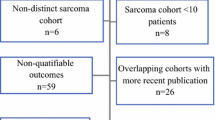
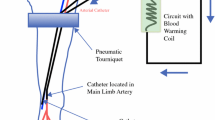
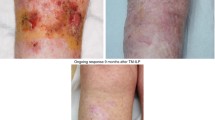
Abstract
Isolated limb perfusion (ILP) is a regional chemotherapy technique used in the treatment of locally advanced or unresectable extremity soft tissue sarcoma (ESTS) or malignant melanoma (MM) of the limbs. It allows for high concentrations of chemotherapeutic agents to be perfused in the limb while minimising the risk of systemic side-effects. While the technique has been utilized for decades, the role of ILP has evolved as other treatment strategies have become available. Current indications for ILP in sarcoma include induction in unresectable ESTS to allow for future definitive limb preservation procedures as well as definitive treatment of unresectable, multifocal ESTS. In MM, ILP is typically used in unresectable in-transit melanoma, and rarely as an alternative to amputation in bulky, symptomatic extremity disease. This review seeks to summarise the current evidence base and indications for ILP as well as present some technical insights from a high-volume United Kingdom (UK) unit.





Similar content being viewed by others
Data Availability
Data is available by request to corresponding author.
References
Jakob J et al (2018) Regional chemotherapy by isolated limb perfusion prior to surgery compared with surgery and post-operative radiotherapy for primary, locally advanced extremity sarcoma: a comparison of matched cohorts. Clin Sarcoma Res 8:1–9
Sevilla-Ortega L et al (2021) Role of isolated limb perfusion in the era of targeted therapies and immunotherapy in melanoma. A systematic review of the literature. Cancers (Basel) 13:5485
Bhangu A, Broom L, Nepogodiev D, Gourevitch D, Desai A (2013) Outcomes of isolated limb perfusion in the treatment of extremity soft tissue sarcoma: A systematic review. Eur J Surg Oncol 39:311–319
Davies EJ, Reijers SJM, Van Akkooi ACJ, Van Houdt WJ, Hayes AJ (2022) Isolated limb perfusion for locally advanced melanoma in the immunotherapy era. Eur J Surg Oncol 48:1288–1292
Klopp CT, Alford TC, Bateman J, Berry GN, Winship T (1950) Fractionated intra-arterial cancer; chemotherapy with methyl bis amine hydrochloride; a preliminary report. Ann Surg 132:811–832
Creech O Jr, Ryan RF, Krementz ET (1959) Treatment of melanoma by isolation-perfusion technique. J Am Med Assoc 169:339–343
Cavaliere R et al (1967) Selective heat sensitivity of cancer cells. Biochem Clin Stud Cancer 20:1351–1381
Grünhagen DJ, de Wilt JHW, ten Hagen TLM, Eggermont AMM (2006) Technology Insight: utility of TNF-α-based isolated limb perfusion to avoid amputation of irresectable tumors of the extremities. Nat Clin Pract Oncol 3:94–103
Moreno-Ramirez D, Cruz-Merino L, Ferrandiz L, Villegas-Portero R, Nieto-Garcia A (2010) Isolated Limb Perfusion for Malignant Melanoma: Systematic Review on Effectiveness and Safety. Oncologist 15:416–427
Vrouenraets BC, Nieweg OE, Kroon BBR (1996) Thirty-five years of isolated limb perfusion for melanoma: Indications and results. Br J Surg 83:1319–1328
Lienard D, Ewalenko P, Delmotte JJ, Renard N, Lejeune FJ (1992) High-dose recombinant tumor necrosis factor alpha in combination with interferon gamma and melphalan in isolation perfusion of the limbs for melanoma and sarcoma. J Clin Oncol Off J Am Soc Clin Oncol 10:52–60
Lejeune FJ (2002) Clinical use of TNF revisited: improving penetration of anti-cancer agents by increasing vascular permeability. J Clin Invest 110:433–435
