et al (2018) The potential role of natural gas flaring in meeting greenhouse gas mitigation targets. Energy Strateg Rev 20:156–162. https://doi.org/10.1016/j.esr.2017.12.012
Farhang Y, Taheri-Nassaj E, Rezaei M (2018) Pd doped LaSrCuO4 perovskite nano-catalysts synthesized by a novel solid state method for CO oxidation and Methane combustion. Ceram Int 44:21499–21506. https://doi.org/10.1016/j.ceramint.2018.08.211
Ferrante F, Armata N, Lazzara G (2015) Modeling of the halloysite spiral nanotube. J Phys Chem C 119:16700–16707. https://doi.org/10.1021/acs.jpcc.5b04281
Freitas ACD, Guirardello R (2014) Thermodynamic analysis of methane reforming with CO2, CO2+H2O, CO2+O2 and CO2+air for hydrogen and synthesis gas production. J CO2 Util 7:30–38. https://doi.org/10.1016/j.jcou.2014.06.004
Hampson NB, Hauschildt KL, Deru K, Weaver LK (2019) Carbon monoxide poisonings in hotels and motels: the problem silently continues. Prev Med Reports 16:100975. https://doi.org/10.1016/j.pmedr.2019.100975
Han B, Zhao L, Wang F et al (2020) Effect of calcination temperature on the performance of the Ni@SiO2 catalyst in methane dry reforming. Ind Eng Chem Res 59:13370–13379. https://doi.org/10.1021/acs.iecr.0c01213
Hazlett MJ, Epling WS (2020) Mechanistic effects of Water on Carbon monoxide and propylene oxidation on platinum and palladium bimetallic catalysts. Catal Today. https://doi.org/10.1016/j.cattod.2020.01.024
Hernández-Fontes C, Mendoza-Nieto JA, Lara-García HA, Pfeiffer H (2019) Pentalithium ferrite (Li5FeO4) as highly active material for hydrogen production in the chemical looping partial oxidation of methane. Top Catal 62:884–893. https://doi.org/10.1007/s11244-019-01175-0
Hou Z, Chen P, Fang H et al (2006) Production of synthesis gas via methane reforming with CO2 on noble metals and small amount of noble-(Rh-) promoted Ni catalysts. Int J Hydrogen Energy 31:555–561. https://doi.org/10.1016/j.ijhydene.2005.06.010
Hu D, Gao J, Ping Y et al (2012) Enhanced investigation of CO methanation over Ni/Al2O3 catalysts for synthetic natural gas production. Ind Eng Chem Res 51:4875–4886. https://doi.org/10.1021/ie300049f
Huang T, Huang W, Huang J, Ji P (2011) Methane reforming reaction with carbon dioxide over SBA-15 supported Ni–Mo bimetallic catalysts. Fuel Process Technol 92:1868–1875. https://doi.org/10.1016/j.fuproc.2011.05.002
Jang W-J, Shim J-O, Kim H-M et al (2019) A review on dry reforming of methane in aspect of catalytic properties. Catal Today 324:15–26. https://doi.org/10.1016/j.cattod.2018.07.032
Ji Y, Zhao Z, Duan A et al (2009) Comparative study on the formation and reduction of bulk and Al2O3-supported cobalt oxides by H2-TPR technique. J Phys Chem C 113:7186–7199. https://doi.org/10.1021/jp8107057
Jones G, Jakobsen JG, Shim SS et al (2008) First principles calculations and experimental insight into methane steam reforming over transition metal catalysts. J Catal 259:147–160. https://doi.org/10.1016/j.jcat.2008.08.003
Kehres J, Jakobsen JG, Andreasen JW et al (2012) Dynamical properties of a Ru/MgAl2O4 catalyst during reduction and dry methane reforming. J Phys Chem C 116:21407–21415. https://doi.org/10.1021/jp3069656
Kerkar RD, Salker AV (2021) Highly active nano-composite of cobalt–copper–manganese oxides for room temperature CO oxidation. Appl Nanosci 11:2861–2867. https://doi.org/10.1007/s13204-021-02232-5
