Abstract
Introduction
Cyanide is a major chemical threat, and cyanide ingestion carries a higher risk for a supra-lethal dose exposure compared to inhalation but provides an opportunity for effective treatment due to a longer treatment window and a gastrointestinal cyanide reservoir that could be neutralized prior to systemic absorption. We hypothesized that orally administered cobinamide may function as a high-binding affinity scavenger and that gastric alkalinization would reduce cyanide absorption and concurrently increase cobinamide binding, further enhancing antidote effectiveness.
Methods
Thirty New Zealand white rabbits were divided into five groups and were given a lethal dose of oral cyanide poisoning (50 mg). The survival time of animals was monitored with oral cyanide alone, oral cyanide with gastric alkalinization with oral sodium bicarbonate buffer (500 mg), and in combination with either aquohydroxocobinamide or dinitrocobinamide (250 mM). Red blood cell cyanide concentration, plasma cobinamide, and thiocyanate concentrations were measured from blood samples.
Results
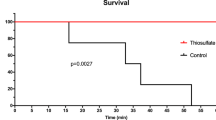
In cyanide ingested animals, oral sodium bicarbonate alone significantly prolonged survival time to 20.3 ± 8.6 min compared to 10.5 ± 4.3 min in saline-treated controls, but did not lead to overall survival. Aquohydroxocobinamide and dinitrocobinamide increased survival time to 64 ± 41 (p < 0.05) and 75 ± 16.4 min (p < 0.001), respectively. Compared to aquohydroxocobinamide, dinitrocobinamide showed greater systemic absorption and reduced blood pressure. Dinitrocobinamide also markedly increased the red blood cell cyanide concentration. Under all conditions, the plasma thiocyanate concentration gradually increased with time.
Conclusion
This study demonstrates a promising new approach to treat high-dose cyanide ingestion, with gastric alkalinization alone and in combination with oral cobinamide for treating a supra-lethal dose of orally administered cyanide in rabbits.





Similar content being viewed by others
Abbreviations
- DOS:
-
Diffuse optical spectroscopy
- FD:
-
Frequency domain
- SS:
-
Steady state
- OxyHb:
-
Oxyhemoglobin
- DeoxyHb:
-
Deoxyhemoglobin
- CytcOx:
-
Cytochrome C oxidase
References
SI B, TG B. Medical aspects of chemical and biological warfare. Chapter 10, cyanide poisoning. In: FR S, ET T, DR F, Borden Institute (U.S.), editors. Textbook of military medicine. Part I, warfare, weaponry, and the casualty. Washington, D.C.: Borden Institute, Walter Reed Army Medical Center; Office of the Surgeon General, U.S. Army; U.S. Army Medical Dept. Center and School; U.S. Army Medical Research and Material Command; Uniformed Services University of the Health Sciences; 1997. p. 272–86.
Eckstein M. Cyanide as a chemical terrorism weapon. JEMS. 2004;29(8):suppl 22–31.
Gracia R, Shepherd G. Cyanide poisoning and its treatment. Pharmacotherapy. 2004;24(10):1358–65.
Martin CO, Adams Jr HP. Neurological aspects of biological and chemical terrorism: a review for neurologists. Arch Neurol. 2003;60(1):21–5.
Dzombak DA, Ghosh RS, Wong-Chong GM. Cyanide in water and soil: chemistry, risk, and management. Boca Raton, FL: CRC Press; 2006.
Suskind R, editor. The one percent doctrine: deep inside America’s pursuit of its enemies since 9/11. New York, NY: Simon & Schuster; 2006.
Beering P. Threats on tap: understanding the terrorist threat to water. J Water Resour Plan Manag. 2002;128(3):163–7. doi:10.1061/(ASCE)0733-9496(2002)128:3(163).
Keim ME. Terrorism involving cyanide: the prospect of improving preparedness in the prehospital setting. Prehosp Disaster Med. 2006;21(SupplementS2):s56–60. doi:10.1017/S1049023X00015910.
