Abstract
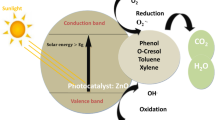
In this study, TiO2 nanoparticles (NPs) and TiO2 nanowires (NWs) were synthesized via modified sol–gel and hydrothermal methods, respectively. The TiO2 NPs and TiO2 NWs were characterized employing various analytical techniques. The performance of TiO2 NPs and TiO2 NWs was evaluated and compared for phenols’ removal from model oil refinery wastewater under visible light irradiation. The TiO2 NPs and TiO2 NWs were pure anatase crystalline phases with average particle size of 10.17 nm and 6.82 nm and a narrow bandgap of 2.41 and 2.66 eV, respectively. 3D view surface plots confirm that the long-range atomic arrangement of the atom was intact, indicated by atomically flat surfaces. The recombination rate of electrons and holes was significantly lower in TiO2 NWs compared to TiO2 NPs. Both the samples were able to remove more than 90% of phenols from model oil refinery wastewater. Phenol removal from neutralized spent caustic took a longer time (300 min) compared to desalter effluent and tank water drain (<180 min). Phenol removal efficiency increased when TiO2 NPs and TiO2 NWs were employed combinedly.. TiO2 NWs were highly stable, as suggested by their performance for photocatalytic removal of phenols after five consecutive cycles.











Similar content being viewed by others
References
Abdelnasser S, Al Sakkaf R, Palmisano G (2021) Environmental and energy applications of TiO2 photoanodes modified with alkali metals and polymers. J Environ Chem Eng 9:104873. https://doi.org/10.1016/j.jece.2020.104873
Abdelwahab O, Amin NK, El-Ashtoukhy ESZ (2009) Electrochemical removal of phenol from oil refinery wastewater. J Hazard Mater 163:711–716. https://doi.org/10.1016/j.jhazmat.2008.07.016
Al Hashemi W, Maraqa MA, Rao MV, Hossain MM (2015) Characterization and removal of phenolic compounds from condensate-oil refinery wastewater. Desalin Water Treat 54:660–671. https://doi.org/10.1080/19443994.2014.884472
Al Zarooni M, Elshorbagy W (2006) Characterization and assessment of Al Ruwais refinery wastewater. J Hazard Mater 136:398–405. https://doi.org/10.1016/j.jhazmat.2005.09.060
Amano F, Nakata M, Yamamoto A, Tanaka T (2016) Effect of Ti3+ ions and conduction band electrons on photocatalytic and photoelectrochemical activity of rutile titania for water oxidation. J Phys Chem C 120:6467–6474. https://doi.org/10.1021/acs.jpcc.6b01481
Anku WW, Mamo MA, Govender PP (2017) Phenolic compounds in water: sources, reactivity, toxicity and treatment methods. Phenolic compounds-natural sources, importance and applications. IntechOpen, London, pp 419–443. https://doi.org/10.5772/66927
Arano J, Nieto JM, Melián JH, Rodrıguez JD, Dıaz OG, Peña JP, Bergasa O, Alvarez C, Méndez J (2004) Photocatalytic degradation of formaldehyde containing wastewater from veterinarian laboratories. Chemosphere 55:893–904. https://doi.org/10.1016/j.chemosphere.2003.11.060
Ariyanti D, Mills L, Dong J, Yao Y, Gao W (2017) NaBH4 modified TiO2: defect site enhancement related to its photocatalytic activity. Mater Chem Phys 199:571–576. https://doi.org/10.1016/j.matchemphys.2017.07.054
Asaithambi P, Sajjadi B, Abdul Aziz AR, Daud WM (2017) Ozone (O3) and sono (US) based advanced oxidation processes for the removal of color, COD and determination of electrical energy from landfill leachate. Sep Purif Technol 172:442–449. https://doi.org/10.1016/j.seppur.2016.08.041