Kinoshita H, Türkan H, Vucinic S et al (2020) Carbon monoxide poisoning. Toxicol Reports 7:169–173. https://doi.org/10.1016/j.toxrep.2020.01.005
Köck E-M, Kogler M, Bielz T et al (2013) In situ FT-IR spectroscopic study of CO2 and CO adsorption on Y2O3, ZrO2, and yttria-stabilized ZrO2. J Phys Chem C 117:17666–17673. https://doi.org/10.1021/jp405625x
Li W-J, Wey M-Y (2020a) Design of a thermally resistant core@shell/halloysite catalyst with optimized structure and surface properties for a Pd-only three-way catalyst. Appl Catal A Gen 602:117732. https://doi.org/10.1016/j.apcata.2020a.117732
Li W-J, Wey M-Y (2020b) Sintering-resistant, highly thermally stable and well-dispersed Pd@CeO2/halloysite as an advanced three-way catalyst. Sci Total Environ 707:136137. https://doi.org/10.1016/j.scitotenv.2019.136137
Li L, Chen J, Zhang Q et al (2020) Methane dry reforming over activated carbon supported Ni-catalysts prepared by solid phase synthesis. J Clean Prod 274:122256. https://doi.org/10.1016/j.jclepro.2020.122256
Liang T-Y, Senthil Raja D, Chin KC et al (2020) Bimetallic metal-organic framework-derived hybrid nanostructures as high-performance catalysts for methane dry reforming. ACS Appl Mater Interfaces 12:15183–15193. https://doi.org/10.1021/acsami.0c00086
Liu BS, Au CT (2003) Carbon deposition and catalyst stability over La2NiO4/γ-Al2O3 during CO2 reforming of methane to syngas. Appl Catal A Gen 244:181–195. https://doi.org/10.1016/S0926-860X(02)00591-4
Liu S, Guan L, Li J et al (2008) CO2 reforming of CH4 over stabilized mesoporous Ni–CaO–ZrO2 composites. Fuel 87:2477–2481. https://doi.org/10.1016/j.fuel.2008.02.009
Liu D, Cheo WNE, Lim YWY et al (2010) A comparative study on catalyst deactivation of nickel and cobalt incorporated MCM-41 catalysts modified by platinum in methane reforming with carbon dioxide. Catal Today 154:229–236. https://doi.org/10.1016/j.cattod.2010.03.054
Liu H, Hadjltaief HB, Benzina M et al (2019) Natural clay based nickel catalysts for dry reforming of methane: on the effect of support promotion (La, Al, Mn). Int J Hydrogen Energy 44:246–255. https://doi.org/10.1016/j.ijhydene.2018.03.004
Lorite I, Romero JJ, Fernández JF (2012) Effects of the agglomeration state on the Raman properties of Co3O4 nanoparticles. J Raman Spectrosc 43:1443–1448. https://doi.org/10.1002/jrs.4098
Luo P, Zhang J, Zhang B et al (2011) Preparation and characterization of silane coupling agent modified halloysite for Cr(VI) removal. Ind Eng Chem Res 50:10246–10252. https://doi.org/10.1021/ie200951n
Massaro M, Colletti CG, Lazzara G et al (2017) Halloysite nanotubes as support for metal-based catalysts. J Mater Chem A 5:13276–13293. https://doi.org/10.1039/C7TA02996A
Mendoza-Nieto JA, Puente-Lee I, Salcedo-Luna C, Klimova T (2012) Effect of titania grafting on behavior of NiMo hydrodesulfurization catalysts supported on different types of silica. Fuel 100:100–109. https://doi.org/10.1016/j.fuel.2012.02.005
Mendoza-Nieto JA, Duan Y, Pfeiffer H (2018a) Alkaline zirconates as effective materials for hydrogen production through consecutive carbon dioxide capture and conversion in methane dry reforming. Appl Catal B Environ 238:576–585. https://doi.org/10.1016/j.apcatb.2018a.07.065
Mendoza-Nieto JA, Tehuacanero-Cuapa S, Arenas-Alatorre J, Pfeiffer H (2018b) Nickel-doped sodium zirconate catalysts for carbon dioxide storage and hydrogen production through dry methane reforming process. Appl Catal B Environ 224:80–87. https://doi.org/10.1016/j.apcatb.2017.10.050
