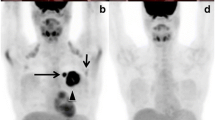
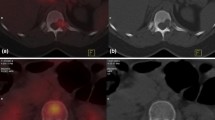
Abstract
Locally advanced breast cancer (LABC) patients require an accurate staging of the disease to rule out distant metastases. Various imaging investigations are used to stage LABC patients. The present study is a prospective comparison of conventional imaging (CI) with fusion positron-emission tomography and computed tomography (PET-CT) scans in the staging of LABC patients. Seventy-three consecutive LABC patients presenting to the breast cancer clinic of the tertiary care cancer institute were included in the study. All patients underwent contrast-enhanced computed tomography, Tv99m bone scintigraphy, and fusion PET-CT. Histology of the metastatic site was confirmed wherever possible. The disparity between the two imaging findings was compared. Doubtful lesions were observed clinically for at least 2 years to confirm their nature. PET-CT detected a higher number of lymph nodes in the axilla, internal mammary, and supraclavicular region as compared to CI. PET-CT upstaged 36.98% and downstaged 5.4% of the patients respectively leading to a change in the management in 30.13% of the patients. Sensitivity, specificity, positive predictive value, and negative predictive value of CI and PET-CT were 71.87%, 87.80%, 82.14%, and 80%, and 90.90%, 90%, 88.23%, and 92.30% respectively. PET-CT was more accurate in staging the LABC patients as compared to CI. PET-CT is more accurate then contrast-enhanced CT and bone scintigraphy for staging locally advanced breast carcinoma patients. It can replace multiple organ–directed imaging in staging breast cancer. It can provide accurate staging of the disease so that patients can be prognosticated and can be directed to the most appropriate treatment plans.

Similar content being viewed by others
References
Gerber B, Seitz E, Müller H, Krause A, Reimer T, Kundt G, Friese K (2003) Perioperative screening for metastatic disease is not indicated in patients with primary breast cancer and no clinical signs of tumor spread. Breast Cancer Res Treat 82:29–37
Müller D, Kohler G, Ohlinger R (2008) Staging procedures in primary breast cancer. Anticancer Res 28:2397–2400
Mahner S, Schirrmacher S, Brenner W, Jenicke L, Habermann CR, Avril N, Dose-Schwarz J (2008) Comparison between positron emission tomography using 2-[fluorine-18] fluoro-2-deoxy-D-glucose, conventional imaging, and computed tomography for staging of breast cancer. Ann Oncol 19(7):1249–1254
Isasi CR, Moadel RM, Blaufox MD (2005) A meta-analysis of FDG-PET for the evaluation of breast cancer recurrence and metastases. Breast Cancer Res Treat 90:105–112
Tatsumi M, Cohade C, Mourtzikos KA et al (2006) Initial experience with FDG-PET/CT in the evaluation of breast cancer. Eur J Nucl Med Mol Imaging 33(3):254–262
Rosen EL, Eubank WB, Mankoff DA (2007) FDG PET, PET/CT, and breast cancer imaging. Radiographics. 27(Suppl 1):S215–S229
Wahl RL, Siegel BA, Coleman RE, Gatsonis CG, PET Study Group (2004) Prospective multicenter study of axillary nodal staging by positron emission tomography in breast cancer: a report of the Staging Breast Cancer with PET Study Group. J Clin Oncol 22(2):277–285
Fehr MK, Hornung R, Varga Z et al (2004) Axillary staging using positron emission tomography in breast cancer patients qualifying for sentinel lymph node biopsy. Breast J 10(2):89–93
Fink D, Steinert HC et al (2004) Axillary staging using positron emission tomography in breast cancer patients qualifying for sentinel lymph node biopsy. Breast J 10:89–93
Barranger E, Grahek D, Antoine M et al (2003) Evaluation of fluorodeoxyglucose positron emission tomography in the detection of axillary lymph node metastases in patients with early-stage breast cancer. Ann Surg Oncol 10(6):622–627
Danforth DN, Aloj L, Carrasquillo JA et al (2002) The role of 18F-FDG-PET in the local/regional evaluation of women with breast cancer. Breast Cancer Res Treat 75(2):135–146
