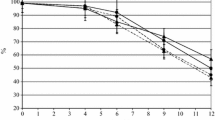
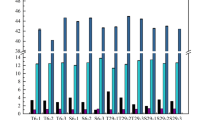
Abstract
The use of non-Saccharomyces yeast strains in winemaking is becoming a common trend. In fact, consumers are demanding new and healthier styles of wine. On the other hand, these strains are a challenge for the starting process due to winery-resident strains, especially with regard to industrial-scale fermentations. Current assay focuses on the scale-up of the laboratorial inoculum inside the winery environment to ferment 15,000 and 25,000 L of Vitis labrusca Bordô must, using a Hanseniaspora uvarum β-glucosidase-producer strain as starter culture. This scale-up could confirm the viability of using non-Saccharomyces yeast, as it presented promising results on a laboratory scale. The non-Saccharomyces strain was selected in a previous study since it proved to increase resveratrol concentration in lab scale winemaking. The yeast diversity was followed by the plate culturing method. Species identification and strain typing were determined by ITS-RFLP and PCR-fingerprinting, respectively. Physical and chemical analyses and resveratrol quantification were performed in the elaborated wines.


Similar content being viewed by others
Availability of data and material
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Agustini BC, da Silva GA, Bonfim TMB (2018) MALDI-TOF MS Supplementary database for species identification employing the yeast diversity encountered on southern Brazil grapes. Folia Microbiol (praha) 63(6):685–693. https://doi.org/10.1007/s12223-018-0607-2
Agustini BC, Silva LP, Bloch C et al (2014) Evaluation of MALDI-TOF mass spectrometry for identification of environmental yeasts and development of supplementary database. Appl Microbiol Biotechnol 98:5645–5654. https://doi.org/10.1007/s00253-014-5686-7
Aranda A, Matallana E, del Marcell lı´ O (2011) Saccharomyces Yeasts I: Primary Fermentation. Molecular Wine Microbiology, 1st edn. Elsevier, Spain, pp 1–31
Ashalou TJ (2019) A review on selection of fermentative microorganisms for functional foods and beverages: the production and future perspectives. Food Sci Technol 54:2511–2519. https://doi.org/10.1111/ijfs.14181
Ashalou TJ (2020) A holistic review on Euro-Asian lactic acid bacteria fermented cereals and vegetables. MDPI 8:1176. https://doi.org/10.3390/microorganisms8081176
Barreiro-Hurlé J, Colombo S, Cantos-villar E (2008) Is there a market for functional wines? Consumer preferences and willingness to pay for resveratrol-enriched red wine. Food Qual Prefer 19:360–371. https://doi.org/10.1016/j.foodqual.2007.11.004
Belda I, Conchillo LB, Ruiz J et al (2016) Selection and use of pectinolytic yeasts for improving clarification and phenolic extraction in winemaking. Int J Food Microbiol 223:1–8. https://doi.org/10.1016/j.ijfoodmicro.2016.02.003
Blanco P et al (2020) Saccharomyces cerevisiae strain diversity associated with spontaneous fermentations in organic wineries from Galicia (NW Spain). Fermentation 6:89. https://doi.org/10.3390/fermentation6030089
Bonciani T, De VL (2018) Qualitative and quantitative screening of the b-glucosidase activity in Saccharomyces cerevisiae and Saccharomyces uvarum strains isolated from refrigerated must. Lett Appl Microbiol 67(1):72–78. https://doi.org/10.1111/lam.12891
Brasil (2005) Instrução normativa nº 24, de 08 de setembro de 2005. Aprova o Manual Operacional de Bebidas e Vinagres do Ministério da Agricultura, Pecuária e Abastecimento. Caderno 04. Fermentados Alcoólicos. Diário Oficial da União. Brasília, DF, 20 set.2005
Brasil (2014) Decreto nº 8.198 de 20 de fevereiro de 2014. Regulamenta a Lei nº 7.678, de 8 de novembro de 1988, que dispõe sobre a produção, circulação e comercialização do vinho e derivados da uva e do vinho. Diário Oficial da União. Brasília, DF, 21 fev.2014
Brasil (2018) Instrução normativa nº 14 de 08 de fevereiro de 2018. Estabelece a complementação dos Padrões de Identidade e Qualidade do Vinho e Derivados da Uva e do Vinho. Diário Oficial da União. Brasília, DF, 09 mar. 2018
Chen AC, Shyu LY, Hsin YL et al (2017) Resveratrol relieves Angiostrongylus cantonensis—Induced meningoencephalitis by activating sirtuin-1. Acta Trop 173:76–84. https://doi.org/10.1016/j.actatropica.2017.05.023
Gaensly F, Agustini BC, da Silva GA et al (2015) Autochthonous yeasts with β-glucosidase activity increase resveratrol concentration during the alcoholic fermentation of Vitis labrusca grape must. J Funct Foods 19:288–295. https://doi.org/10.1016/j.jff.2015.09.041
International Organization of Vine and Wine (2011) Maximum acceptable limits of various substances contained in wine. Commendium of international methods of analysis.
