Abstract
Background
Prevention and improvement of disease symptoms are important issues, and probiotics are suggested as a good treatment for controlling the obesity. Human gut microbiota has different community structures. Because gut microbial composition is assumed to be linked to probiotic function, this study evaluated the efficacy of probiotics on obesity-related clinical markers according to gut microbial enterotype.
Methods
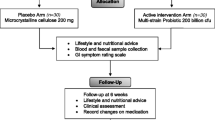
Fifty subjects with body mass index over 25 kg/m2 were randomly assigned to either the probiotic or placebo group. Each group received either unlabeled placebo or probiotic capsules for 12 weeks. Body weight, waist circumference, and body composition were measured every 3 weeks. Using computed tomography, total abdominal fat area and visceral fat area were measured. Blood and fecal samples were collected before and after the intervention for biochemical parameters and gut microbial compositions analysis.
Results
Gut microbial compositions of all the subjects were classified into two enterotypes according to Prevotella/Bacteroides ratio. The fat percentage, blood glucose, and insulin significantly increased in the Prevotella-rich enterotype of the placebo group. The obesity-related markers, such as waist circumference, total fat area, visceral fat, and ratio of visceral to subcutaneous fat area, were significantly reduced in the probiotic group. The decrease of obesity-related markers was greater in the Prevotella-rich enterotype than in the Bacteroides-rich enterotype.
Conclusion
Administration of probiotics improved obesity-related markers in obese people, and the efficacy of probiotics differed per gut microbial enterotype and greater responses were observed in the Prevotella-dominant enterotype.




Similar content being viewed by others
References
Agha M, Agha R. The rising prevalence of obesity: part A: impact on public health. Int J Surg Oncol (N Y). 2017;2(7):e17. https://doi.org/10.1097/IJ9.0000000000000017.
Van Gaal LF, Mertens IL, De Block CE. Mechanisms linking obesity with cardiovascular disease. Nature. 2006;444(7121):875–80. https://doi.org/10.1038/nature05487.
Chan JM, Rimm EB, Colditz GA, Stampfer MJ, Willett WC. Obesity, fat distribution, and weight gain as risk factors for clinical diabetes in men. Diabetes Care. 1994;17(9):961–9.
Global BMIMC, Di Angelantonio E, Bhupathiraju S, Wormser D, Gao P, Kaptoge S, et al. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet. 2016;388(10046):776–86. https://doi.org/10.1016/S0140-6736(16)30175-1.
Anandacoomarasamy A, Fransen M, March L. Obesity and the musculoskeletal system. Curr Opin Rheumatol. 2009;21(1):71–7. https://doi.org/10.1097/bor.0b013e32831bc0d7.
Harris R, Card TR, Delahooke T, Aithal GP, Guha IN. Obesity is the most common risk factor for chronic liver disease: results from a risk stratification pathway using transient elastography. Am J Gastroenterol. 2019;114(11):1744–52. https://doi.org/10.14309/ajg.0000000000000357.
Dietrich P, Hellerbrand C. Non-alcoholic fatty liver disease, obesity and the metabolic syndrome. Best Pract Res Clin Gastroenterol. 2014;28(4):637–53. https://doi.org/10.1016/j.bpg.2014.07.008.
Castro AM. Macedo-de la concha LE, Pantoja-Meléndez CA. Low-grade inflammation and its relation to obesity and chronic degenerative diseases. Revista Médica del Hospital General de México. 2017;80(2):101–5. https://doi.org/10.1016/j.hgmx.2016.06.011.
Lord GM, Matarese G, Howard JK, Baker RJ, Bloom SR, Lechler RI. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature. 1998;394(6696):897–901. https://doi.org/10.1038/29795.
Youssef DM, Elbehidy RM, Shokry DM, Elbehidy EM. The influence of leptin on Th1/Th2 balance in obese children with asthma. J Bras Pneumol. 2013;39(5):562–8. https://doi.org/10.1590/S1806-37132013000500006.
Rogero MM, Calder PC. Obesity, inflammation, Toll-like receptor 4 and fatty acids. Nutrients. 2018;10(4):432. https://doi.org/10.3390/nu10040432.
Galli C, Calder PC. Effects of fat and fatty acid intake on inflammatory and immune responses: a critical review. Ann Nutr Metab. 2009;55(1–3):123–39. https://doi.org/10.1159/000228999.
Simopoulos AP, DiNicolantonio JJ. The importance of a balanced ω-6 to ω-3 ratio in the prevention and management of obesity. Open Heart. 2016;3(2):e000385–e. https://doi.org/10.1136/openhrt-2015-000385.
