Abstract
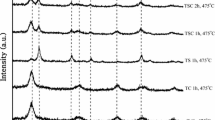
TiO2–ZnO nanocomposites with a constant Ti:Zn molar ratio of 1:0.1 were prepared via sol–gel process followed by calcination at 300, 400, 500, 600, and 700 °C. The structural and compositional characterizations of these nanocomposites were performed through XRD, FTIR, SEM, and EDAX. Bandgap was measured using DRS. Photocatalytic performance of the nanocomposites was evaluated by decolorization of methyl orange dye under UV and visible irradiation with and without aeration. The results showed that increase in calcination temperature resulted in nanocomposites with well-defined morphology. Although the particle size increased with increase in calcination temperature, the crystallinity of the particles also increased, resulting in enhanced photocatalytic activity. A temperature-dependent anatase-to-rutile phase transformation was observed in TiO2–ZnO nanocomposite beyond 600 °C. The calcination temperature influenced both dye adsorption on the nanocomposites and also dye decolorization by photocatalysis. Even when present at low molar concentration, ZnO in the nanocomposite caused sufficient decrease in bandgap (2.6 eV) at temperatures as low as 400 °C, such that visible irradiation could cause dye decolorization. However, the best decolorization performance was observed in the presence of the nanocomposite calcined at 600 °C. Aerated systems showed better performance in all cases. Desorption of the dye remaining adsorbed on the nanocomposite at the end of the photocatalytic reaction, confirmed that adsorption accounted for only 6.6 and 3% of dye removal in the nanocomposites calcined at 600 °C with UV and visible irradiation, respectively. However, in other systems, ignoring adsorption may cause significant overestimation in photocatalytic loss of dye from the system.









Similar content being viewed by others
References
Al-Qaradawi S, Salman SR (2002) Photocatalytic degradation of methyl orange as a model compound. J Photochem Photobiol A Chem 148(1–3):161–168
Araña J, Doña-Rodríguez JM, Portillo-Carrizo D, Fernández-Rodríguez C, Pérez-Peña J, Díaz OG, Navio JA, Macías M (2010) Photocatalytic degradation of phenolic compounds with new TiO2 catalysts. Appl Catal B 100(1):346–354. https://doi.org/10.1016/j.apcatb.2010.08.011
Athauda TJ, Neff JG, Sutherlin L, Butt U, Ozer RR (2012) Systematic study of the structure–property relationships of branched hierarchical TiO2/ZnO nanostructures. ACS Appl Mater Interfaces 4(12):6917–6926. https://doi.org/10.1021/am302061z
Bojinova A, Kralchevska R, Poulios I, Dushkin C (2007) Anatase/rutile TiO2 composites: influence of the mixing ratio on the photocatalytic degradation of malachite green and orange II in slurry. Mater Chem Phys 106(2):187–192. https://doi.org/10.1016/j.matchemphys.2007.05.035
Cai Y, Strømme M, Welch K, Cai YL (2014) Disinfection kinetics and contribution of reactive oxygen species when eliminating bacteria with TiO2 Induced photocatalysis. J Biomater Nanobiotechnol 5(5):200–209. https://doi.org/10.4236/jbnb.2014.53024
Cheng C, Amini A, Zhu C, Xu Z, Song H, Wang N (2014) Enhanced photocatalytic performance of TiO2–7ZnO hybrid nanostructures. Sci rep 4(4181):1–5. https://doi.org/10.1038/srep04181