Monroy TG, Abella LC, Gallardo SM, Hinode H (2010) Catalytic dry reforming of methane using Ni/MgO-ZrO2 catalyst. In: Benyahia F, Eljack FTBT-P of the 2nd AGPS (eds) Advances in Gas processing. Elsevier, Amsterdam, pp 145–152
Osazuwa OU, Setiabudi HD, Rasid RA, Cheng CK (2017) Syngas production via methane dry reforming: a novel application of SmCoO3 perovskite catalyst. J Nat Gas Sci Eng 37:435–448. https://doi.org/10.1016/j.jngse.2016.11.060
Papoulis D (2019) Halloysite based nanocomposites and photocatalysis: a review. Appl Clay Sci 168:164–174. https://doi.org/10.1016/j.clay.2018.11.009
Pham Minh D, Pham X-H, Siang TJ, N. Vo D-V, (2021) Review on the catalytic tri-reforming of methane—Part I: impact of operating conditions, catalyst deactivation and regeneration. Appl Catal A Gen 621:118202. https://doi.org/10.1016/j.apcata.2021.118202
Pinheiro AN, Valentini A, Sasaki JM, Oliveira AC (2009) Highly stable dealuminated zeolite support for the production of hydrogen by dry reforming of methane. Appl Catal A Gen 355:156–168. https://doi.org/10.1016/j.apcata.2008.12.007
Pudukudy M, Kadier A, Yaakob Z, Takriff MS (2016) Non-oxidative thermocatalytic decomposition of methane into COx free hydrogen and nanocarbon over unsupported porous NiO and Fe2O3 catalysts. Int J Hydrogen Energy 41:18509–18521. https://doi.org/10.1016/j.ijhydene.2016.08.160
Rezaei E, Dzuryk S (2019) Techno-economic comparison of reverse water gas shift reaction to steam and dry methane reforming reactions for syngas production. Chem Eng Res Des 144:354–369. https://doi.org/10.1016/j.cherd.2019.02.005
Rood SC, Ahmet HB, Gomez-Ramon A et al (2019) Enhanced ceria nanoflakes using graphene oxide as a sacrificial template for CO oxidation and dry reforming of methane. Appl Catal B Environ 242:358–368. https://doi.org/10.1016/j.apcatb.2018.10.011
Royer S, Duprez D (2011) Catalytic oxidation of carbon monoxide over transition metal oxides. ChemCatChem 3:24–65. https://doi.org/10.1002/cctc.201000378
Ruckenstein E, Wang HY (2002) Carbon deposition and catalytic deactivation during CO2 reforming of CH4 over Co/γ-Al2O3 catalysts. J Catal 205:289–293. https://doi.org/10.1006/jcat.2001.3458
Shalini Arora RP (2016) An overview on dry reforming of methane: strategies to reduce carbonaceous deactivation of catalysts. RSC Adv. https://doi.org/10.1039/C6RA20450C
Shen J, Reule AAC, Semagina N (2019) Ni/MgAl2O4 catalyst for low-temperature oxidative dry methane reforming with CO2. Int J Hydrogen Energy 44:4616–4629. https://doi.org/10.1016/j.ijhydene.2019.01.027
Sie M-C, Jeng P-D, Chen P-H et al (2017) Evaluation of CO oxidation over Co3O4-supported NiO catalysts. AIP Conf Proc 1877:70004. https://doi.org/10.1063/1.4999890
Song Z, Wang Q, Guo C et al (2020) Improved effect of Fe on the stable NiFe/Al2O3 catalyst in low-temperature dry reforming of methane. Ind Eng Chem Res 59:17250–17258. https://doi.org/10.1021/acs.iecr.0c01204
Usman M, Wan Daud WMA, Abbas HF (2015) Dry reforming of methane: influence of process parameters—a review. Renew Sustain Energy Rev 45:710–744. https://doi.org/10.1016/j.rser.2015.02.026
Wang HY, Ruckenstein E (2001) CO2 reforming of CH4 over Co/MgO solid solution catalysts—effect of calcination temperature and Co loading. Appl Catal A Gen 209:207–215. https://doi.org/10.1016/S0926-860X(00)00753-5