Roberts CK, Barnard RJ. Effects of exercise and diet on chronic disease. J Appl Physiol (Bethesda, Md : 1985). 2005;98(1):3–30. https://doi.org/10.1152/japplphysiol.00852.2004.
Feinberg AP, Irizarry RA, Fradin D, Aryee MJ, Murakami P, Aspelund T, et al. Personalized epigenomic signatures that are stable over time and covary with body mass index. Sci Transl Med. 2010;2(49):49ra67. https://doi.org/10.1126/scitranslmed.3001262.
Alonso R, Farías M, Alvarez V, Cuevas A. Chapter 7 - the genetics of obesity. In: Rodriguez-Oquendo A, editor. Translational cardiometabolic genomic medicine. Boston: Academic Press; 2016. p. 161–77.
Romieu I, Dossus L, Barquera S, Blottiere HM, Franks PW, Gunter M, et al. Energy balance and obesity: what are the main drivers? Cancer Causes Control : CCC. 2017;28(3):247–58. https://doi.org/10.1007/s10552-017-0869-z.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027–31. https://doi.org/10.1038/nature05414.
Turnbaugh PJ, Gordon JI. The core gut microbiome, energy balance and obesity. J Physiol. 2009;587(Pt 17):4153–8. https://doi.org/10.1113/jphysiol.2009.174136.
Thaiss CA, Zmora N, Levy M, Elinav E. The microbiome and innate immunity. Nature. 2016;535:65–74. https://doi.org/10.1038/nature18847.
Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, et al. A core gut microbiome in obese and lean twins. Nature. 2008a;457:480. https://doi.org/10.1038/nature07540 https://www.nature.com/articles/nature07540#supplementary-information.
Turnbaugh PJ, Bäckhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008b;3(4):213–23. https://doi.org/10.1016/j.chom.2008.02.015.
Turnbaugh PJ, Ridaura VK, Faith JJ, Rey FE, Knight R, Gordon JI. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med. 2009;1(6):6ra14–6ra. https://doi.org/10.1126/scitranslmed.3000322.
Hughes RL, Marco ML, Hughes JP, Keim NL, Kable ME. The role of the gut microbiome in predicting response to diet and the development of precision nutrition models—part I: overview of current methods. Adv Nutr. 2019a;10(6):953–78. https://doi.org/10.1093/advances/nmz022.
Hughes RL, Kable ME, Marco M, Keim NL. The role of the gut microbiome in predicting response to diet and the development of precision nutrition models. Part II: Results. Adv Nutr. 2019b;10(6):979–98. https://doi.org/10.1093/advances/nmz049.
Bubnov R, Babenko L, Lazarenko L, Kryvtsova M, Shcherbakov O, Zholobak N, et al. Can tailored nanoceria act as a prebiotic? Report on improved lipid profile and gut microbiota in obese mice. EPMA J. 2019;10(4):317–35. https://doi.org/10.1007/s13167-019-00190-1.
Markowiak P, Śliżewska K. Effects of probiotics, prebiotics, and Synbiotics on human health. Nutrients. 2017;9(9):1021. https://doi.org/10.3390/nu9091021.
Wang L, Guo M-J, Gao Q, Yang J-F, Yang L, Pang X-L, et al. The effects of probiotics on total cholesterol: a meta-analysis of randomized controlled trials. Medicine. 2018;97(5):e9679–e. https://doi.org/10.1097/MD.0000000000009679.
Collado MC, Meriluoto J, Salminen S. Role of commercial probiotic strains against human pathogen adhesion to intestinal mucus. Lett Appl Microbiol. 2007;45(4):454–60. https://doi.org/10.1111/j.1472-765X.2007.02212.x.
Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506–14. https://doi.org/10.1038/nrgastro.2014.66.
Bubnov RV, Babenko LP, Lazarenko LM, Mokrozub VV, Demchenko OA, Nechypurenko OV, et al. Comparative study of probiotic effects of Lactobacillus and Bifidobacteria strains on cholesterol levels, liver morphology and the gut microbiota in obese mice. EPMA J. 2017;8(4):357–76. https://doi.org/10.1007/s13167-017-0117-3.
Park D-Y, Ahn Y-T, Park S-H, Huh C-S, Yoo S-R, Yu R, et al. Supplementation of Lactobacillus curvatus HY7601 and Lactobacillus plantarum KY1032 in diet-induced obese mice is associated with gut microbial changes and reduction in obesity. PLoS One. 2013;8(3):e59470. https://doi.org/10.1371/journal.pone.0059470.