Bavykin DV, Cressey BA, Light ME, Walsh FC (2008) An aqueous, alkaline route to titanate nanotubes under atmospheric pressure conditions. Nanotechnology 19:275604
Bharagava RN, Saxena G, Mulla SI (2020) Introduction to industrial wastes containing organic and inorganic pollutants and bioremediation approaches for environmental management. In: Saxena G, Kishor R, Bharagava RN (eds) Bioremediation of industrial waste for environmental safety. Springer, New York, pp 1–18. https://doi.org/10.1007/978-981-13-1891-7_1
Casa R, D’Annibale A, Pieruccetti F, Stazi SR, Sermanni GG, Cascio BL (2003) Reduction of the phenolic components in olive-mill wastewater by an enzymatic treatment and its impact on durum wheat (Triticum durum Desf.) germinability. Chemosphere 50:959–966. https://doi.org/10.1016/S0045-6535(02)00707-5
Chantho P, Musikavong C, Suttinun O (2016) Removal of phenolic compounds from palm oil mill effluent by thermophilic Bacillus thermoleovorans strain A2 and their effect on anaerobic digestion. Int Biodeterior Biodegradation 115:293–301. https://doi.org/10.1016/j.ibiod.2016.09.010
Chen LY, Cheng CW, Liang JY (2015a) Effect of esterification condensation on the Folin-Ciocalteu method for the quantitative measurement of total phenols. Food Chem 170:10–15. https://doi.org/10.1016/j.foodchem.2014.08.038
Chen X, Liu L, Huang F (2015b) Black titanium dioxide (TiO2) nanomaterials. Chem Soc Rev 44:1861–1885. https://doi.org/10.1039/C4CS00330F
Clément L, Hurel C, Marmier N (2013) Toxicity of TiO2 nanoparticles to cladocerans, algae, rotifers and plants–effects of size and crystalline structure. Chemosphere 90:1083–1090. https://doi.org/10.1016/j.chemosphere.2012.09.013
D’Annibale A, Casa R, Pieruccetti F, Ricci M, Marabottini R (2004) Lentinula edodes removes phenols from olive-mill wastewater: impact on durum wheat (Triticum durum Desf.) germinability. Chemosphere 54:887–894. https://doi.org/10.1016/j.chemosphere.2003.10.010
David B (2009) Sonochemical degradation of PAH in aqueous solution. Part I: monocomponent PAH solution. Ultrason Sonochem 16:260–265. https://doi.org/10.1016/j.ultsonch.2008.07.013
Do TC, Nguyen DQ, Nguyen KT, Le PH (2019) TiO2 and Au-TiO2 nanomaterials for rapid photocatalytic degradation of antibiotic residues in aquaculture wastewater. Materials 12:2434. https://doi.org/10.3390/ma12152434
Ergül FE, Sargın S, Öngen G, Sukan FV (2011) Dephenolization and decolorization of olive mill wastewater through sequential batch and co-culture applications. World J Microbiol Biotechnol 27:107–114. https://doi.org/10.1007/s11274-010-0433-4
Fonseca CR, Paiva JL, Rodriguez EM, Beltran FJ, Teixeira AC (2017) Degradation of phenolic compounds in aqueous sucrose solutions by ozonation. Ozone Sci Eng 39:255–263. https://doi.org/10.1080/01919512.2017.1322486
Fuentes KM, Venuti D, Betancourt P (2020) Black titania with increased defective sites for phenol photodegradation under visible light. React Kinet 131:423–435. https://doi.org/10.1007/s11144-020-01832-6
Guo H, Zheng Z, Chen J, Weng W, Huang M (2016) Facile template-free one-pot fabrication of TiO2@C microspheres with high visible-light photocatalytic degradation activity. J Ind Eng Chem 36:306–313. https://doi.org/10.1016/j.jiec.2016.02.018