Wang F, Xu L, Zhang J et al (2016) Tuning the metal-support interaction in catalysts for highly efficient methane dry reforming reaction. Appl Catal B Environ 180:511–520. https://doi.org/10.1016/j.apcatb.2015.07.001
Wang H, Su Y, Wang D et al (2020) Optimal design and energy-saving investigation of the triple CO2 feeds for methanol production system by combining steam and dry methane reforming. Ind Eng Chem Res. https://doi.org/10.1021/acs.iecr.9b05296
Wei Y, Song M, Yu L et al (2018) Promotion effect of SiO2 on the catalytic performance of Ni/CF for biomass derived gas reforming. Ind Eng Chem Res 57:10851–10858. https://doi.org/10.1021/acs.iecr.8b01452
Yañez-Aulestia A, Gómez-García JF, Mendoza-Nieto JA et al (2018) Thermocatalytic analysis of CO2-CO selective chemisorption mechanism on lithium cuprate (Li2CuO2) and oxygen addition effect. Thermochim Acta 660:144–151. https://doi.org/10.1016/j.tca.2017.12.027
Yuan P, Southon PD, Liu Z et al (2008) Functionalization of halloysite clay nanotubes by grafting with γ-aminopropyltriethoxysilane. J Phys Chem C 112:15742–15751. https://doi.org/10.1021/jp805657t
Yuan P, Tan D, Nnabi-Bergaya F, Yan W, Fan M, Liu D, He H (2012) Changes in structure, morphology, porosity, and surface activity of mesoporous halloysite nanotubes under heating. Clays Clay Miner 60(6):561–573
Zeng S, Fu X, Zhou T et al (2013) Influence of pore distribution on catalytic performance over inverse CeO2/Co3O4 catalysts for CH4/CO2 reforming. Fuel Process Technol 114:69–74. https://doi.org/10.1016/j.fuproc.2013.03.040
Zhang J, Wang H, Dalai AK (2008) Effects of metal content on activity and stability of Ni-Co bimetallic catalysts for CO2 reforming of CH4. Appl Catal A Gen 339:121–129. https://doi.org/10.1016/j.apcata.2008.01.027
Zhao Z, Ren P, Li W (2016) Supported Ni catalyst on a natural halloysite derived silica–alumina composite oxide with unexpected coke-resistant stability for steam-CO2 dual reforming of methane. RSC Adv 6:49487–49496. https://doi.org/10.1039/C6RA09203A
Zhao X, Joseph B, Kuhn J, Ozcan S (2020) Biogas reforming to syngas: a review. iScience 23:101082. https://doi.org/10.1016/j.isci.2020.101082
Zhong L, Kropp T, Baaziz W et al (2019) Correlation between reactivity and oxidation state of cobalt oxide catalysts for CO preferential oxidation. ACS Catal 9:8325–8336. https://doi.org/10.1021/acscatal.9b02582
Acknowledgements
C. Daza-Gómez thanks CONACYT for the Ph.D. scholarship (595211). D. Diaz thanks FQUNAM for the 5000-90-39 PAIP grant. J. Arturo Mendoza-Nieto thanks PAPIIT-UNAM IA-106321 and PAIP 5000-91-77 grant. We thank Ivan Puente (UNAM) for his technical assistance in HR-TEM and SEM techniques, and M. en C. Viridiana Maturano Rojas (Instituto de Ciencias Aplicadas y Tecnología, ICAT) in H2-TPR profiles.
Author information
Authors and Affiliations
Contributions
The manuscript was written through the contributions of all authors. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Daza-Gómez, LC., Ruiz-Ruiz, VF., Mendoza-Nieto, J.A. et al. Investigation of H2 production via an integrated pathway of consecutive CO oxidation and dry methane reforming in the presence of Co3O4@HNTs catalyst. Appl Nanosci 12, 2459–2476 (2022). https://doi.org/10.1007/s13204-022-02510-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-022-02510-w