Huang Y, Wang J, Quan G, Wang X, Yang L, Zhong L. <span class="named-content genus-species" id="named-content-1">Lactobacillus acidophilus</span> ATCC 4356 prevents atherosclerosis via inhibition of intestinal cholesterol absorption in apolipoprotein e-knockout mice. Appl Environ Microbiol. 2014;80(24):7496. https://doi.org/10.1128/AEM.02926-14.
Aronsson L, Huang Y, Parini P, Korach-André M, Håkansson J, Gustafsson J-Å, et al. Decreased fat storage by Lactobacillus paracasei is associated with increased levels of angiopoietin-like 4 protein (ANGPTL4). PLoS One. 2010;5(9):e13087. https://doi.org/10.1371/journal.pone.0013087.
Ma X, Hua J, Li Z. Probiotics improve high fat diet-induced hepatic steatosis and insulin resistance by increasing hepatic NKT cells. J Hepatol. 2008;49(5):821–30. https://doi.org/10.1016/j.jhep.2008.05.025.
Hidalgo-Cantabrana C, Delgado S, Ruiz L, Ruas-Madiedo P, Sánchez B, Margolles A. Bifidobacteria and their health-promoting effects. Microbiol Spectr. 2017;5(3). https://doi.org/10.1128/microbiolspec.BAD-0010-2016.
Thomson P, Medina DA, Garrido D. Human milk oligosaccharides and infant gut bifidobacteria: molecular strategies for their utilization. Food Microbiol. 2018;75:37–46. https://doi.org/10.1016/j.fm.2017.09.001.
Ruiz L, Delgado S, Ruas-Madiedo P, Sánchez B, Margolles A. Bifidobacteria and their molecular communication with the immune system. Front Microbiol. 2017;8:2345. https://doi.org/10.3389/fmicb.2017.02345.
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, et al. Human gut microbiome viewed across age and geography. Nature. 2012;486(7402):222–7. https://doi.org/10.1038/nature11053.
Moschen AR, Wieser V, Tilg H. Dietary factors: major regulators of the gut’s microbiota. Gut Liver. 2012;6(4):411–6. https://doi.org/10.5009/gnl.2012.6.4.411.
Wu GD, Chen J, Hoffmann C, Bittinger K, Chen YY, Keilbaugh SA, et al. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334(6052):105–8. https://doi.org/10.1126/science.1208344.
Lim MY, Rho M, Song Y-M, Lee K, Sung J, Ko G. Stability of gut enterotypes in Korean monozygotic twins and their association with biomarkers and diet. Sci Rep. 2014;4:7348. https://doi.org/10.1038/srep07348 https://www.nature.com/articles/srep07348#supplementary-information.
Liang C, Tseng H-C, Chen H-M, Wang W-C, Chiu C-M, Chang J-Y, et al. Diversity and enterotype in gut bacterial community of adults in Taiwan. BMC Genomics. 2017;18(1):932. https://doi.org/10.1186/s12864-016-3261-6.
Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, et al. Enterotypes of the human gut microbiome. Nature. 2011;473(7346):174–80. https://doi.org/10.1038/nature09944.
de Moraes AC, Fernandes GR, da Silva IT, Almeida-Pititto B, Gomes EP, Pereira AD, et al. Enterotype may drive the dietary-associated cardiometabolic risk factors. Front Cell Infect Microbiol. 2017;7:47. https://doi.org/10.3389/fcimb.2017.00047.
Roager HM, Licht TR, Poulsen SK, Larsen TM, Bahl MI. Microbial enterotypes, inferred by the prevotella-to-bacteroides ratio, remained stable during a 6-month randomized controlled diet intervention with the new nordic diet. Appl Environ Microbiol. 2014;80(3):1142–9. https://doi.org/10.1128/aem.03549-13.
Hjorth MF, Roager HM, Larsen TM, Poulsen SK, Licht TR, Bahl MI, et al. Pre-treatment microbial Prevotella-to-Bacteroides ratio, determines body fat loss success during a 6-month randomized controlled diet intervention. Int J Obes. 2017;42:580–3. https://doi.org/10.1038/ijo.2017.220.
Hjorth MF, Blaedel T, Bendtsen LQ, Lorenzen JK, Holm JB, Kiilerich P, et al. Prevotella-to-Bacteroides ratio predicts body weight and fat loss success on 24-week diets varying in macronutrient composition and dietary fiber: results from a post-hoc analysis. Int J Obes (Lond). 2019;43(1):149–57. https://doi.org/10.1038/s41366-018-0093-2.