Ling L, Gu S, Cheng Y (2017) Resveratrol activates endogenous cardiac stem cells and improves myocardial regeneration following acute myocardial infarction. Mol Med Rep 15:1188–1194. https://doi.org/10.3892/mmr.2017.6143
Lleixà J, Martín V, del Portillo MC et al (2016) Comparison of fermentation and wines produced by inoculation of Hanseniaspora vineae and Saccharomyces cerevisiae. Front Microbiol 7:1–12. https://doi.org/10.3389/fmicb.2016.00338
Lombardi SJ, Pannella G, Iorizzo M et al (2018) Sequential inoculum of Hanseniaspora guilliermondii and Saccharomyces cerevisiae for winemaking Campanino on an industrial scale. World J Microbiol Biotechnol 34:161. https://doi.org/10.1007/s11274-018-2540-6
Manzanares P, Vallés S, Viana F (2011) Non-Saccharomyces Yeasts in the Winemaking Process. Carrascosa A V, Muñoz R, Gonzales R Molecular Wine Microbiology, 1st edn. Elsevier, UK, pp 85–111
Martiniuk JT, Pacheco B, Russell G et al (2016) Impact of commercial strain use on Saccharomyces cerevisiae population structure and dynamics in pinot noir vineyards and spontaneous fermentations of a canadian Winery. PLoS ONE 11:1–19. https://doi.org/10.1371/journal.pone.0160259
Mateo JJ, Maicas S (2016) Application of Non-Saccharomyces Yeasts to Wine-Making Process. Fermentation 2:14. https://doi.org/10.3390/fermentation2030014
Padilla B, Zulian L, Ferreres À et al (2017) Sequential inoculation of native non-saccharomyces and saccharomyces cerevisiae strains for wine making. Front Microbiol 8:1–12. https://doi.org/10.3389/fmicb.2017.01293
Plessis HW, Toit M, Hoff JW et al (2017) Characterisation of non-saccharomyces yeasts using different methodologies and evaluation of their compatibility with malolactic fermentation. S Afr J Enol Vitic 38:46–63
Polizzotto G, Barone E, Ponticello G et al (2016) Isolation, identification and oenological characterization of non-Saccharomyces yeasts in a Mediterranean island. Lett Appl Microbiol 63(2):131–138. https://doi.org/10.1111/lam.12599
Pretorius IS (2000) Tailoring wine yeast for the new millennium: Novel approaches to the ancient art of winemaking. Yeast 16:675–729. https://doi.org/10.1002/1097-0061(20000615)16:8%3c675::AID-YEA585%3e3.0.CO;2-B
da Silva LF, Guerra CC, Klein D, Bergold AM (2017) Solid cation exchange phase to remove interfering anthocyanins in the analysis of other bioactive phenols in red wine. Food Chem 227:158–165. https://doi.org/10.1016/j.foodchem.2017.01.087
Silva GA, Bernardi TL, Schaker PDC et al (2012) Rapid yeast DNA extraction by boiling and freeze-thawing without using chemical reagents and dna purification. Brazilian Arch Biol Technol 55:319–327
Spagna G, Barbagallo RN, Palmeri R et al (2002) Properties of endogenous β-glucosidase of a Pichia anomala strain isolated from Sicilian musts and wines. Enzyme Microb Technol 31:1036–1041
Todaro A, Palmeri R, Barbagallo RN et al (2008) Food Chemistry Increase of trans-resveratrol in typical Sicilian wine using β-Glucosidase from various sources. Food Chem 107:1570–1575. https://doi.org/10.1016/j.foodchem.2007.09.075
Tristezza M, Tufariello M, Capozzi V et al (2016) The oenological potential of Hanseniaspora uvarum in simultaneous and sequential co-fermentation with Saccharomyces cerevisiae for industrial wine production. Front Microbiol 7:1–14. https://doi.org/10.3389/fmicb.2016.00670
Varela C, Barker A, Tran T et al (2017) Sensory profile and volatile aroma composition of reduced alcohol Merlot wines fermented with Metschnikowia pulcherrima and Saccharomyces uvarum. Int J Food Microbiol 252:1–9. https://doi.org/10.1016/j.ijfoodmicro.2017.04.002
Wang C, Liu Y (2013) Dynamic study of yeast species and Saccharomyces cerevisiae strains during the spontaneous fermentations of Muscat blanc in Jingyang, China. Food Microbiol 33:172–177. https://doi.org/10.1016/j.fm.2012.09.014
White TJ, Bruns TD, Lee E, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Acknowledgements
The authors would like to thank the technicians of the Embrapa Grape and Wine for their technical support in resveratrol and molecular analyses.
Funding
Current study was funded by the Brazilian Coordination for the Upgrading of Higher Education Personnel (CAPES).
Author information
Authors and Affiliations
Contributions
CS carried out the experiments and wrote the MS; TMBB was responsible for conceiving the idea, leader of the study; BCA and DB edited the manuscript and supervised the experiments; FD assisted in the execution of the experiments; MGAS edited the manuscript; GAS supervised the work and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rompkovksi, C., Agustini, B.C., Deffert, F. et al. Microbial dynamics in industrial-scale wine fermentation employing Hanseniaspora uvarum β-glucosidase-producer strain. J Food Sci Technol 59, 1570–1576 (2022). https://doi.org/10.1007/s13197-021-05168-4
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-021-05168-4