Khan AS, Swerdlow DL, Juranek DD. Precautions against biological and chemical terrorism directed at food and water supplies. Public Health Rep. 2001;116(1):3–14.
Bhattacharya R. Antidotes to cyanide poisoning: present status. Indian J Pharm. 2000;32(2):94–101.
Cummings TF. The treatment of cyanide poisoning. Occup Med (Lond). 2004;54(2):82–5.
Bebarta VS, Tanen DA, Boudreau S, Castaneda M, Zarzabal LA, Vargas T, et al. Intravenous cobinamide versus hydroxocobalamin for acute treatment of severe cyanide poisoning in a swine (Sus scrofa) model. Ann Emerg Med. 2014. doi:10.1016/j.annemergmed.2014.02.009.
Brenner M, Kim JG, Lee J, Mahon SB, Lemor D, Ahdout R, et al. Sulfanegen sodium treatment in a rabbit model of sub-lethal cyanide toxicity. Toxicol Appl Pharmacol. 2010;248(3):269–76. doi:10.1016/j.taap.2010.08.002.
Brenner M, Kim JG, Mahon SB, Lee J, Kreuter KA, Blackledge W, et al. Intramuscular cobinamide sulfite in a rabbit model of sublethal cyanide toxicity. Ann Emerg Med. 2010;55(4):352–63. doi:10.1016/j.annemergmed.2009.12.002.
Brenner M, Mahon SB, Lee J, Kim J, Mukai D, Goodman S, et al. Comparison of cobinamide to hydroxocobalamin in reversing cyanide physiologic effects in rabbits using diffuse optical spectroscopy monitoring. J Biomed Opt. 2010;15(1):017001. doi:10.1117/1.3290816.
Broderick KE, Potluri P, Zhuang S, Scheffler IE, Sharma VS, Pilz RB, et al. Cyanide detoxification by the cobalamin precursor cobinamide. Exp Biol Med. 2006;231(5):641–9.
Chan A, Crankshaw DL, Monteil A, Patterson SE, Nagasawa HT, Briggs JE, et al. The combination of cobinamide and sulfanegen is highly effective in mouse models of cyanide poisoning. Clin Toxicol. 2011;49(5):366–73. doi:10.3109/15563650.2011.584879.
Kim JG, Lee J, Mahon SB, Mukai D, Patterson SE, Boss GR, et al. Noninvasive monitoring of treatment response in a rabbit cyanide toxicity model reveals differences in brain and muscle metabolism. J Biomed Opt. 2012;17(10):105005. doi:10.1117/1.JBO.17.10.105005.
Chan A, Balasubramanian M, Blackledge W, Mohammad OM, Alvarez L, Boss GR, et al. Cobinamide is superior to other treatments in a mouse model of cyanide poisoning. Clin Toxicol (Phila). 2010;48(7):709–17. doi:10.3109/15563650.2010.505197.
Chan A, Jiang J, Fridman A, Guo LT, Shelton GD, Liu M-T, et al. Nitrocobinamide, a new cyanide antidote that can be administered by intramuscular injection. J Med Chem. 2015;58(4):1750–9. doi:10.1021/jm501565k.
Hayward GC, Hill HA, Pratt JM, Vanston NJ, Williams RJ. The chemistry of vitamin B 12. IV. The thermodynamic trans-effect. J Chem Soc Perkin 1. 1965:6485–93.
Bebarta VS, Pitotti RL, Boudreau S, Tanen DA. Intraosseous versus intravenous infusion of hydroxocobalamin for the treatment of acute severe cyanide toxicity in a swine model. Acad Emerg Med Off J Soc Acad Emerg Med. 2014;21(11):1203–11. doi:10.1111/acem.12518.
Broderick KE, Balasubramanian M, Chan A, Potluri P, Feala J, Belke DD, et al. The cobalamin precursor cobinamide detoxifies nitroprusside-generated cyanide. Exp Biol Med. 2007;232(6):789–98.