Choudhury B, Choudhury A (2013) Local structure modification and phase transformation of TiO2 nanoparticles initiated by oxygen defects, grain size, and annealing temperature. Int Nano Lett 3(1):55. https://doi.org/10.1186/2228-5326-3-55
Çomaklı O, Yazıcı M, Yetim T, Yetim AF, Çelik A (2016) The effect of calcination temperatures on structural and electrochemical properties of TiO2 film deposited on commercial pure titanium. Surf Coat Technol 285:298–303. https://doi.org/10.1016/j.surfcoat.2015.11.055
Conesa JJC (2012). Modeling with hybrid density functional theory the electronic band alignment at the zinc oxide–anatase interface. J Phys Chem C, 116(35): 18884–18890. https://doi.org/10.1021/jp306160c
Das R, Sarkar S, Chakraborty S, Choi H, Bhattacharjee C (2014) Remediation of antiseptic components in wastewater by photocatalysis using TiO2 nanoparticles. Ind Eng Chem Res 53(8):3012–3020. https://doi.org/10.1021/ie403817z
De Angelis F, Di Valentin C, Fantacci S, Vittadini A, Selloni A (2014) Theoretical studies on anatase and less common TiO2 phases: bulk, surfaces, and nanomaterials. Chem Rev 114(19):9708–9753. https://doi.org/10.1021/cr500055q
Derikvandi H, Nezamzadeh-Ejhieh A (2017) Increased photocatalytic activity of NiO and ZnO in photodegradation of a model drug aqueous solution: effect of coupling, supporting, particles size and calcination temperature. J Hazard Mater 321:629–638. https://doi.org/10.1016/j.jhazmat.2016.09.056
Devi LG, Kavitha R (2013) A review on non metal ion doped titania for the photocatalytic degradation of organic pollutants under UV/solar light: role of photogenerated charge carrier dynamics in enhancing the activity. Appl Catal B 140:559–587. https://doi.org/10.1016/j.apcatb.2013.04.035
Fox MA, Dulay MT (1993) Heterogeneous photocatalysis. Chem rev 93(1):341–357
Gallardo JM, Vicente A, Escribano S, Buscab G (1995) Anatase crystal growth and phase transformation to Rutile in high- area TiO2, MOO,-TiO, and other TiO,-supported oxide catalytic systems. J Mater Chem, 5(8):1245–1249. http://pubs.rsc.org/en/content/articlepdf/1995/JM/JM9950501245
Gao J, Luan X, Wang J, Wang B, Li K, Li Y, Kang P, Han G (2011) Preparation of Er3+:YAlO3 /Fe-doped TiO2 –ZnO and its application in photocatalytic degradation of dyes under solar light irradiation. Desalination 268:68–75. https://doi.org/10.1016/j.desal.2010.09.052
Gribb AA, Banfield JF (1997) Particle size effects on transformation kinetics and phase stability in nanocrystalline TiO2. Am Mineral 82(7–8):717–728. https://doi.org/10.2138/am-1997-7-809
Janitabar-DarziAli S, Mahjoub R (2009) Investigation of phase transformations and photocatalytic properties of sol–gel prepared nanostructured ZnO/TiO2 composites. J Alloys Compd 486(1–2):805–808. https://doi.org/10.1016/J.JALLCOM.2009.07.071
Jing C, Rawson FJ, Zhou H, Shi X, Li WH, Li DW, Long YT (2014) New insights into electrocatalysis based on plasmon resonance for the real-time monitoring of catalytic events on single gold nanorods. Anal Chem 86(11):5513–5518. https://doi.org/10.1021/ac500785u
Lei JF, Li LB, Shen XH, Du K, Ni J, Liu CJ, Li WS (2013) Fabrication of ordered ZnO/TiO2 heterostructures via a templating technique. Langmuir 29(45):13975–13981
Li G, Li L, Boerio-Goates J, Woodfield BF (2005) High purity anatase TiO2 nanocrystals: near room-temperature synthesis, grain growth kinetics, and surface hydration chemistry. J Am Chem Soc 127(24):8659–8666. https://doi.org/10.1021/ja050517g