Kumar R, Chauhan A, Zhuang H, Chandra P, Schnall M, Alavi A (2006) Clinicopathologic factors associated with false-negative FDG-PET in primary breast cancer. Breast Cancer Res Treat 98(3):267–274
Zhang YJ, Oh JL, Whitman GJ, Iyengar P, Yu TK, Tereffe W, Woodward WA, Perkins G, Buchholz TA, Strom EA (2010) Clinically apparent internal mammary nodal metastasis in patients with advanced breast cancer: incidence and local control. Int J Radiat Oncol Biol Phys 77(4):1113–1119
Savaridas SL, Spratt JD, Cox J (2015) Incidence and potential significance of internal mammary lymphadenopathy on computed tomography in patients with a diagnosis of primary breast cancer. Breast Cancer (Auckl) 9:59–65
Nakai T, Okuyama C, Kubota T, Yamada K, Ushijima Y, Taniike K, Suzuki T, Nishimura T (2005) Pitfalls of FDG-PET for the diagnosis of osteoblastic bone metastases in patients with breast cancer. Eur J Nucl Med Mol Imaging 32(11):1253–1258
Cook GJ, Houston S, Rubens R, Maisey MN, Fogelman I (1998) Detection of bone metastases in breast cancer by 18FDG PET: differing metabolic activity in osteoblastic and osteolytic lesions. J Clin Oncol 16(10):3375–3379
O’Sullivan GJ, Carty FL, Cronin CG (2015) Imaging of bone metastasis: an update. World J Radiol 7(8):202–211
Clavo AC, Brown RS, Wahl RL (1995) Fluorodeoxyglucose uptake in human cancer cell lines is increased by hypoxia. J Nucl Med 36(9):1625–1632
Piccardo A, Altrinetti V, Bacigalupo L, Puntoni M, Biscaldi E, Gozza A, Cabria M, Iacozzi M, Pasa A, Morbelli S, Villavecchia G, DeCensi A (2012) Detection of metastatic bone lesions in metastatic breast cancer patient fused (18)F-Flouride- PET-MDCT has higher accuracy than MDCT. Preliminary experience. Eur J Radiol 81:2632–2638
Eubank WB, Mankoff D, Bhattacharya M, Gralow J, Linden H, Ellis G, Lindsley S, Austin-Seymour M, Livingston R (2004) Impact of FDG PET on defining the extent of disease and on the treatment of patients with recurrent or metastatic breast cancer. AJR Am J Roentgenol 183(2):479–486
Yap CS, Seltzer MA, Schippers C (2001) Impact of whole-body 18 FDG PET on staging and imaging patients with breast cancer: the referring physicians prospective. J Nucl Med 42:1334–1337
Fuster D, Duch J, Paredes P, Velasco M, Muñoz M, Santamaría G, Fontanillas M, Pons F (2008) Pre-operative staging in large primary breast cancer with 18 Fluorodeoxyglucose positron emission tomography/computed tomography compared with conventional imaging procedure. J Clin Oncol 26:4746–4751
Piperkova E, Raphael B, Altinyay ME, Castellon I, Libes R, Sandella N, Heiba S, Abdel-Dayem H (2007) Impact of PET/CT in comparison with same day contrast-enhanced CT in breast cancer management. Clin Nucl Med 32(6):429–434
Radan L, Ben-Haim S, Bar-Shalom R et al (2006) The role of FDG-PET/CT in suspected recurrence of breast cancer. Cancer 107:2545–2551
Kamel EM, Wyss MT, Fehr MK, von Schulthess GK, Goerres GW (2003) [18F]-Flourodeoxyglucose positron emission tomography in patients with suspected recurrence of breast cancer. J Cancer Res Clin Oncol 129(3):147–153
Moon DH, Maddahi J, Silverman DH, Glaspy JA, Phelps ME, Hoh CK (1998) Accuracy of whole-body fluorine-18-FDG PET for the detection of recurrent or metastatic breast carcinoma. J Nucl Med 39(3):431–435
Kim TS, Moon WK, Lee DS, Chung JK, Lee MC, Youn YK, Oh SK, Choe KJ, Noh DY (2001) Fluorodeoxyglucose positron emission tomography for detection of recurrent or metastatic breast cancer. World J Surg 25(7):829–834
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhoriwal, S., Deo, S.V.S., Kumar, R. et al. A Prospective Study Comparing the Role of 18 FDG PET-CT with Contrast-Enhanced Computed Tomography and Tc99m Bone Scan for Staging Locally Advanced Breast Cancer. Indian J Surg Oncol 12, 266–271 (2021). https://doi.org/10.1007/s13193-021-01299-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-021-01299-4