Herrmann JM (1999) Heterogeneous photocatalysis: fundamentals and applications to the removal of various types of aqueous pollutants. Catal Today 53:115–129. https://doi.org/10.1016/S0920-5861(99)00107-8
Hu A, Zhang X, Oakes KD, Peng P, Zhou YN, Servos MR (2011) Hydrothermal growth of free standing TiO2 nanowire membranes for photocatalytic degradation of pharmaceuticals. J Hazard Mater 189:278–285. https://doi.org/10.1016/j.jhazmat.2011.02.033
Hu A, Liang R, Zhang X, Kurdi S, Luong D, Huang H, Peng P, Marzbanrad E, Oakes KD, Zhou Y, Servos MR (2013) Enhanced photocatalytic degradation of dyes by TiO2 nanobelts with hierarchical structures. J Photochem Photobiol A Chem 256:7–15. https://doi.org/10.1016/j.jphotochem.2013.01.015
Ilie AG, Scarisoareanu M, Morjan I, Dutu E, Badiceanu M, Mihailescu I (2017) Principal component analysis of Raman spectra for TiO2 nanoparticle characterization. Appl Surf Sci 417:93–103. https://doi.org/10.1016/j.apsusc.2017.01.193
Ji L, Zhang Y, Miao S, Gong M, Liu X (2017) In situ synthesis of carbon doped TiO2 nanotubes with an enhanced photocatalytic performance under UV and visible light. Carbon 125:544–550. https://doi.org/10.1016/j.carbon.2017.09.094
Jusoh N, Rosly MB, Othman N, Rahman HA, Noah NF, Sulaiman RN (2020) Selective extraction and recovery of polyphenols from palm oil mill sterilization condensate using emulsion liquid membrane process. Environ Sci Pollut Res 27:23246–23257. https://doi.org/10.1007/s11356-020-07972-5
Khan ME, Khan MM, Min BK, Cho MH (2018) Microbial fuel cell assisted band gap narrowed TiO2 for visible light-induced photocatalytic activities and power generation. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-19617-2
Khongkhaem P, Suttinun O, Intasiri A, Pinyakong O, Luepromchai E (2016) Degradation of phenolic compounds in palm oil mill effluent by silica-immobilized bacteria in internal loop airlift bioreactors. Clean: Soil, Air, Water 44:383–392. https://doi.org/10.1002/clen.201300853
Kong L, Wang C, Zheng H, Zhang X, Liu Y (2015) Defect-induced yellow color in Nb-doped TiO2 and its impact on visible-light photocatalysis. J Phys Chem C 119:16623–16632. https://doi.org/10.1021/acs.jpcc.5b03448
Lee SM, Kim KS, Pippel E, Kim S, Kim JH, Lee HJ (2012) Facile route toward mechanically stable superhydrophobic copper using oxidation-reduction induced morphology changes. J Phys Chem C 116:2781–2790. https://doi.org/10.1021/jp2109626
Magalhaes LM, Santos F, Segundo MA, Reis S, Lima JL (2010) Rapid microplate high-throughput methodology for assessment of Folin-Ciocalteu reducing capacity. Talanta 83:441–447. https://doi.org/10.1016/j.talanta.2010.09.042
Martín MMB, Pérez JAS, Fernández FGA, Sánchez JLG, López JLC, Rodríguez SM (2008) A kinetics study on the biodegradation of synthetic wastewater simulating effluent from an advanced oxidation process using Pseudomonas putida CECT 324. J Hazard Mater 151:780–788. https://doi.org/10.1016/j.jhazmat.2007.06.053
Miklos DB, Remy C, Jekel M, Linden KG, Drewes JE, Hübner U (2018) Evaluation of advanced oxidation processes for water and wastewater treatment–a critical review. Water Res 139:118–131. https://doi.org/10.1016/j.watres.2018.03.042
Moya A, Cherevan A, Marchesan S, Gebhardt P, Prato M, Eder D, Vilatela JJ (2015) Oxygen vacancies and interfaces enhancing photocatalytic hydrogen production in mesoporous CNT/TiO2 hybrids. Appl Catal B Environ 179:574–582. https://doi.org/10.1016/j.apcatb.2015.05.052