Organization WH. The Asia-Pacific perspective: redefining obesity and its treatment. Sydney: Health Communications Australia; 2000.
Organization WH. Waist circumference and waist-hip ratio: report of a WHO expert consultation, Geneva, 8-11 December 2008. 2011.
Perry AC, Applegate EB, Jackson ML, Deprima S, Goldberg RB, Ross R, et al. Racial differences in visceral adipose tissue but not anthropometric markers of health-related variables. J Appl Physiol (Bethesda, Md : 1985). 2000;89(2):636–43. https://doi.org/10.1152/jappl.2000.89.2.636.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502.
Lim MY, Song E-J, Kim SH, Lee J, Nam Y-D. Comparison of DNA extraction methods for human gut microbial community profiling. Syst Appl Microbiol. 2018;41(2):151–7. https://doi.org/10.1016/j.syapm.2017.11.008.
Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30(15):2114–20. https://doi.org/10.1093/bioinformatics/btu170.
Zhang J, Kobert K, Flouri T, Stamatakis A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics. 2014;30(5):614–20. https://doi.org/10.1093/bioinformatics/btt593.
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics. 2011;27(16):2194–200. https://doi.org/10.1093/bioinformatics/btr381.
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet C, Al-Ghalith GA, et al. QIIME 2: reproducible, interactive, scalable, and extensible microbiome data science. Peer J Preprints. 2018;6:e27295v2. https://doi.org/10.7287/peerj.preprints.27295v2.
Janssen I, Katzmarzyk PT, Ross R. Body mass index, waist circumference, and health risk: evidence in support of current National Institutes of Health guidelines. Arch Intern Med. 2002;162(18):2074–9.
Janssen I, Katzmarzyk PT, Ross R. Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr. 2004;79(3):379–84.
Jung SP, Lee KM, Kang JH, Yun SI, Park HO, Moon Y, et al. Effect of Lactobacillus gasseri BNR17 on overweight and obese adults: a randomized, double-blind clinical trial. Korean J Fam Med. 2013;34(2):80–9. https://doi.org/10.4082/kjfm.2013.34.2.80.
Matsuzawa Y, Nakamura T, Shimomura I, Kotani K. Visceral fat accumulation and cardiovascular disease. Obes Res. 1995;3(Suppl 5):645s–7s.
Despres JP. Cardiovascular disease under the influence of excess visceral fat. Crit Pathw Cardiol. 2007;6(2):51–9. https://doi.org/10.1097/HPC.0b013e318057d4c9.
Fukuda T, Bouchi R. Ratio of visceral-to-subcutaneous fat area predicts cardiovascular events in patients with type 2 diabetes. J Diabetes Investig. 2018;9:396-402. https://doi.org/10.1111/jdi.12713.
Kaess BM, Pedley A, Massaro JM, Murabito J, Hoffmann U, Fox CS. The ratio of visceral to subcutaneous fat, a metric of body fat distribution, is a unique correlate of cardiometabolic risk. Diabetologia. 2012;55(10):2622–30. https://doi.org/10.1007/s00125-012-2639-5.
Barreto FM, Colado Simão AN, Morimoto HK, Batisti Lozovoy MA, Dichi I, Helena da Silva Miglioranza L. Beneficial effects of Lactobacillus plantarum on glycemia and homocysteine levels in postmenopausal women with metabolic syndrome. Nutrition 2014;30(7):939–942. doi:https://doi.org/10.1016/j.nut.2013.12.004.
Takemura N, Okubo T, Sonoyama K. Lactobacillus plantarum strain No. 14 reduces adipocyte size in mice fed high-fat diet. Exp Biol Med (Maywood, NJ). 2010;235(7):849–56. https://doi.org/10.1258/ebm.2010.009377.
Takahashi S, Anzawa D, Takami K, Ishizuka A, Mawatari T, Kamikado K, et al. Effect of Bifidobacterium animalis ssp. lactis GCL2505 on visceral fat accumulation in healthy Japanese adults: a randomized controlled trial. Biosci Microbiota Food Health. 2016;35(4):163–71. https://doi.org/10.12938/bmfh.2016-002.
Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, et al. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur J Clin Nutr. 2010;64(6):636–43. https://doi.org/10.1038/ejcn.2010.19.
Kortman GAM, Raffatellu M, Swinkels DW, Tjalsma H. Nutritional iron turned inside out: intestinal stress from a gut microbial perspective. FEMS Microbiol Rev. 2014;38(6):1202–34. https://doi.org/10.1111/1574-6976.12086.