Liang R, Hu A, Li W, Zhou YN (2013). Enhanced degradation of persistent pharmaceuticals found in wastewater treatment effluents using TiO2 nanobelt photocatalysts. J Nanoparticle Res, 15(10):1990. https://doi.org/10.1007/s11051-013-1990-x
Liao DL, Badour CA, Liao BQ (2008) Preparation of nanosized TiO2/ZnO composite catalyst and its photocatalytic activity for degradation of methyl orange. J Photochem Photobiol A 194(1):11–19. https://doi.org/10.1016/j.jphotochem.2007.07.008
Little ME, Kordesch ME (2001) Band-gap engineering in sputter-deposited ScxGa1–xN. Appl Phys Lett 78(19):2891–2892. https://doi.org/10.1063/1.1370548
Luttrell T, Halpegamage S, Tao J, Kramer A, Sutter E, Batzill M (2014) Why is anatase a better photocatalyst than rutile? Model studies on epitaxial TiO2 films. Sci Rep 4:4043. https://doi.org/10.1038/srep04043
Méndez-Arriaga F, Esplugas S, Gime´nez J (2008) Photocatalytic degradation of non-steroidal anti-inflammatory drugs with TiO2 and simulated solar irradiation. Water Res 42:585–594. https://doi.org/10.1016/j.watres.2007.08.002
Minero C, Catozzo F, Pelizzett E (1992). Role of adsorption in photocatalyzed reactions of organic molecules in aqueous TiO2 suspensions. Langmuir 8:481–486. https://doi.org/10.1021/la00038a029
Mo SD, Ching WY (1995) Electronic and optical properties of three phases of titanium dioxide: rutile, anatase, and brookite. Phys Rev B 51(19):13023–13032. https://doi.org/10.1103/PhysRevB.51.13023
Mozaffari S, Li W, Thompson C, Ivanov S, Seifert S, Lee B, Koverick L, Karim AM (2017) Colloidal nanoparticle size control: experimental and kinetic modeling investigation of the ligand–metal binding role in controlling the nucleation and growth kinetics. Nanoscale 9(36):13772–13785
Nguyen NC, Lin BL, Chen SS (2013) A novel treatment method for acetaminophen in pharmaceutical wastewater by photocatalysis with various electron accepters. J Eng Technol Educ 9(5):8–14
Nishio J, Tokumura M, Znad HT, Kawase Y (2006) Photocatalytic decolorization of azo-dye with zinc oxide powder in an external UV light irradiation slurry photoreactor. J Hazard Mater 138(1):106–115. https://doi.org/10.1016/j.jhazmat.2006.05.039
Odling G, Robertson N (2015) Why is anatase a better photocatalyst than rutile? The importance of free hydroxyl radicals. Chem Sus Chem 8(11):1838–1840. https://doi.org/0.1002/cssc.201500298
Pei CC, Leung WWF (2013) Photocatalytic degradation of rhodamine B by TiO2/ZnO nanofibers under visible-light irradiation. Sep Purif Technol 114:108–116. https://doi.org/10.1016/j.seppur.2013.04.032
Pelaez M, Nolan NT, Pillai SC, Seery MK, Falaras P, Kontos AG, Dunlop PSM, Hamilton JWJ, Byrne JA, O’Shea K, Entezari MH (2012) A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl Catal B 125:331–349. https://doi.org/10.1016/j.apcatb.2012.05.036
Rajbongshi BM, Samdarshi SK, Boro B (2015) Multiphasic bi-component TiO2–ZnO nanocomposite: synthesis, characterization and investigation of photocatalytic activity under different wavelengths of light irradiation. J Mater Sci Mater Electron 26(1):377–384. https://doi.org/10.1007/s10854-014-2410-4
Rizzo L, Meric S, Guida M, Kassinos D, Belgiorno V (2009) Heterogenous photocatalytic degradation kinetics and detoxification of an urban wastewater treatment plant effluent contaminated with pharmaceuticals. Water Res 43:4070–4078. https://doi.org/10.1016/j.watres.2009.06.046
