Abstract
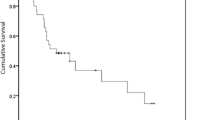
Fungating breast cancer (FBC) is a rare entity in developed nations. But this occurrence is not uncommon in our country. The aim of this study was to review clinico-pathologic profile and outcomes of FBC in a developing country. This retrospective study consisted of patients with FBC managed at our institute (Jan 2005–Dec 2015). Clinico-pathologic profile, management details, and outcomes were analyzed. The Kaplan-Meier method was used to determine overall survival (OS). Log-rank test was performed to compare survival in various subgroups. Seventy-nine patients were detected to have FBC constituting 3.3% of all breast cancers and 24.8% of those having T4b lesions. Mean age of the patients was 55 + 11 years. Ninety-six percent were women and 67% belonged to rural areas. A total of 75% women were postmenopausal. Mean duration of lump was 16 + 11 months. The mean tumor size was 8+ 2 cm. Eighty-seven percent had axillary lymph node involvement and 42% distant metastases. Fifty-eight percent (n = 46) patients had stage III and 42% (n = 33) stage had IV tumors. Hormone receptor (HR) positivity was noted in 44% (n = 35) and HER2/neu overexpression in 39% (n = 31) tumors, whereas 32% (n = 25) were triple negative. Overall, 95% (n = 75) of patients received chemotherapy, 91% (n = 72) patients underwent mastectomy, and 76% (n = 60) loco-regional radiotherapy. Median duration of follow-up was 40 (2–93) months. Median survival was 36 months, and 5-year OS was 40%. Except for stage (53% vs 22%, p = 0.005), no other factor influenced OS. Multimodality therapy in FBS results in good symptom palliation and comparable survival to stage III and IV patients without fungating tumors.

Similar content being viewed by others
References
IARC. Breast cancer screening. In: Vainio H, Bianchini F, eds. International Agency for Research on Cancer (IARC) Handbooks of Cancer Prevention. Lyon, France: IARC Press; 2002:87–117
Desantis C, Ma J, Bryan L, Jemal A (2014) Breast cancer statistics, 2013. CA Cancer J Clin 64:52–62
2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018 68(6):394–424. https://doi.org/10.3322/caac.21492
Parkin DM, Fernandez LM (2006) Use of statistics to assess the global burden of breast cancer. Breast J 12(Suppl 1):S70–S80
Farmer P, Frenk J, Knaul FM, Shulman LN, Alleyne G, Armstrong L, Atun R, Blayney D, Chen L, Feachem R, Gospodarowicz M, Gralow J, Gupta S, Langer A, Lob-Levyt J, Neal C, Mbewu A, Mired D, Piot P, Reddy KS, Sachs JD, Sarhan M, Seffrin JR (2010) Expansion of cancer care and control in countries of low and middle income: a call to action. Lancet 376(9747):1186–1193
Agarwal G, Pradeep PV, Aggarwal V, Yip CH, Cheung PS (2007) Spectrum of breast cancer in Asian women. World J Surg 31:1031–1040. https://doi.org/10.1007/s00268-005-0585-9
Charles H. Weaver CH, Deuter D NCCN guidelines for the treatment of breast cancer: changes for 2005
Hortobagyi GN et al. Breast. In Amin MB et al (edi.). AJCC Cancer Staging Manual. Eight editon: p-589
Ugalde I, Pirela D Fungating breast mass in a developed country. BMJ Case Rep Published Online First. https://doi.org/10.1136/bcr-2018-224727
Quackenbush K, Amini A, Fisher CM et al (2017) Regression of a fungating tumor after hypofractionated radiation therapy in a patient with metastatic breast cancer. Cureus 9(7):e1417. https://doi.org/10.7759/cureus.1417
Kumar A, Shah LL, Khanna S, Khanna NN (1987) Preoperative chemotherapy for fungating breast cancer. J SurgOncol 36(4):295–298
Borger JH et al (1992) Primary radiotherapy of breast cancer: treatment results in locally advanced breast cancer and in operable patients selected by positive axillary apex biopsy. RadiotherOncol 25(1):1–11