Das NK, Schwartz AJ, Barthel G, Inohara N, Liu Q, Sankar A, et al. Microbial metabolite signaling is required for systemic iron homeostasis. Cell Metabolism. 2019;S1550–4131(19):30560–1. https://doi.org/10.1016/j.cmet.2019.10.005.
Skrypnik K, Suliburska J. Association between the gut microbiota and mineral metabolism. J Sci Food Agric. 2018;98(7):2449–60. https://doi.org/10.1002/jsfa.8724.
Raimondi S, Amaretti A, Leonardi A, Quartieri A, Gozzoli C, Rossi M. Conjugated linoleic acid production by Bifidobacteria: screening, kinetic, and composition. Biomed Res Int. 2016;2016:8654317. https://doi.org/10.1155/2016/8654317.
Kim JH, Kim Y, Kim YJ, Park Y. Conjugated linoleic acid: potential health benefits as a functional food ingredient. Annu Rev Food Sci Technol. 2016a;7:221–44. https://doi.org/10.1146/annurev-food-041715-033028.
Patterson E, Wall R, Lisai S, Ross RP, Dinan TG, Cryan JF, et al. Bifidobacterium breve with α-linolenic acid alters the composition, distribution and transcription factor activity associated with metabolism and absorption of fat. Sci Rep. 2017;7:43300. https://doi.org/10.1038/srep43300 https://www.nature.com/articles/srep43300#supplementary-information.
Kondo S, Kamei A, Xiao JZ, Iwatsuki K, Abe K. Bifidobacterium breve B-3 exerts metabolic syndrome-suppressing effects in the liver of diet-induced obese mice: a DNA microarray analysis. Benefic Microbes. 2013;4(3):247–51. https://doi.org/10.3920/BM2012.0019.
Kondo S, Xiao J-z, Satoh T, Odamaki T, Takahashi S, Sugahara H, et al. Antiobesity effects of Bifidobacterium breve strain B-3 supplementation in a mouse model with high-fat diet-induced obesity. Biosci Biotechnol Biochem. 2010;74(8):1656–61. https://doi.org/10.1271/bbb.100267.
Molly K, Smet ID, Nollet L, Woestyne MV, Verstraete W. Effect of lactobacilli on the ecology of the gastro-intestinal microbiota cultured in the SHIME reactor. Microb Ecol Health Dis. 1996;9(2):79–89. https://doi.org/10.3109/08910609609166446.
Walter J. Ecological role of lactobacilli in the gastrointestinal tract: implications for fundamental and biomedical research. Appl Environ Microbiol. 2008;74(16):4985–96. https://doi.org/10.1128/AEM.00753-08.
Wu C-C, Weng W-L, Lai W-L, Tsai H-p, Liu W-H, Lee M-H et al. Effect of Lactobacillus plantarum strain K21 on high-fat diet-fed obese mice. Evid Based Complement Alternat Med. 2015;2015:391767. doi:DOI:https://doi.org/10.1155/2015/391767.
Million M, Angelakis E, Paul M, Armougom F, Leibovici L, Raoult D. Comparative meta-analysis of the effect of Lactobacillus species on weight gain in humans and animals. Microb Pathog. 2012;53(2):100–8. https://doi.org/10.1016/j.micpath.2012.05.007.
Michael DR, Davies TS, Moss JWE, Calvente DL, Ramji DP, Marchesi JR, et al. The anti-cholesterolaemic effect of a consortium of probiotics: an acute study in C57BL/6J mice. Sci Rep. 2017;7(1):2883. https://doi.org/10.1038/s41598-017-02889-5.
Mastromarino P, Macchia S, Meggiorini L, Trinchieri V, Mosca L, Perluigi M, et al. Effectiveness of Lactobacillus-containing vaginal tablets in the treatment of symptomatic bacterial vaginosis. Clin Microbiol Infect. 2009;15(1):67–74. https://doi.org/10.1111/j.1469-0691.2008.02112.x.
Kobyliak N, Conte C, Cammarota G, Haley AP, Styriak I, Gaspar L, et al. Probiotics in prevention and treatment of obesity: a critical view. Nutri Metab. 2016;13:14. https://doi.org/10.1186/s12986-016-0067-0.
Brusaferro A, Cozzali R, Orabona C, Biscarini A, Farinelli E, Cavalli E, et al. Is it time to use probiotics to prevent or treat obesity? Nutrients. 2018;10(11). https://doi.org/10.3390/nu10111613.
X-M L, HA L, M K, E-S P, K-Y P. Probiotic effects of Lactobacillus plantarum strains isolated from Kimchi. J Korean Soc Food Sci Nutr. 2016;45(12):1717–24.
