Abstract
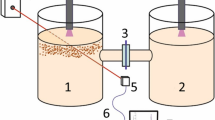
The influence of the initial pH of the solution on ferritization in the Fe2+–Co2+–SO42−–OH− system was investigated. Cobalt ferrite was obtained by a combined method of co-precipitation and treatment with contact low-temperature non-equilibrium plasma. The pH varied in the range of 7–12. The phase composition was determined by X-ray phase analysis. Magnetic properties by vibration magnetometry. The stability of the formed complexes was evaluated by the cyclic voltammograms method. Studies have shown that PNC treatment promotes ferritization. In this case, by varying the initial pH value, you can get products with different technological characteristics.
As the initial pH value increases, the oxidation rate decreases. According to X-ray examination, with increasing initial pH of the suspension, the degree of crystallinity of the final product increases, and the magnetic parameters (saturation magnetization, forced force) also increase rapidly. The formation of cobalt ferrite occurs at an initial pH 11–12







Similar content being viewed by others
References
Ai L et al (2010) Activated carbon/CoFe2O4 composites: facile synthesis, magnetic performance and their potential application for the removal of malachite green from water. Chem Eng J. 156(2):243–249
Ciomaga CE et al (2019) Functional properties of percolative CoFe2O4-PbTiO3 composite ceramics. J Alloys Compounds 775:90–99
Etier MF et al. (2012) Synthesis and magnetic properties of cobalt ferrite nanoparticles. MRS Online Proceedings Library Archive 1398
Frolova LA, Derhachov MP (2017) The effect of contact non-equilibrium plasma on structural and magnetic properties of MnXFe3− XO4 spinels. Nanoscale Res Lett 12(1):505
Frolova LA, Derimova AV (2019) Factors controlling magnetic properties of cofe2o4 nanoparticles prepared by contact low-temperature non-equilibrium plasma method. J Chem Technol Metallurgy 54(5):1040–1046
Frolova L, Derimova A, Galivets I, Savchenko M, Khlopytskyi A (2016) Investigation of phase formation in the system Fe2+/Co2+/O2/H2O. Eastern-Eur J Enterprise Technol 6(6):64–68
Gorski CA et al (2016) Thermodynamic characterization of iron oxide–aqueous Fe2+ redox couples. Environ Sci Technol 50(16):8538–8547
Idayanti N et al. (2020) Structural change and magnetic properties of mechanically alloyed spinel ferrite CoFe2O4. Key Eng Mater. Trans Tech Publications Ltd 855: 108–116
Kumbhar VS et al (2012) Chemical synthesis of spinel cobalt ferrite (CoFe2O4) nano-flakes for supercapacitor application. Appl Surface Sci 259:39–43
Munjal S et al (2019) Citric acid coated CoFe2O4 nanoparticles transformed through rapid mechanochemical ligand exchange for efficient magnetic hyperthermia applications. J Magn Magn Mater 477:388–395
Naik TRR et al (2019) Low-temperature microwave-assisted synthesis and antifungal activity of CoFe2O4 nanoparticles. J Mater NanoSci 6(2):67–72
Pourgolmohammad B, Masoudpanah SM, Aboutalebi MR (2017) Effect of starting solution acidity on the characteristics of CoFe2O4 powders prepared by solution combustion synthesis. J Magn Magn Mater. 424:352–358
Routray KL, Saha S, Behera D (2020) Insight into the anomalous electrical behavior, dielectric and magnetic study of Ag-Doped CoFe 2 O 4 synthesised by Okra extract-assisted green synthesis. J Electronic Mater 49(12):7244–7258
Senthil VP et al (2018) Study of structural and magnetic properties of cobalt ferrite (CoFe2O4) nanostructures. Chem Phys Lett 695:19–23
Venturini J et al (2019) The influence of solvent composition in the sol−gel synthesis of cobalt ferrite (CoFe2O4): a route to tuning its magnetic and mechanical properties. J Eur Ceramic Soc 39(12):3442–3449
Vignesh H et al. (2019) Structural and Magnetic properties of Cobalt Ferrite (CoFe2O4) Nanoparticles by Sol–Gel Technique using Yeast. IOP Conference Series: Materials Science and Engineering. IOP Publishing 577(1): 012092
Wiedmann MK et al. (2011) Atomic layer deposition for improved stability of catalysts for the conversion of biomass to chemicals and fuels. MRS Online Proceedings Library Archive 1366
Wu X et al (2016) PEG-assisted hydrothermal synthesis of CoFe2O4 nanoparticles with enhanced selective adsorption properties for different dyes. Appl Surface Sci 389:1003–1011
Zhang JH, Chen L, Williams XL (2012) Template assisted fabrication and magnetic properties of cobalt ferrite nanostructures. MRS Online Proceedings Library Archive 1408
Acknowledgements
We would like to acknowledge the support provided by the Ministry of Science and Education of Ukraine (project «Obtaining nanodispersed metal oxide materials and nanocomposites based on them for environmental protection»). The author expresses his sincere gratitude to T. Butyrina, Associate Professor of the Department of Technology of Inorganic Substances and Ecology, for her help in obtaining CVA metric curves.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Frolova, L., Sukhyy, K. Investigation of the ferritization process in the Co2+–Fe2+–SO42−–OH− system under the action of contact non-equilibrium low-temperature plasma. Appl Nanosci 12, 1029–1036 (2022). https://doi.org/10.1007/s13204-021-01755-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-01755-1