Abstract
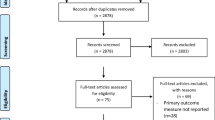
Patients with breast cancer are increasingly being offered breast conserving surgery (BCS) following neoadjuvant chemotherapy (NACT). We aimed to conduct a systematic review to assess the advantage of tumor marking in patients undergoing BCS after NACT. After registering the protocol for a systematic review with PROSPERO, a systematic search was conducted through September 30, 2020, for all studies involving patients undergoing BCS post NACT after tumor marking. Margin status on final histology was the primary outcome. Oxford Centre for Evidence Based Medicine (OCEBM) levels were used to assess internal validity. A total of 636 records from Medline/PubMed, 1381 from Embase, and 1422 from Cochrane library were extracted. After screening, 15 articles (1520 patients) were included for data synthesis. For marking, 6 studies used metallic markers and 5 used 125I-radioactive seeds (RSL) followed by skin tattoo and radio-guided occult lesion localization using 99mTc (ROLL) in one study each. Most studies used a single marker at the center except for two (143 patients), who practiced the bracketing technique. Incidence of unsatisfactory margins (positive/close) ranged from 5 to 23.5%. After excluding patients with complete pathological response, the “adjusted unsatisfactory margin” rate was found to be 19.3% (10.4–33%). Overall 20.6 recurrences (locoregional/distant) were reported per 1000 patient-years follow-up. Overall survival (OS) was only reported by one study as 96.6% and 84.7% in patients with and without marker placement (p = .01). Re-excision and secondary mastectomy rates (reported by nine studies) were 7.3% and 5.7% respectively. There is limited evidence that tumor marking before neoadjuvant chemotherapy improves the rate of unsatisfactory margins or survival outcomes in a patient undergoing BCS after NACT.

Similar content being viewed by others
References
Mauri D, Pavlidis N, Ioannidis JP (2005) Neoadjuvant versus adjuvant systemic treatment in breast cancer: a meta-analysis. J Natl Cancer Inst 97(3):188–94. https://doi.org/10.1093/jnci/dji021 (PMID: 15687361)
Butler-Henderson K, Lee AH, Price RI, Waring K (2014) Intraoperative assessment of margins in breast conserving therapy: a systematic review. Breast 23(2):112–9. https://doi.org/10.1016/j.breast.2014.01.002
McCahill LE, Single RM, Aiello Bowles EJ, Feigelson HS, James TA, Barney T et al (2012) Variability in reexcision following breast conservation surgery. JAMA 307(5):467–75. https://doi.org/10.1001/jama.2012.43
Heelan Gladden AA, Sams S, Gleisner A, Finlayson C, Kounalakis N, Hosokawa P et al (2017) Re-excision rates after breast conserving surgery following the 2014 SSO-ASTRO guidelines. Am J Surg 214(6):1104–1109. https://doi.org/10.1016/j.amjsurg.2017.08.023
Hill-Kayser CE, Vachani C, Hampshire MK, Di Lullo GA, Metz JM (2012) Cosmetic outcomes and complications reported by patients having undergone breast-conserving treatment. Int J Radiat Oncol Biol Phys 83(3):839–44. https://doi.org/10.1016/j.ijrobp.2011.08.013
Volders JH, Haloua MH, Krekel NM, Negenborn VL, Barbé E, Sietses C, “the nationwide network and registry of histo- and cytopathology in the Netherlands (PALGA)” et al (2016) Neoadjuvant chemotherapy in breast-conserving surgery - Consequences on margin status and excision volumes: a nationwide pathology study. Eur J Surg Oncol 42(7):986–93. https://doi.org/10.1016/j.ejso.2016.02.252
Lannin DR, Grube B, Black DS, Ponn T (2007) Breast tattoos for planning surgery following neoadjuvant chemotherapy. Am J Surg 194(4):518–20. https://doi.org/10.1016/j.amjsurg.2007.07.009
Sever AR, O’Brien ME, Humphreys S, Singh I, Jones SE, Jones PA (2005) Radiopaque coil insertion into breast cancers prior to neoadjuvant chemotherapy. Breast 14(2):108–17. https://doi.org/10.1016/j.breast.2004.08.008
Janssen NN, Nijkamp J, Alderliesten T, Loo CE, Rutgers EJ, Sonke JJ et al (2016) Radioactive seed localization in breast cancer treatment. Br J Surg 103(1):70–80. https://doi.org/10.1002/bjs.9962
Donker M, Straver ME, Rutgers EJ, Valdés Olmos RA, Loo CE, Sonke GS et al (2012) Radioguided occult lesion localisation (ROLL) in breast-conserving surgery after neoadjuvant chemotherapy. Eur J Surg Oncol 38(12):1218–1224. https://doi.org/10.1016/j.ejso.2012.07.107
Aggarwal V, Agarwal G, Lal P, Krishnani N, Mishra A, Verma AK et al (2008) Feasibility study of safe breast conservation in large and locally advanced cancers with use of radiopaque markers to mark pre-neoadjuvant chemotherapy tumor margins. World J Surg 32(12):2562–2569. https://doi.org/10.1007/s00268-007-9289-7
Rubio IT, Esgueva-Colmenarejo A, Espinosa-Bravo M, Salazar JP, Miranda I, Peg V (2016) Intraoperative ultrasound-guided lumpectomy versus mammographic wire localization for breast cancer patients after neoadjuvant treatment. Ann Surg Oncol 23(1):38–43. https://doi.org/10.1245/s10434-015-4935-z
Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535. https://doi.org/10.1136/bmj.b2535