Kim HW, Hong R, Choi EY, Yu K, Kim N, Hyeon JY, et al. A probiotic mixture regulates T cell balance and reduces atopic dermatitis symptoms in mice. Front Microbiol. 2018a;9(2414). https://doi.org/10.3389/fmicb.2018.02414.
Lee SJ, Bose S, Seo J-G, Chung W-S, Lim C-Y, Kim H. The effects of co-administration of probiotics with herbal medicine on obesity, metabolic endotoxemia and dysbiosis: a randomized double-blind controlled clinical trial. Clin Nutr. 2014;33(6):973–81. https://doi.org/10.1016/j.clnu.2013.12.006.
Kwak M-J, Yoon J-K, Kwon S-K, Chung M-J, Seo J-G, Kim JF. Complete genome sequence of the probiotic bacterium Bifidobacterium breve KCTC 12201BP isolated from a healthy infant. J Biotechnol. 2015;214:156–7. https://doi.org/10.1016/j.jbiotec.2015.09.035.
Shin J-H, Chung M-J, Seo J-G. A multistrain probiotic formulation attenuates skin symptoms of atopic dermatitis in a mouse model through the generation of CD4(+)Foxp3(+) T cells. Food Nutr Res. 2016;60:32550. https://doi.org/10.3402/fnr.v60.32550.
Kim MS, Byun JS, Yoon YS, Yum DY, Chung MJ, Lee JC. A probiotic combination attenuates experimental colitis through inhibition of innate cytokine production. Benefic Microbes. 2016b;8(2):231–41. https://doi.org/10.3920/BM2016.0031.
J-S L, M-J C, J-G S. In vitro evaluation of antimicrobial activity of lactic acid bacteria against Clostridium difficile. Toxicological Research. 2013;29(2):99–106.
Chung M-J. Efficacy and safety evaluation of anti-obesity probiotics. Unpublished raw data. 2017.
Canada Go. Accepted claims about the nature of probiotic microorganisms in food. 2019. http://www.hc-sc.gc.ca/fn-an/label-etiquet/claims-reclam/probiotics_claims-allegations_probiotiques-eng.php%20.
Hobbs CA, Saigo K, Koyanagi M, Hayashi S-M. Magnesium stearate, a widely-used food additive, exhibits a lack of in vitro and in vivo genotoxic potential. Toxicol Rep. 2017;4:554–9. https://doi.org/10.1016/j.toxrep.2017.10.003.
Gibson GR, Hutkins R, Sanders ME, Prescott SL, Reimer RA, Salminen SJ, et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol. 2017;14(8):491–502. https://doi.org/10.1038/nrgastro.2017.75.
Reid G, Abrahamsson T, Bailey M, Bindels LB, Bubnov R, Ganguli K, et al. How do probiotics and prebiotics function at distant sites? Benefic Microbes. 2017;8(4):521–33. https://doi.org/10.3920/BM2016.0222.
Andersen CJ, Murphy KE, Fernandez ML. Impact of obesity and metabolic syndrome on immunity. Adv Nutr. 2016;7(1):66–75. https://doi.org/10.3945/an.115.010207.
Cheru L, Saylor CF, Lo J. Gastrointestinal barrier breakdown and adipose tissue inflammation. Curr Obes Rep. 2019;8(2):165–74. https://doi.org/10.1007/s13679-019-00332-6.
Chang C-C, Sia K-C, Chang J-F, Lin C-M, Yang C-M, Huang K-Y, et al. Lipopolysaccharide promoted proliferation and adipogenesis of preadipocytes through JAK/STAT and AMPK-regulated cPLA2 expression. Int J Med Sci. 2019;16(1):167–79. https://doi.org/10.7150/ijms.24068.
Moss RB, Moll T, El-Kalay M, Kohne C, Soo Hoo W, Encinas J, et al. Th1/Th2 cells in inflammatory disease states: therapeutic implications. Expert Opin Biol Ther. 2004;4(12):1887–96. https://doi.org/10.1517/14712598.4.12.1887.
Charlton B, Lafferty KJ. The Th1/Th2 balance in autoimmunity. Curr Opin Immunol. 1995;7(6):793–8. https://doi.org/10.1016/0952-7915(95)80050-6.
Mazzarella G, Bianco A, Catena E, De Palma R, Abbate GF. Th1/Th2 lymphocyte polarization in asthma. Allergy. 2000;55(Suppl 61):6–9. https://doi.org/10.1034/j.1398-9995.2000.00511.x.