Mu R, Xu Z, Li L, Shao Y, Wan H, Zheng S (2010) On the photocatalytic properties of elongated TiO2 nanoparticles for phenol degradation and Cr(VI) reduction. J Hazard Mater 176:495–502. https://doi.org/10.1016/j.jhazmat.2009.11.057
Myung ST, Kikuchi M, Yoon CS, Yashiro H, Kim SJ, Sun YK, Scrosati B (2013) Black anatase titania enabling ultra high cycling rates for rechargeable lithium batteries. Energy Environ Sci 6:2609–2614. https://doi.org/10.1039/C3EE41960F
Nasr M, Abou Chaaya A, Abboud N, Bechelany M, Viter R, Eid C, Khoury A, Miele P (2015) Photoluminescence: a very sensitive tool to detect the presence of anatase in rutile phase electrospun TiO2 nanofibers. Superlattices Microstruct 77:18–24. https://doi.org/10.1016/j.spmi.2014.10.034
Nawaz R, Kait CF, Chia HY, Isa MH, Huei LW (2019) Glycerol-mediated facile synthesis of colored titania nanoparticles for visible light photodegradation of phenolic compounds. Nanomaterials 9:1586. https://doi.org/10.3390/nano9111586
Nawaz R, Chong FK, Ho YC, Isa MH, Lim WH (2020a) Restoration of pretreated palm oil mill effluent using TiO2 based photocatalytic system: an optimization study. IOP Conf Ser Mater Sci Eng 736:042035
Nawaz R, Kait CF, Chia HY, Isa MH, Huei LW (2020b) Photocatalytic remediation of treated palm oil mill effluent contaminated with phenolic compounds using TiO2 nanomaterial. Desalination Water Treat 183:355–365. https://doi.org/10.5004/dwt.2020.25218
Nawaz R, Kait CF, Chia HY, Isa MH, Huei LW (2020c) Structural elucidation of core–shell TiO2 nanomaterials for environmental pollutants removal: a focused mini review. Environ Technol Innov 19:101007. https://doi.org/10.1016/j.eti.2020.101007
Negi C, Kandwal P, Rawat J, Sharma M, Sharma H, Dalapati G, Dwivedi C (2021) Carbon-doped titanium dioxide nanoparticles for visible light driven photocatalytic activity. Appl Surf Sci 554:149553. https://doi.org/10.1016/j.apsusc.2021.149553
Neoh CH, Lam CY, Ghani SM, Ware I, Sarip SH, Ibrahim Z (2016) Bioremediation of high-strength agricultural wastewater using Ochrobactrum sp. strain SZ1. Biotech 6:143. https://doi.org/10.1007/s13205-016-0455-1
Ni Y, Xia Z, Kokot S (2011) A kinetic spectrophotometric method for simultaneous determination of phenol and its three derivatives with the aid of artificial neural network. J Hazard Mater 192:722–729. https://doi.org/10.1016/j.jhazmat.2011.05.081
Pan X, Yang MQ, Fu X, Zhang N, Xu YJ (2013) Defective TiO2 with oxygen vacancies: synthesis, properties and photocatalytic applications. Nanoscale 5:3601–3614. https://doi.org/10.1039/C3NR00476G
Panomsuwan G, Watthanaphanit A, Ishizaki T, Saito N (2015) Water-plasma-assisted synthesis of black titania spheres with efficient visible-light photocatalytic activity. Phys Chem Chem Phys 17:13794–13799. https://doi.org/10.1039/C5CP00171D
Pelaez M, Nolan NT, Pillai SC, Seery MK, Falaras P, Kontos AG, Dunlop PS, Hamilton JW, Byrne JA, O’shea K (2012) A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl Catal B Environ 125:331–349. https://doi.org/10.1016/j.apcatb.2012.05.036
Rab N, Chong FK, Mohamed HI, Lim WH (2018) Preparation of TiO2 nanoparticles by hydrolysis of TiCl4 using water and glycerol solvent system. IOP Conf Ser J Phys 1123:012065