Volders JH, Negenborn VL, Spronk PE, Krekel NMA, Schoonmade LJ, Meijer S et al (2018) Breast-conserving surgery following neoadjuvant therapy-a systematic review on surgical outcomes. Breast Cancer Res Treat 168(1):1–12. https://doi.org/10.1007/s10549-017-4598-5
Oh JL, Nguyen G, Whitman GJ, Hunt KK, Yu TK, Woodward WA et al (2007) Placement of radiopaque clips for tumor localization in patients undergoing neoadjuvant chemotherapy and breast conservation therapy. Cancer 110(11):2420–2427. https://doi.org/10.1002/cncr.23068.PMID:17941034;PMCID:PMC4329780
Cha C, Lee J, Kim D, Park S, Bae SJ, Eun NL et al (2020) Comparison of resection margin status after single or double radiopaque marker insertion for tumor localization in breast cancer patients receiving neoadjuvant chemotherapy. Breast Cancer Res Treat 184(3):797–803. https://doi.org/10.1007/s10549-020-05907-9
Alonso-Bartolome P, Ortega Garcia E, Garijo Ayensa F, de Juan FA, Vega BA (2002) Utility of the tumor bed marker in patients with breast cancer receiving induction chemotherapy. Acta Radiol 43(1):29–33
Ramos M, Díez JC, Ramos T, Ruano R, Sancho M, González-Orús JM (2014) Intraoperative ultrasound in conservative surgery for non-palpable breast cancer after neoadjuvant chemotherapy. Int J Surg 12(6):572–577. https://doi.org/10.1016/j.ijsu.2014.04.003
Shalaby LASED, KhallafESed, Moussa MM, (2019) Clip and wire localization of locally advanced malignant breast masses in patients undergoing neoadjuvant chemotherapy and breast conservation therapy. Egypt J Radiol Nucl Med 50:96. https://doi.org/10.1186/s43055-019-0066-z
van Riet YE, Maaskant AJ, Creemers GJ, van Warmerdam LJ, Jansen FH, van de Velde CJ et al (2010) Identification of residual breast tumour localization after neo-adjuvant chemotherapy using a radioactive 125 Iodine seed. Eur J Surg Oncol 36(2):164–169. https://doi.org/10.1016/j.ejso.2009.10.009
Alderliesten T, Loo CE, Pengel KE, Rutgers EJ, Gilhuijs KG, VranckenPeeters MJ (2011) Radioactive seed localization of breast lesions: an adequate localization method without seed migration. Breast J17(6):594–601. https://doi.org/10.1111/j.1524-4741.2011.01155.x
Gobardhan PD, de Wall LL, van der Laan L, ten Tije AJ, van der Meer DC, Tetteroo E et al (2013) The role of radioactive iodine-125 seed localization in breast-conserving therapy following neoadjuvant chemotherapy. Ann Oncol 24(3):668–673. https://doi.org/10.1093/annonc/mds475
Donker M, Drukker CA, Valdés Olmos RA, Rutgers EJ, Loo CE, Sonke GS et al (2013) Guiding breast-conserving surgery in patients after neoadjuvant systemic therapy for breast cancer: a comparison of radioactive seed localization with the ROLL technique. Ann Surg Oncol 20(8):2569–2575. https://doi.org/10.1245/s10434-013-2921-x
Espinosa-Bravo M, Sao Avilés A, Esgueva A, Córdoba O, Rodriguez J, Cortadellas T et al (2011) Breast conservative surgery after neoadjuvant chemotherapy in breast cancer patients: comparison of two tumor localization methods. Eur J Surg Oncol 37(12):1038–1043. https://doi.org/10.1016/j.ejso.2011.08.136
OCEBM Levels of Evidence Working Group . Howick J, Chalmers I, Glasziou P, et al. Levels of evidence—Centre for Evidence-Based Medicine (CEBM). University of Oxford: University of Oxford: 2011. https://www.cebm.ox.ac.uk/resources/levels-of-evidence/ocebm-levels-of-evidence
Moran MS, Schnitt SJ, Giuliano AE, Harris JR, Khan SA, Horton J et al (2014) Society of Surgical Oncology-American Society for Radiation Oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Int J Radiat Oncol Biol Phys 88(3):553–564. https://doi.org/10.1016/j.ijrobp.2013.11.012
Bossuyt V, Provenzano E, Symmans WF, Boughey JC, Coles C, Curigliano G, Breast International Group-North American Breast Cancer Group (BIG-NABCG) collaboration et al (2015) Recommendations for standardized pathological characterization of residual disease for neoadjuvant clinical trials of breast cancer by the BIG-NABCG collaboration. Ann Oncol 26(7):1280–91. https://doi.org/10.1093/annonc/mdv161
Schaefgen B, Mati M, Sinn HP, Golatta M, Stieber A, Rauch G et al (2016) Can routine imaging after neoadjuvant chemotherapy in breast cancer predict pathologic complete response? Ann Surg Oncol 23(3):789–795. https://doi.org/10.1245/s10434-015-4918-0
Gampenrieder SP, Peer A, Weismann C, Meissnitzer M, Rinnerthaler G, Webhofer J et al (2019) Radiologic complete response (rCR) in contrast-enhanced magnetic resonance imaging (CE-MRI) after neoadjuvant chemotherapy for early breast cancer predicts recurrence-free survival but not pathologic complete response (pCR). Breast Cancer Res 21(1):19. https://doi.org/10.1186/s13058-018-1091-y.PMID:30704493;PMCID:PMC6357474
Author information
Authors and Affiliations
Contributions
CKJ: conceptualization, literature search, screening of the selected articles, data collection and analysis, drafting of the manuscript. GJ: literature search, screening of the selected articles, data collection, and drafting of the manuscript. SKY and US: conceptualization, screening of the selected articles, and revision of the manuscript. PKS: conceptualization, literature search, screening of the selected articles, and supervision of the study.