Han JM, Levings MK. Immune regulation in obesity-associated adipose inflammation. J Immunol. 2013;191(2):527. https://doi.org/10.4049/jimmunol.1301035.
Richardson VR, Smith KA, Carter AM. Adipose tissue inflammation: feeding the development of type 2 diabetes mellitus. Immunobiology. 2013;218(12):1497–504. https://doi.org/10.1016/j.imbio.2013.05.002.
Savcheniuk OA, Virchenko OV, Falalyeyeva TM, Beregova TV, Babenko LP, Lazarenko LM, et al. The efficacy of probiotics for monosodium glutamate-induced obesity: dietology concerns and opportunities for prevention. EPMA J. 2014;5(1):2. https://doi.org/10.1186/1878-5085-5-2.
Tymoshok NO, Lazarenko LM, Bubnov RV, Shynkarenko LN, Babenko LP, Mokrozub VV, et al. New aspects the regulation of immune response through balance Th1/Th2 cytokines. EPMA J. 2014;5(1):A134. https://doi.org/10.1186/1878-5085-5-S1-A134.
Мokrozub VV, Lazarenko LM, Sichel LM, Babenko LP, Lytvyn PM, Demchenko OM, et al. The role of beneficial bacteria wall elasticity in regulating innate immune response. EPMA J. 2015;6(1):13–5. https://doi.org/10.1186/s13167-015-0035-1.
Shockman GD, Barren JF. Structure, function, and assembly of cell walls of gram-positive bacteria. Annu Rev Microbiol 1983;37(1):501–527. doi:https://doi.org/10.1146/annurev.mi.37.100183.002441.
Zeuthen LH, Fink LN, Frøkiær H. Toll-like receptor 2 and nucleotide-binding oligomerization domain-2 play divergent roles in the recognition of gut-derived lactobacilli and bifidobacteria in dendritic cells. Immunology. 2008;124(4):489–502. https://doi.org/10.1111/j.1365-2567.2007.02800.x.
Borriello SP, Hammes WP, Holzapfel W, Marteau P, Schrezenmeir J, Vaara M, et al. Safety of probiotics that contain lactobacilli or bifidobacteria. Clin Infect Dis. 2003;36(6):775–80. https://doi.org/10.1086/368080.
Boyle RJ, Robins-Browne RM, Tang MLK. Probiotic use in clinical practice: what are the risks? Am J Clin Nutr. 2006;83(6):1256–447. https://doi.org/10.1093/ajcn/83.6.1256.
Schork NJ. Personalized medicine: time for one-person trials. Nature. 2015;520(7549):609–11. https://doi.org/10.1038/520609a.
Golubnitschaja O, Baban B, Boniolo G, Wang W, Bubnov R, Kapalla M, et al. Medicine in the early twenty-first century: paradigm and anticipation - EPMA position paper 2016. EPMA J. 2016;7(1):23. https://doi.org/10.1186/s13167-016-0072-4.
Nebert DW, Zhang G, Vesell ES. From human genetics and genomics to pharmacogenetics and pharmacogenomics: past lessons. Future Directions Drug Metabolism Reviews. 2008;40(2):187–224. https://doi.org/10.1080/03602530801952864.
John GK, Wang L, Nanavati J, Twose C, Singh R, Mullin G. Dietary alteration of the gut microbiome and its impact on weight and fat mass: a systematic review and meta-analysis. Genes. 2018;9(3). https://doi.org/10.3390/genes9030167.
Park S, Bae J-H. Probiotics for weight loss: a systematic review and meta-analysis. Nutr Res. 2015;35(7):566–75. https://doi.org/10.1016/j.nutres.2015.05.008.
Borgeraas H, Johnson LK, Skattebu J, Hertel JK, Hjelmesaeth J. Effects of probiotics on body weight, body mass index, fat mass and fat percentage in subjects with overweight or obesity: a systematic review and meta-analysis of randomized controlled trials. Obes Rev. 2018;19(2):219–32. https://doi.org/10.1111/obr.12626.
Korem T, Zeevi D, Zmora N, Weissbrod O, Bar N, Lotan-Pompan M, et al. Bread affects clinical parameters and induces gut microbiome-associated personal glycemic responses. Cell Metab. 2017;25(6):1243–53.e5. https://doi.org/10.1016/j.cmet.2017.05.002.
Kang C, Zhang Y, Zhu X, Liu K, Wang X, Chen M, et al. Healthy subjects differentially respond to dietary capsaicin correlating with specific gut enterotypes. J Clin Endocrinol Metab. 2016;101(12):4681–9. https://doi.org/10.1210/jc.2016-2786.