Rahman S, Nawaz R, Khan JA, Ullah H, Irfan M, Glowacz A, Lyp-Wronska K, Wzorek L, Asif Khan MK, Jalalah M, Alsaiari MA, Almawgani AH (2021) Synthesis and characterization of carbon and carbon-nitrogen doped black TiO2 nanomaterials and their application in sonophotocatalytic remediation of treated agro-industrial wastewater. Materials 14:6175. https://doi.org/10.3390/ma14206175
Ramos RL, Moreira VR, Lebron YAR, Santos AV, Santos LVS, Amaral MCS (2021) Phenolic compounds seasonal occurrence and risk assessment in surface and treated waters in Minas Gerais−Brazil. Environ Pollut 268:115782. https://doi.org/10.1016/j.envpol.2020.115782
Samal A, Das DP (2018) Transfiguring UV light active “metal oxides” to visible light active photocatayst by reduced graphene oxide hypostatization. Catal Today 300:124–135. https://doi.org/10.1016/j.cattod.2017.03.052
Schwarzenbach RP, Escher BI, Fenner K, Hofstetter TB, Johnson CA, Von Gunten U, Wehrli B (2006) The challenge of micropollutants in aquatic systems. Science 313:1072–1077. https://doi.org/10.1126/science.1127291
Surkatti R, Al-Zuhair S (2018) Microalgae cultivation for phenolic compounds removal. Environ Sci Pollut Res 25:33936–33956. https://doi.org/10.1007/s11356-018-3450-8
Tetteh EK, Rathilal S, Asante-Sackey D, Chollom MN (2021) Prospects of synthesized magnetic TiO2-based membranes for wastewater treatment: a review. Materials 14:3524. https://doi.org/10.3390/ma14133524
Tian M, Mahjouri-Samani M, Eres G, Sachan R, Yoon M, Chisholm MF, Wang K, Puretzky AA, Rouleau CM, Geohegan DB (2015) Structure and formation mechanism of black TiO2 nanoparticles. ACS Nano 9:10482–10488. https://doi.org/10.1021/acsnano.5b04712
Treschev SY, Chou PW, Tseng YH, Wang JB, Perevedentseva EV, Cheng CL (2008) Photoactivities of the visible-light-activated mixed-phase carbon-containing titanium dioxide: the effect of carbon incorporation. Appl Catal B Environ 79:8–16. https://doi.org/10.1016/j.apcatb.2007.09.046
Tufarelli V, Laudadio V, Casalino E (2016) An extra-virgin olive oil rich in polyphenolic compounds has antioxidant effects in meat-type broiler chickens. Environ Sci Pollut Res 23:6197–6204. https://doi.org/10.1007/s11356-015-5852-1
Tziotzios G, Teliou M, Kaltsouni V, Lyberatos G, Vayenas DV (2005) Biological phenol removal using suspended growth and packed bed reactors. Biochem Eng J 26:65–71. https://doi.org/10.1016/j.bej.2005.06.006
Villegas LGC, Mashhadi N, Chen M, Mukherjee D, Taylor KE, Biswas N (2016) A short review of techniques for phenol removal from wastewater. Curr Pollut Rep 2:157–167. https://doi.org/10.1007/s40726-016-0035-3
Wang H, Lin T, Zhu G, Yin H, Lü X, Li Y, Huang F (2015) Colored titania nanocrystals and excellent photocatalysis for water cleaning. Catal Commun 60:55–59. https://doi.org/10.1016/j.catcom.2014.11.004
Wang J, Wang Y, Wang W, Peng T, Liang J, Li P, Pan D, Fan Q, Wu W (2020) Visible light driven Ti3+ self-doped TiO2 for adsorption-photocatalysis of aqueous U(VI). Environ Pollut 262:114373. https://doi.org/10.1016/j.envpol.2020.114373
Wang Y, Wang H, Yang Y, Xin B (2021) Magnetic NiFe2O4 3D nanosphere photocatalyst: glycerol-assisted microwave solvothermal synthesis and photocatalytic activity under microwave electrodeless discharge lamp. Ceram Intern 47:14594–14602. https://doi.org/10.1016/j.ceramint.2021.02.041