Van Der Veen AH et al (2000) TNF-α augments intratumoural concentrations of doxorubicin in TNF-α-based isolated limb perfusion in rat sarcoma models and enhances anti-tumour effects. Br J Cancer 82:973–980
Eggermont AMM et al (1996) Isolated limb perfusion with tumor necrosis factor and melphalan for limb salvage in 186 patients with locally advanced soft tissue extremity sarcomas: The cumulative multicenter European experience. Ann Surg 224:756–765
Bonvalot S et al (2009) Hyperthermic isolated limb perfusion in locally advanced soft tissue sarcoma and progressive desmoid-type fibromatosis with TNF 1 mg and melphalan (T1-M HILP) is safe and efficient. Ann Surg Oncol 16:3350–3357
Hayes AJ, Neuhaus SJ, Clark MA, Thomas JM (2007) Isolated limb perfusion with melphalan and tumor necrosis factor α for advanced melanoma and soft-tissue sarcoma. Ann Surg Oncol 14:230–238
Bonvalot S et al (2005) Limb salvage with isolated perfusion for soft tissue sarcoma: Could less TNF-α be better? Ann Oncol 16:1061–1068
Podleska LE et al (2014) Drug dosage in isolated limb perfusion: Evaluation of a limb volume model for extremity volume calculation. World J Surg Oncol 12:1–9
Deroose JP et al (2011) Long-term results of tumor necrosis factor α- and melphalan-based isolated limb perfusion in locally advanced extremity soft tissue sarcomas. J Clin Oncol 29:4036–4044
Van Ginkel RJ et al (2007) Isolated limb perfusion with tumor necrosis factor alpha and melphalan for locally advanced soft tissue sarcoma: Three time periods at risk for amputation. Ann Surg Oncol 14:1499–1506
Erdmann F et al (2013) International trends in the incidence of malignant melanoma 1953–2008-are recent generations at higher or lower risk? Int J Cancer 132:385–400
Gershenwald JE, Scolyer RA (2018) Melanoma staging: American joint committee on cancer (AJCC) 8th edition and beyond. Ann Surg Oncol 25:2105–2110
Wolchok JD et al (2022) Long-term outcomes with nivolumab plus ipilimumab or nivolumab alone versus ipilimumab in patients with advanced melanoma. J Clin Oncol 40:127–137
Lebbé C et al (2019) Evaluation of two dosing regimens for nivolumab in combination with ipilimumab in patients with advanced melanoma: Results from the phase IIIb/IV CheckMate 511 trial. J Clin Oncol 37:867–875
Ascierto PA et al (2013) Phase II trial (BREAK-2) of the BRAF inhibitor dabrafenib (GSK2118436) in patients with metastatic melanoma. J Clin Oncol 31:3205–3211
Pasquali S, Hadjinicolaou AV, Chiarion Sileni V, Rossi CR, Mocellin S (2018) Systemic treatments for metastatic cutaneous melanoma. Cochrane Database Syst Rev 2018
Hayes AJ, Clark MA, Harries M, Thomas JM (2004) Management of in-transit metastases from cutaneous malignant melanoma. Br J Surg 91:673–682
Read RL et al (2015) In-transit Melanoma Metastases: Incidence, Prognosis, and the Role of Lymphadenectomy. Ann Surg Oncol 22:475–481
Andtbacka RHI et al (2019) Final analyses of OPTiM: A randomized phase III trial of talimogene laherparepvec versus granulocyte-macrophage colony-stimulating factor in unresectable stage III-IV melanoma. J Immunother Cancer 7:1–11
Nan Tie E, Henderson MA, Gyorki DE (2019) Management of in-transit melanoma metastases: a review. ANZ J Surg 89:647–652
Patel A, Carr MJ, Sun J, Zager JS (2022) In-transit metastatic cutaneous melanoma: current management and future directions. Clin Exp Metastasis 39:201–211
Eggermont AMM et al (2016) Prolonged Survival in Stage III Melanoma with Ipilimumab Adjuvant Therapy. N Engl J Med 375:1845–1855