Rodríguez-Méndez A, Guzmán C, Elizalde-Peña EA, Escobar-Alarcón L, Vega M, Rivera JA, Esquivel K (2017) Effluent disinfection of real wastewater by Ag–TiO2 nanoparticles photocatalysis. J Nanosci Nanotechnol 17(1):711–719. https://doi.org/10.1166/jnn.2017.13066
Schneider J, Matsuoka M, Takeuchi M, Zhang J, Horiuchi Y, Anpo M, Bahnemann DW (2014) Understanding TiO2 photocatalysis: mechanisms and materials. Chem Rev 114:9919 – 9986. https://doi.org/10.1021/cr5001892
Shahram M, Aberoomand-Azar P, Raeis-Farshid S, Abedini-Khorrami S, Hadi Givianrad M (2016) The effect of different molar ratios of ZnO on characterization and photocatalytic activity of TiO2/ZnO nanocomposite. J Saudi Chem Soc 20(4):373–378. https://doi.org/10.1016/J.JSCS.2012.08.002
Spurr RA, Myers H (1957). Quantitative analysis of anatase-rutile mixtures with an X-ray diffractometer. Anal Chem, 29(5): 760–762. https://doi.org/10.1021/ac60125a006
Su W, Zhang J, Feng Z, Chen T, Ying P, Li C (2008) Surface phases of TiO2 nanoparticles studied by UV Raman spectroscopy and FT-IR spectroscopy. J Phys ChemC 112(20):7710–7716. https://doi.org/10.1021/jp7118422
Tsuang YH, Sun JS, Huang YC, Lu CH, Chang WHS, Wang CC (2008) Studies of photokilling of bacteria using titanium dioxide nanoparticles. Int J Artif Organs 32(2):167–174. https://doi.org/10.1111/j.1525-1594.2007.00530.x
Wang J, Mi W, Tian J, Dai J, Wang X, Liu X (2013) Effect of calcinations of TiO2/ZnO composite powder at high temperature on photodegradation of methyl orange. Compos Part B 45(1):758–767. https://doi.org/10.1016/j.compositesb.2012.09.053
Yakuphanoglu F, Ilican S, Caglar M, Caglar Y (2007) The determination of the optical band and optical constants of non-crystalline and crystalline ZnO thin films deposited by spray pyrolysis. J Optoelectron Adv Mater 9(7):2180–2185
Yu J, Wang B (2010). Effect of calcination temperature on morphology and photoelectrochemical properties of anodized titanium dioxide nanotube arrays. Appl Catal B, 94(3): 295–302. https://doi.org/10.1016/j.apcatb.2009.12.003
Zhang Q, Gao L, Guo J (2000) Effects of calcination on the photocatalytic properties of nanosized TiO2 powders prepared by TiCl4 hydrolysis. Appl Catal B 26:207–215. https://doi.org/10.1016/S0926-3373(00)00122-3
Zhang J, Zhou P, Liu J, Yu J (2014) New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2. Phys Chem 16(16):20382–20386. https://doi.org/10.1039/c4cp02201g
Acknowledgements
The authors would like to extend their gratitude to the Sophisticated Analytical Instrument Facility (SAIF), IIT Bombay for FEG–SEM, EDAX, ICP-AES, and FTIR analyses, the Department of Chemistry for DRS measurements and the Department of Metallurgy Engineering and Materials Science (MEMS) for XRD analysis. Partial funding for this work was provided by the Department of Science and Technology, New Delhi, India under the Water Technology Initiative (WTI) scheme (Project code: DST/TM/WTI/2K15/101(G)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Menon, N.G., Tatiparti, S.S.V. & Mukherji, S. Effect of calcination temperature on the microstructure and electronic properties of TiO2–ZnO nanocomposites and implications on photocatalytic activity. Appl Nanosci 8, 915–930 (2018). https://doi.org/10.1007/s13204-018-0783-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-018-0783-z