Wu Y, Xing M, Zhang J (2011) Gel-hydrothermal synthesis of carbon and boron co-doped TiO2 and evaluating its photocatalytic activity. J Hazard Mater 192:368–373. https://doi.org/10.1016/j.jhazmat.2011.05.037
Xiang Q, Yu J, Wong PK (2011) Quantitative characterization of hydroxyl radicals produced by various photocatalysts. J Colloid Interface Sci 357:163–167. https://doi.org/10.1016/j.jcis.2011.01.093
Xiong W, Ni P, Chen Y, Gao YLS, Zhan A (2019) Biological consequences of environmental pollution in running water ecosystems: a case study in zooplankton. Environ Pollut 252:1483–1490. https://doi.org/10.1016/j.envpol.2019.06.055
Yao Y (2016) Enhancement of mass transfer by ultrasound: Application to adsorbent regeneration and food drying/dehydration. Ultrason Sonochem 31:512–531. https://doi.org/10.1016/j.ultsonch.2016.01.039
Yin H, Lin T, Yang C, Wang Z, Zhu G, Xu T, Xie X, Huang F, Jiang M (2013) Gray TiO2 nanowires synthesized by aluminum-mediated reduction and their excellent photocatalytic activity for water cleaning. Chem Eur J 19:13313–13316. https://doi.org/10.1002/chem.201302286
Yu J, Yu H, Cheng B, Zhao X, Zhang Q (2006) Preparation and photocatalytic activity of mesoporous anatase TiO2 nanofibers by a hydrothermal method. J Photochem Photobiol A Chem 182:121–127. https://doi.org/10.1016/j.jphotochem.2006.01.022
Zangeneh H, Zinatizadeh A, Habibi M, Akia M, Isa MH (2015) Photocatalytic oxidation of organic dyes and pollutants in wastewater using different modified titanium dioxides: a comparative review. J Ind Eng Chem 26:1–36. https://doi.org/10.1016/j.jiec.2014.10.043
Zhong J, Chen F, Zhang J (2010) Carbon-deposited TiO2: synthesis, characterization, and visible photocatalytic performance. J Phys Chem C 114:933–939. https://doi.org/10.1021/jp909835m
Zhou M, Zhang J, Sun C (2017) Occurrence, ecological and human health risks, and seasonal variations of phenolic compounds in surface water and sediment of a potential polluted river basin in China. Int J Environ Res Public Healt 14:1140. https://doi.org/10.3390/ijerph14101140
Acknowledgements
The authors sincerely appreciate funding from Researchers Supporting Project number (RSP-2021/399), King Saud University, Riyadh, Saudi Arabia.
Funding
This research was funded by Researchers Supporting Project of King Saud University, Riyadh, Saudi Arabia, grant number RSP-2021/399.
Author information
Authors and Affiliations
Contributions
RN: conceptualization, methodology, investigations, writing—original draft preparation, visualization, formal analysis, writing—review and editing; NTS: methodology, investigations, writing—review and editing; SH: validation, resources, writing—review and editing, project administration, funding acquisition; HU: conceptualization, methodology, data curation, writing—review and editing; MJ: formal analysis, visualization; MSA: data curation, validation, writing—review and editing, project administration, funding acquisition; SK: validation, writing—review and editing, resources. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nawaz, R., Sahrin, N.T., Haider, S. et al. Photocatalytic performance of black titanium dioxide for phenolic compounds removal from oil refinery wastewater: nanoparticles vs nanowires. Appl Nanosci 12, 3499–3515 (2022). https://doi.org/10.1007/s13204-021-02240-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-02240-5