Schachter J et al (2017) Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet 390:1853–1862
Weber J et al (2017) Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. N Engl J Med 377:1824–1835
Long GV et al (2017) Adjuvant Dabrafenib plus Trametinib in Stage III BRAF -Mutated Melanoma. N Engl J Med 377:1813–1823
Deroose JP et al (2012) 20 years experience of TNF-based isolated limb perfusion for in-transit melanoma metastases: TNF dose matters. Ann Surg Oncol 19:627–635
Koops HS et al (1998) Prophylactic isolated limb perfusion for localized, high-risk limb melanoma: results of a multicenter randomized phase III trial. European Organization for Research and Treatment of Cancer Malignant Melanoma Cooperative Group Protocol 18832, the World He. J Clin Oncol Off J Am Soc Clin Oncol 16:2906–2912
Cornett WR et al (2006) Randomized multicenter trial of hyperthermic isolated limb perfusion with melphalan alone compared with melphalan plus tumor necrosis factor: American College of Surgeons Oncology Group trial Z0020. J Clin Oncol 24:4196–4201
Lejeune FJ, Eggermont AMM (2007) Hyperthermic isolated limb perfusion with tumor necrosis factor is a useful therapy for advanced melanoma of the limbs [7]. J Clin Oncol 25:1449–1450
Thompson JF, Kam PC, Waugh RC, Harman CR (1998) Isolated limb infusion with cytotoxic agents: a simple alternative to isolated limb perfusion. Semin Surg Oncol 14:238–247
Beasley GM et al (2008) Isolated limb infusion for in-transit malignant melanoma of the extremity: A well-tolerated but less effective alternative to hyperthermic isolated limb perfusion. Ann Surg Oncol 15:2195–2205
Dossett LA, Ben-Shabat I, OlofssonBagge R, Zager JS (2016) Clinical response and regional toxicity following isolated limb infusion compared with isolated limb perfusion for in-transit melanoma. Ann Surg Oncol 23:2330–2335
Dangoor A et al (2016) UK guidelines for the management of soft tissue sarcomas. Clin Sarcoma Res 6:1–26
Grimer R, Judson I, Peake D, Seddon B (2010) Guidelines for the management of soft tissue sarcomas. Sarcoma 2010
Coindre JM et al (2001) Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas: a study of 1240 patients from the French Federation of Cancer Centers Sarcoma Group. Cancer 91:1914–1926
Wray CJ et al (2011) Isolated limb perfusion for unresectable extremity sarcoma. Cancer 117:3235–3241
Kaushal A, Citrin D (2008) The Role of Radiation Therapy in the Management of Sarcomas. Surg Clin North Am 88:629–646
Woll PJ et al (2012) Adjuvant chemotherapy with doxorubicin, ifosfamide, and lenograstim for resected soft-tissue sarcoma (EORTC 62931): A multicentre randomised controlled trial. Lancet Oncol 13:1045–1054
Gronchi A et al (2017) Histotype-tailored neoadjuvant chemotherapy versus standard chemotherapy in patients with high-risk soft-tissue sarcomas (ISG-STS 1001): an international, open-label, randomised, controlled, phase 3, multicentre trial. Lancet Oncol 18:812–822
Pasquali S et al (2022) Neoadjuvant chemotherapy in high-risk soft tissue sarcomas: A Sarculator-based risk stratification analysis of the ISG-STS 1001 randomized trial. Cancer 128:85–93
Squires MH et al (2022) Extremity soft tissue sarcoma: a multi-institutional validation of prognostic nomograms. Ann Surg Oncol 29:3291–3301
O’Donnell PW et al (2014) The effect of the setting of a positive surgical margin in soft tissue sarcoma. Cancer 120:2866–2875