Christensen L, Roager HM, Astrup A, Hjorth MF. Microbial enterotypes in personalized nutrition and obesity management. Am J Clin Nutr. 2018a;108(4):645–51. https://doi.org/10.1093/ajcn/nqy175.
Gu Y, Wang X, Li J, Zhang Y, Zhong H, Liu R, et al. Analyses of gut microbiota and plasma bile acids enable stratification of patients for antidiabetic treatment. Nat Commun. 2017a;8(1):1785. https://doi.org/10.1038/s41467-017-01682-2.
Chen T, Long W, Zhang C, Liu S, Zhao L, Hamaker BR. Fiber-utilizing capacity varies in Prevotella- versus Bacteroides-dominated gut microbiota. Scientific reports. 2017;7(1):2594. https://doi.org/10.1038/s41598-017-02995-4.
Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016;7(3):189–200. https://doi.org/10.1080/19490976.2015.1134082.
Kim YA, Keogh JB, Clifton PM. Probiotics, prebiotics, synbiotics and insulin sensitivity. Nutr Res Rev. 2018b;31(1):35–51. https://doi.org/10.1017/S095442241700018X.
Zmora N, Zeevi D, Korem T, Segal E, Elinav E. Taking it personally: personalized utilization of the human microbiome in health and disease. Cell Host Microbe. 2016;19(1):12–20. https://doi.org/10.1016/j.chom.2015.12.016.
Bubnov RV, Babenko LP, Lazarenko LM, Mokrozub VV, Demchenko OA, Nechypurenko OV, et al. Comparative study of probiotic effects of Lactobacillus and Bifidobacteria strains on cholesterol levels, liver morphology and the gut microbiota in obese mice. EPMA J. 2017;8(4):357–76. https://doi.org/10.1007/s13167-017-0117-3.
Bubnov RV, Spivak MY, Lazarenko LM, Bomba A, Boyko NV. Probiotics and immunity: provisional role for personalized diets and disease prevention. EPMA J. 2015b;6(1):14. https://doi.org/10.1186/s13167-015-0036-0.
Christensen L, Roager HM, Astrup A, Hjorth MF. Microbial enterotypes in personalized nutrition and obesity management. Am J Clin Nutr. 2018b;108(4):645–51. https://doi.org/10.1093/ajcn/nqy175.
Costea PI, Hildebrand F, Arumugam M, Bäckhed F, Blaser MJ, Bushman FD, et al. Enterotypes in the landscape of gut microbial community composition. Nat Microbiol. 2018;3(1):8–16. https://doi.org/10.1038/s41564-017-0072-8.
Gu Y, Wang X, Li J, Zhang Y, Zhong H, Liu R, et al. Analyses of gut microbiota and plasma bile acids enable stratification of patients for antidiabetic treatment. Nat Commun. 2017b;8(1):1785. https://doi.org/10.1038/s41467-017-01682-2.
de la Cuesta-Zuluaga J, Kelley ST, Chen Y, Escobar JS, Mueller NT, Ley RE, et al. Age- and sex-dependent patterns of gut microbial diversity in human adults msystems. 2019;4(4):e00261–19. https://doi.org/10.1128/mSystems.00261-19.
Zhang Q, Wang Y. Socioeconomic inequality of obesity in the United States: do gender, age, and ethnicity matter? Soc Sci Med. 2004;58(6):1171–80. https://doi.org/10.1016/s0277-9536(03)00288-0.
Santos-Marcos JA, Rangel-Zuñiga OA, Jimenez-Lucena R, Quintana-Navarro GM, Garcia-Carpintero S, Malagon MM, et al. Influence of gender and menopausal status on gut microbiota. Maturitas. 2018;116:43–53. https://doi.org/10.1016/j.maturitas.2018.07.008.
Funding
This research was supported by Main Research Program (E0170601-03) of the Korea Food Research Institute (KFRI) funded by the Ministry of Science and ICT.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
This study was conducted according to the principles of Declaration of Helsinki and good clinical practice guidelines. The study protocol was approved by the Institutional Review Board of Ilsan Dongguk University Hospital (approval number 2016-02) and registered in Clinical Research Information Service (CRIS identifier: KCT0002292). Written informed consent has also been obtained from all the participants.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Song, EJ., Han, K., Lim, TJ. et al. Effect of probiotics on obesity-related markers per enterotype: a double-blind, placebo-controlled, randomized clinical trial. EPMA Journal 11, 31–51 (2020). https://doi.org/10.1007/s13167-020-00198-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13167-020-00198-y