Corresponding author
Ethics declarations
Ethics Approval
This systematic review was carried out after registering the protocol for a systematic review with International Prospective Register of Systematic Reviews, PROSPERO (ID-CRD42020165806). The PRISMA 2009 guidelines were followed throughout this review.
Consent to Participate
Not applicable.
Consent for Publication
All authors approved the manuscript and gave consent for publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Search Strategy for PubMed
breast AND (cancer OR carcinoma OR neoplasm OR lesion) AND (mapping OR marking OR localisation OR localization OR “tissue marker clip” OR “tissue marker” OR clip OR marker OR “skin tattoo” OR tattoo OR ROLL OR ROSL OR WGL OR “wire guided” OR “needle wire” OR magseed OR hydrogel OR hydromark OR “radioactive seed” OR “radioactive” OR “seed” OR saviscout OR saviSCOUT) AND (“neoadjuvant chemotherapy” OR “neoadjuvant systemic therapy” OR “preoperative chemotherapy” OR “preoperative systemic therapy” OR “induction chemotherapy” OR “anterior chemotherapy” OR “neo-adjuvant chemotherapy” OR “neo-adjuvant systemic therapy”)
Search Strategy for Cochrane
breast in Title Abstract Keyword AND cancer OR carcinoma OR neoplasm OR lesion in Title Abstract Keyword AND mapping OR marking OR localisation OR localization OR “tissue marker clip” OR “tissue marker” OR clip OR marker OR “skin tattoo” OR tattoo OR ROLL OR ROSL OR WGL OR “wire guided” OR “needle wire” OR magseed OR hydrogel OR hydromark OR “radioactive seed” OR “radioactive” OR “seed” OR saviscout OR saviSCOUT in Title Abstract Keyword AND “neoadjuvant chemotherapy” OR “neoadjuvant systemic therapy” OR “preoperative chemotherapy” OR “preoperative systemic therapy” OR “induction chemotherapy” OR “anterior chemotherapy” OR “neo-adjuvant chemotherapy” OR “neo-adjuvant systemic therapy” in Title Abstract Keyword with Cochrane Library publication date to Sep 2020, in Trials with 'Breast Cancer' in Cochrane Groups (Word variations have been searched)
Search Strategy for Embase
(((exp BREAST/ OR (breast).ti,ab) AND (exp NEOPLASM/ OR exp "INVASIVE CARCINOMA"/ OR (carcinoma).ti,ab OR exp NEOPLASM/ OR (neoplasm).ti,ab)) AND ((localisation).ti,ab OR (localization).ti,ab OR (mapping).ti,ab OR (marking).ti,ab OR (tumour marking).ti,ab OR (tissue marking).ti,ab OR (tissue marking clip).ti,ab OR (clip).ti,ab OR (marker).ti,ab OR (skin tattoo).ti,ab OR (ROLL).ti,ab OR (ROSL).ti,ab OR (WGL).ti,ab OR (Wire guided).ti,ab OR (needle wire).ti,ab OR (magseed).ti,ab OR (hydrogel).ti,ab OR (radioactive seed).ti,ab OR (saviscout).ti,ab)) AND ((neoadjuvant chemotherapy).ti,ab OR (neoadjuvant systemic therapy).ti,ab OR (preoperative systemic therapy).ti,ab OR (preoperative chemotherapy).ti,ab OR (induction chemotherapy).ti,ab OR (anterior chemotherapy).ti,ab OR (neo- adjuvant systemic therapy).ti,ab OR (neo-adjuvant chemotherapy).ti,ab)
Rights and permissions
About this article
Cite this article
Jha, C.K., Johri, G., Singh, P.K. et al. Does Tumor Marking Before Neoadjuvant Chemotherapy Helps Achieve Better Outcomes in Patients Undergoing Breast Conservative Surgery? A Systematic Review . Indian J Surg Oncol 12, 624–631 (2021). https://doi.org/10.1007/s13193-021-01393-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-021-01393-7