O’Sullivan B et al (2002) Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet (London, England) 359:2235–2241
Davis AM et al (2005) Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol J Eur Soc Ther Radiol Oncol 75:48–53
Chung PWM et al (2009) Radiosensitivity translates into excellent local control in extremity myxoid liposarcoma: A comparison with other soft tissue sarcomas. Cancer 115:3254–3261
Hoekstra HJ, SchraffordtKoops H, Molenaar WM, Oldhoff J (1987) Results of isolated regional perfusion in the treatment of malignant soft tissue tumors of the extremities. Cancer 60:1703–1707
Stehlin JSJ, de Ipolyi PD, Giovanella BC, Gutierrez AE, Anderson RF (1975) Soft tissue sarcomas of the extremity. Multidisciplinary therapy employing hyperthermic perfusion. Am J Surg 130:643–646
Grunhagen DJ et al (2006) Outcome and prognostic factor analysis of 217 consecutive isolated limb perfusions with tumor necrosis factor-α and melphalan for limb-threatening soft tissue sarcoma. Cancer 106:1776–1784
Gutman M et al (1997) High dose tumor necrosis factor-alpha and melphalan administered via isolated limb perfusion for advanced limb soft tissue sarcoma results in a >90% response rate and limb preservation. Cancer 79:1129–1137
Lejeune FJ et al (2000) Limb salvage by neoadjuvant isolated perfusion with TNFalpha and melphalan for non-resectable soft tissue sarcoma of the extremities. Eur J Surg Oncol J Eur Soc Surg Oncol Br Assoc Surg Oncol 26:669–678
Noorda EM et al (2003) Isolated limb perfusion with tumor necrosis factor-α and melphalan for patients with unresectable soft tissue sarcoma of the extremities. Cancer 98:1483–1490
Neuwirth MG et al (2017) Isolated limb perfusion and infusion for extremity soft tissue sarcoma: a contemporary systematic review and meta-analysis. Ann Surg Oncol 24:3803–3810
Lans TE, Grünhagen DJ, De Wilt JHW, Van Geel AN, Eggermont AMM (2005) Isolated limb perfusions with tumor necrosis factor and melphalan for locally recurrent soft tissue sarcoma in previously irradiated limbs. Ann Surg Oncol 12:406–411
In’t Veld EH et al (2017) Isolated limb perfusion for locally advanced angiosarcoma in extremities: A multi-centre study. Eur J Cancer 85:114–121
Hoven-Gondrie ML et al (2008) Isolated limb perfusion and external beam radiotherapy for soft tissue sarcomas of the extremity: Long-term effects on normal tissue according to the LENT-SOMA scoring system. Ann Surg Oncol 15:1502–1510
Pennacchioli E et al (2007) Advanced extremity soft tissue sarcoma: prognostic effect of isolated limb perfusion in a series of 88 patients treated at a single institution. Ann Surg Oncol 14:553–559
Rastrelli M et al (2016) Hyperthermic isolated limb perfusion in locally advanced limb soft tissue sarcoma: A 24-year single-centre experience. Int J Hyperth Off J Eur Soc Hyperthermic Oncol North Am Hyperth Gr 32:165–172
Hill S et al (1993) Low-dose tumour necrosis factor alpha and melphalan in hyperthermic isolated limb perfusion. Br J Surg 80:995–997
Vaglini M et al (1994) Treatment of primary or relapsing limb cancer by isolation perfusion with high-dose alpha-tumor necrosis factor, gamma-interferon, and melphalan. Cancer 73:483–492
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Russell, M., Wilkinson, M. & Hayes, A. Isolated Limb Perfusion for Extremity Soft Tissue Sarcoma and Malignant Melanoma. Indian J Surg Oncol (2024). https://doi.org/10.1007/s13193-024-01920-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13193-024-01920-2