Abstract
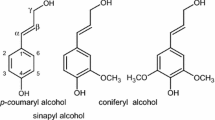
In order to understand the potential of some tropical hardwood wastes as source of utilizable lignins, the chemical structure of acetylated dioxane acid lignins from Testulea gabonensis Pellegr (T. gabonensis), Julbernardia pellegriniana (J. pellegriniana), Aucoumea klaineana Pierre (A. klaineana) and Tieghemella africana Pierre (T. africana) widely used in the tropical timber industry of Congo basin was investigated by 1D 1H and 13C-NMR. T. gabonensis and J. pellegriniana were the richest in guaiacyl (G) units, while A. klaineana and T. africana ones exhibited the highest syringyl (S) content. 1H-NMR exhibited an S/G < 1 ratio for T. gabonensis and J. pellegriniana, while that displayed by A. klaineana and T. africana dioxane acid lignins was S/G > 1. 13C-NMR aliphatic to phenolic hydroxyl group ratios were 1.42; 2.10; 2.0 and 1.41 for T. gabonensis, J. pellegriniana, A. klaineana and T. africana, respectively. The guaiacyl hydroxyl groups of T. gabonensis and J. pellegriniana dioxane acid lignins were more acetylated than A. klaineana and T. africana ones. The lowest acetylation of A. klaineana dioxane acid lignin was reinforced by the weak esterification of its guaiacyl-type hydroxyl groups at the Cα and Cγ position. The same trend was observed for acetylated syringyl-type hydroxyl groups at the Cα position of A. klaineana. Nevertheless, T. gabonensis was the richest in β-O-4 and 5-5′ condensed structures. Those lignins showed a low content on β-5 links for which the weakest signal strength was for T. africana which exhibited, however, the highest content on β–β structures of pinoresinol moieties. The occurrence of guaiacyl-type β-O-4 with Cα=O or β-O-4 threo and erythro forms within the studied hardwood lignins was discussed.







Similar content being viewed by others
References
Adler E (1977) Lignin chemistry—past, present and future. Wood Sci Technol 11:168–218. https://doi.org/10.1007/BF00365615
Alén R, Kuoppala E, Oesch P (1996) Formation of the main degradation compound groups from wood and its components during pyrolysis. J Anal Appl Pyrol 36(2):137–148. https://doi.org/10.1016/0165-2370(96)00932-1
Audu IG, Brosse N, Desharmais L, Rakshit SK (2012) Ethanol pretreatment of Typha capensis for bioethanol production and co-products. BioResources 7(4):5917–5933
Brosse N, Dufour A, Meng X, Sun Q, Ragauskas A (2012) Miscanthus: a fast-growing crop for biofuels and chemicals production. Biofuels Bioprod Bioref 1–19. https://bioenergycenter.org/besc/publications/brosse_miscanthus_yr5.pdf. Accessed 13 Dec 2018
Bylin S, Wells T, Sun Q, Ragauskas A, Theliander A (2017) Lignin structure and aggregation behavior in two component ionic liquid solvent system. BioResources 9(4):6002–6018
Capanema EA, Balakshin Y, Kadla FA (2004) Comprehensive approach for quantitative lignin characterization by NMR spectroscopy. J Agric Food Chem 52:1852–1860. https://pubs.acs.org/doi/abs/10.1021/jf035282b
Chen CL, Robert D (1988) Characterization of lignin by 1H and 13C NMR spectroscopy. Methods Enzymol 16:137–157. https://doi.org/10.1016/0076-6879(88)61017-2
Choi JW, Faix O (2011) NMR study on residual lignins isolated from chemical pulps of beech wood by enzymatic hydrolysis. J Ind Eng Chem 17:25–28. https://doi.org/10.1016/j.jiec.2010.10.004
El Hage R, Perrin D, Brosse N (2012) Effect of the pre-treatment severity on the antioxidant properties of ethanol organosolv Miscanthus × Giganteus lignin. Nat Resour 3:29–34. https://doi.org/10.4236/nr.2012.32005
Glasser WG, Sarkanen S (1989) Lignin, properties and materials, ACS symposium series. American Chemical Society, Washington, p 397
Goldmann WM, Ahola J, Mankinen O, Kantola AM, Komulainen S, Telkki VV, Tanskanen J (2017) Determination of phenolic hydroxyl groups in technical lignins by ionization difference ultraviolet spectrophotometry (Δε-IDUS method). Period Polytechnol Chem Eng 61(2):93–101. https://doi.org/10.3311/PPch.9269