Wieland AW, Louwman MW, Voogd AC, van Beek MW, Vreugdenhil G, Roumen RM (2004) Determinants of prognosis in breast cancer patients with tumor involvement of the skin (pT4b). Breast J 10(2):123–128
Hortobágyi GN, Blumenschein GR, Spanos W et al (1983) Multimodal treatment of locoregionally advanced breast cancer. Cancer 51:763–768 CrossRefMedlineGoogle Scholar
Powles TJ, Hickish TF, Makris A et al (1995) Randomized trial of chemoendocrine therapy started before or after surgery for treatment of primary breast cancer. J ClinOncol 13:547–552
van der Hage JA, van de Velde CJ, Julien JP et al (2001) Preoperative chemotherapy in primary operable breast cancer: results from the European Organization for Research and Treatment of Cancer trial 10902. J ClinOncol 19:4224–4237
Hortobágyi GN, Ames FC, Buzdar AU et al (1988) Management of stage III primary breast cancer with primary chemotherapy, surgery, and radiation therapy. Cancer 62:2507–2516
Pierce LJ, Lippman M, Ben-Baruch N et al (1992) The effect of systemic therapy on local-regional control in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 23:949–960
Giordano SH (2003) Update on locally advanced breast cancer. Oncologist 8:521–530
Yildirim E, Semerci E, Berberoglu U (2000) The analysis of prognostic factors in stage iii-b non-inflammatory breast cancer. Eur J SurgOncol 26:34–38
Stewart JF, King RJ, Winter PJ et al (1982) Oestrogen receptors, clinical features and prognosis in stage III breast cancer. Eur J Cancer ClinOncol 18:1315–1320 CrossRefMedlineGoogle Scholar
Bonnefoi H, Diebold-Berger S, Therasse P, Hamilton A, van de Vijver M, MacGrogan G, Shepherd L, Amaral N, Duval C, Drijkoningen R, Larsimont D, Piccart M (2003) Locally advanced/inflammatory breast cancers treated with intensive epirubicin-based neoadjuvant chemotherapy: are there molecular markers in the primary tumour that predict for 5-year clinical outcome? Ann Oncol 14:406–413
Agarwal G, Nanda G, Lal P, Mishra A, Agarwal A, Agrawal V, Krishnani N (2016) Outcomes of triple-negative breast cancers (TNBC) compared with non-TNBC: does the survival vary for all stages? World J Surg 40(6):1362–1372. https://doi.org/10.1007/s00268-016-3422-4
Author information
Authors and Affiliations
Contributions
Raouef Ahmed Bichoo, Sanjay Kumar Yadav, Anjali Mishra-literature search, figures, study design, data collection, data analysis, data interpretation, and writing.
Saroj Kanta Mishra, Gyan Chand, Gaurav Agarwal, Amit Agarwal, Punita Lal-revision and editing of manuscript.
Corresponding author
Ethics declarations
This is to declare that all authors have contributed to the study. No part of the manuscript has been sent for consideration elsewhere or published in any international or national journal. The authors clearly certify that there is no aspect of plagiarism. All the conflicts of interest have been clearly defined and the source of grant disclosed. Due ethical permission/consent has been obtained for carrying out the study. In case of any dispute, the authors will be held fully responsible for the statement disclosed in the cover letter. The authors are also aware of the copyright rules and also declare that they will not reproduce any published text without due permission from the journal.
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed written consent was obtained from the patient.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bichoo, R.A., Yadav, S.K., Mishra, A. et al. Fungating Breast Cancer: Experience in Low and Middle Income Country. Indian J Surg Oncol 11, 281–286 (2020). https://doi.org/10.1007/s13193-020-01040-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-020-01040-7