Newhouse K, Chiu N. Toxicological review of hydrogen cyanide and cyanide salts. In: Agency USEP, editor. Washington, DC: EPA http://www.epa.gov/iris/toxreviews/0060tr.pdf; Accessed on 20 March 2016.
Atug O, Dobrucali A, Orlando R. Critical pH level of lye (NaOH) for esophageal injury. Dig Dis Sci. 2009;54(5):980–7. doi:10.1007/s10620-009-0767-7.
Bevilacqua F, Berger AJ, Cerussi AE, Jakubowski D, Tromberg BJ. Broadband absorption spectroscopy in turbid media by combined frequency-domain and steady-state methods. Appl Opt (USA). 2000;39(34):6498–507.
Lee J, Armstrong J, Kreuter K, Tromberg BJ, Brenner M. Non-invasive in vivo diffuse optical spectroscopy monitoring of cyanide poisoning in a rabbit model. Physiol Meas. 2007;28(9):1057–66. doi:10.1088/0967-3334/28/9/007.
Lee J, El-Abaddi N, Duke A, Cerussi AE, Brenner M, Tromberg BJ. Noninvasive in vivo monitoring of methemoglobin formation and reduction with broadband diffuse optical spectroscopy. J Appl Physiol. 2006;100(2):615–22. doi:10.1152/japplphysiol.00424.2004.
Lee J, Keuter KA, Kim J, Tran A, Uppal A, Mukai D, et al. Noninvasive in vivo monitoring of cyanide toxicity and treatment using diffuse optical spectroscopy in a rabbit model. Mil Med. 2009;174(6):615–21.
Lee J, Kim JG, Mahon SB, Mukai D, Yoon D, Boss GR, et al. Noninvasive optical cytochrome c oxidase redox state measurements using diffuse optical spectroscopy. J Biomed Opt. 2014;19(5):055001. doi:10.1117/1.JBO.19.5.055001.
Merritt S, Gulsen G, Chiou G, Chu Y, Deng C, Cerussi AE, et al. Comparison of water and lipid content measurements using diffuse optical spectroscopy and MRI in emulsion phantoms. Technol Cancer Res Treat. 2003;2(6):563–9.
Pham TH, Coquoz O, Fishikin JB, Anderson E, Tromberg BJ. Broad bandwidth frequency domain instrument for quantitative tissue optical spectroscopy. Rev Sci Instrum. 2000;71:2500–13.
Zijlstra WG, Buursma A, Assendelft OW. Visible and near-infrared absorption spectra of human and animal haemoglobin determination and application. Zeist, The Netherlands: CRC Press; 2000.
Rich PR, Moody AJ. Chapter 10 cytochrome c oxidase. In: Milazzo G, Graber P, editors. Treatise on bioelectrochemistry, vol. 3 Bioenergetics. Basel: Birkhauser Verlag; 1996.
Lundquist P, Rosling H, Sorbo B. Determination of cyanide in whole blood, erythrocytes, and plasma. Clin Chem. 1985;31(4):591–5.
Blackledge WC, Blackledge CW, Griesel A, Mahon SB, Brenner M, Pilz RB, et al. New facile method to measure cyanide in blood. Anal Chem. 2010;82(10):4216–21. doi:10.1021/ac100519z.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
None
Sources of Funding
This study is supported by CounterACT NIH # 1U54 NS079201, CounterACT NIH # U01 NS058030, LAMMP # 445474-30136, and AMRMC W81XWH-12-2-0098.
Rights and permissions
About this article
Cite this article
Lee, J., Mahon, S.B., Mukai, D. et al. The Vitamin B12 Analog Cobinamide Is an Effective Antidote for Oral Cyanide Poisoning. J. Med. Toxicol. 12, 370–379 (2016). https://doi.org/10.1007/s13181-016-0566-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13181-016-0566-4