Hu WJ, Harding SA, Popko JL, Ralph J, Stokke DD (1999) Repression of lignin biosynthesis promotes cellulose accumulation and growth in transgenic trees. Nat Biotechnol 17:808–8012. htp://lib.dr.iastate.edu/for_pubs/2
Kishimoto T, Ueki A, Sano Y (2003) Delignification mechanism during high-boiling solvent pulping: part 3. Structural changes in lignin analyzed by 13C-NMR spectroscopy. Holzforschung 57:602–610. https://doi.org/10.1515/HF.2003.091
Lewis NG, Yamamoto E (1990) Lignin: occurrence, biogenesis and biodegradation. Ann Rev Plant Physiol Plant Mol Biol 41:455–496. https://doi.org/10.1146/annurev.pp.41.060190.002323
Ludwig CH, Nist BJ, McCarthy JL (1964a) Lignin. XII. The high resolution nuclear magnetic resonance spectroscopy of protons in compounds related to lignin. J Am Chem Soc 86:1186–1196. https://pubs.acs.org/doi/abs/10.1021/ja01060a046
Ludwig CH, Nist BJ, McCarthery JL (1964b) Lignin III. The high resolution nuclear magnetic resonance spectroscopy of protons in acetylated lignins. J Am Chem Soc 86:1196–1202. https://doi.org/10.1021/ja01060a047
Lundquist K (1979) NMR studies of lignin. 3. 1H NMR spectroscopic data for lignin model compounds. Acta Chem Scand B 33:418–420. http://actachemscand.org/pdf/acta_vol_33b_p0418-0420.pdf. Accessed 13 Dec 2018
Lundquist K (1980) NMR study of lignin. 4. Investigation of spruce lignin by 1H NMR spectroscopy. Acta chem scandin B 34:21–26. http://actachemscand.org/pdf/acta_vol_34b_p0021-0026.pdf. Accessed 13 Dec 2018
Lundquist K, Stern K (1989) Analysis of lignin by 1H NMR spectroscopy. Nord Pulp Pap Res 3(4):2010–2013. https://doi.org/10.3183/npprj-1989-04-03-p210-213
Nimz HH, Ebel J, Grisebach H (1975) On the structure of lignin from soybean cell. Suspension cultures. Z Naturforsch 30:442–444
Nimz HH, Robert D, Faix O, Nemr N (1981) Carbone 13-NMR spectra of lignin, 8*. Holzforschung 35:16–21. https://doi.org/10.1515/hfsg.1981.35.1.16
Nze Nguema S (2009) Présentation du secteur forestier au Gabon: rapport sur l’évolution de la mise en œuvre de la politique du Gouvernement dans les secteurs Forêts, Pêches et Aquaculture, Aires protégées et Formation. http://www.euflegt.efi.int/documents/10180/23275/Pr%C3%A9sentation+du+Secteur+Forestier+au+Gabon/eb4427e5-f61c-4b09-83c3-6ab5bcfa14b4?version=1.0. Accessed 15 Dec 2018
Pandey KK (2008) Study of effect of photo-irradiation on surface chemistry of wood. Polym Degrad Stab 90:9–20. https://doi.org/10.1016/j.polymdegradstab.2005.02.009
Pinto PC, Evtuguin DV, Neto CP, Silvestre AJD, Amado FML (2002) Behavior of Eucalyptus globulus lignin during kraft pulping. II. Analysis by NMR, ESI/MS, and GPC. J Wood Chem Technol 22(2–3):109–125. https://doi.org/10.1081/WCT-120013356
Pu Y, Hu F, Huang F, Davison BH, Ragauskas AJ (2013) Assessing the molecular structure basis for biomass recalcitrance during dilute acid and hydrothermal pretreatments. Biotechnol Biofuels 6:15. https://doi.org/10.1186/1754-6834-6-15
Ralph J (1996) An unusual lignin from kenaf. J Nat Prod 59(4):1–2. https://doi.org/10.1021/np960143s
Ralph J, Landucci LL (2010) NMR of lignins. Lignin and lignans: advances in chemistry. Lignin and lignans: advances in chemistry/editors, Cyril Heitner, Don Dimmel, John A. Schmidt, New York, p 174. https://www.fpl.fs.fed.us/documnts/pdf2010/fpl_2010_ralph001.pdf. Accessed 13 Dec 2018
Saberikhah E, Mohammadi-Rovshandeh J, Mamaghani M (2013) Spectroscopic comparison of organosolv lignins isolated from wheat straw. Cellul Chem Technol 47(5–6):409–418. http://www.cellulosechemtechnol.ro/pdf/CCT5-6(2013)/p.409-418.pdf. Accessed 13 Dec 2018
Safou Tchiama R, Soulounganga P, Engozogho Anris PS, Bikoro Bi Athomo A, Andzi Barhé T, De Jeso B, Charrier B, Akagah AG (2018) Understanding the natural durability of some African tropical heartwoods toward Pycnoporus sanguineus and Antrodia sp.: lignin structure and cellulose morphology control. J Ind Acad Wood Sci 15(2):162–171. https://doi.org/10.1007/s13196-018-0222-5
Safou-Tchiama R, Ngwa Obame S, Brosse N, Soulounganga S, Andzi Barhé T (2016) Investigating the potential of Aucoumea klaineana Pierre sapwood and heartwood wastes to produce cellulosic ethanol. Afr J Biotechnol 15(46):2587–2595. https://doi.org/10.5897/AJB2016.15515
Safou-Tchiama R, Andzi Barhé T, Soulounganga P, Ngwa Obame S, Mabicka Iwangou SB, Athomo ABB, Engozogho Anris PS, De Jeso B, Akagah AG (2017a) Chemical reactivity and supramolecular susceptibility of hardwood celluloses towards succinic anhydride. Int J Biol Chem Sci 11(6):3110–3131. https://doi.org/10.4314/ijbcs.v11i6.44
Safou-Tchiama R, Andzi Barhé T, Soulounganga P, Akagah AG, De Jeso B (2017b) A comparative study of the syringyl, guaiacyl and hydroxyl groups units distribution in some African tropical hardwoods’ lignin by Py-GC/MS and spectroscopic techniques. J Mater Environ Sci 8(17):2530–2540. http://www.jmaterenvironsci.com/Document/vol8/vol8_N7/272-JMES-2734-Tchiama.pdf. Accessed 13 Dec 2018
Sette M, Lange H, Crestini C (2013) Quantitative HSQC analyses of lignin: a practical comparison. Comput Struct Biotechnol J 6(7):1–7. https://doi.org/10.5936/csbj.201303016
Silva EJ, Rocha e Silva MNP, Rufino RQ, Luna JM, Silva O, Sarubbo A (2014) Characterization of a biosurfactant produced by Pseudomonas cepacia CCT6659 in the presence of industrial wastes and its application in the biodegradation of hydrophobic compounds in soil. Colloids Surf B Biointerfaces 117:36–41. https://doi.org/10.1016/j.colsurfb.2014.02.012
Wen JL, Sun SL, Xue BL, Sun RC (2013) Recent advances in characterization of lignin polymer by solution-state nuclear magnetic resonance (NMR) methodology. Materials 6:359–391. https://doi.org/10.3390/ma6010359
Yoan AO, Xue Y, Kiki MJM (2018) Gabon wood industry and Chinese companies activities. OALib 05:1–15. https://file.scirp.org/pdf/OALibJ_2018053014425381.pdf. Accessed 17 Dec 2018
Zhi-Hua J (1997) Advances and applications of quantitative 31P-NMR for the structural elucidation of lignin. PhD Thesis. McGill University, Montréal, Quebec, Canada. http://digitool.Library.McGill.CA:80/R/-?func=dbin-jump-full&object_id=34650&silo_library=GEN01. Accessed 13 Dec 2018
Zhou XF, Lu XJ (2014) Structural characterization of kraft lignin for its green utilization. Wood Res 59(4):583–592. http://www.centrumdp.sk/wr/201404/06.pdf. Accessed 13 Dec 2018
Acknowledgements
We would like to thank the “Programme de Mobilité Scientifique et Universitaire” of the “Agence Universitaire de la Francophonie (AUF)” for its financial support. The Laboratoire de Chimie des Polymères Organiques (LCPO) and the Université de Bordeaux are thanked for the scientific support they brought to this research program.
Author information
Authors and Affiliations
Contributions
Rodrigue SAFOU-TCHIAMA was the principal investigator of the subject. He acted as group leader of a research program dealing with the natural durability and molecular structure of lignocellulosic polymers of tropical hardwoods in the LASNSOM (USTM) and the LaReVa (ENSET). Arsène BIKORO BI ATHOMO and Péguy Starlin ENGOZOGHO acted as PhD student in Wood Aristide Gervais AKAGAH, and Bernard De JESO acted as scientific directors.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Safou Tchiama, R., Arsène, B.B.A., Anris, P.S.E. et al. Characterization of some African tropical heartwood lignins by 1D 13C and 1H-NMR: molecular structure and hydroxyl groups’ distribution. J Indian Acad Wood Sci 16, 73–86 (2019). https://doi.org/10.1007/s13196-019-00239-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13196-019-00239-8