Abstract
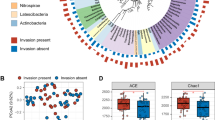
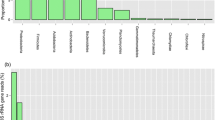
The microbial community in peat is responsible for organic matter degradation and greenhouse gas emissions, yet its response to peat extraction and peatland restoration remains poorly understood. We investigated how different physicochemical conditions in natural, restored, unrestored, and actively extracted peatlands influenced the methanogenic and methanotrophic community characteristics. 16S rRNA amplicon sequencing allowed for the comparison of the abundance of both groups. The communities were similar at sites restored in 1991 and 2009 (25 and 7 years prior to our research) that had high water table and were dominated by sedges. A different, shared pattern of microbial membership was observed at natural, unrestored, and actively extracted sites, and a site restored in 2012 (4 years prior to the research). These similarities were reflected in peat chemistry and hydrology in canonical correspondence analysis (CCA) ordination that was used to investigate how these factors affected the abundances of methane cycling taxa. Based on the CCA, methanotrophs reached their highest abundances close to the water table, at high and moderate concentrations of phosphate and propionate, and low concentrations of formate. Methanogens exhibited a more distributed pattern, with organisms responding to opposite conditions along these environmental gradients. Methane-cycling community in older restored sites departed from the reference community at the natural bog likely due to the fen-like conditions that developed at these sites.







Similar content being viewed by others
Data Availability
The raw reads are available in the NCBI SRA database under BioProject number: PRJNA640052, BioSample numbers: SAMN15297727—SAMN15297845. Raw peat chemistry and quality control data are available on Scholars Portal Dataverse https://doi.org/10.5683/SP2/R6FCJV.
References
Aggenbach CJ, Backx H, Emsens WJ, Grootjans AP, Lamers LP, Smolders AJ, Stuyfzand P, Wolejko P, Van Diggelen R (2013) Do high iron concentrations in rewetted rich fens hamper restoration. Preslia 85(3):405–420
Andersen R, Chapman SJ, and Artz R (2013a) Microbial communities in natural and disturbed peatlands: A review. Soil Biology and Biochemistry 57:979–994. http://dx.doi.org.proxy.lib.uwaterloo.ca/https://doi.org/10.1016/j.soilbio.2012.10.003
Andersen R, Francez A, Rochefort L (2006) The physicochemical and microbiological status of a restored bog in Quebec: Identification of relevant criteria to monitor success. Soil Biology and Biochemistry 38(6):1375–1387. https://doi.org/10.1016/j.soilbio.2005.10.012
Andersen R, Grasset L, Thormann MN, Rochefort L, Francez A (2010) Changes in microbial community structure and function following sphagnum peatland restoration. Soil Biology and Biochemistry 42(2):291–301. https://doi.org/10.1016/j.soilbio.2009.11.006
Andersen R, Wells C, Macrae M, Price J (2013b) Nutrient mineralisation and microbial functional diversity in a restored bog approach natural conditions 10 years post restoration. Soil Biology and Biochemistry 64:37–47. https://doi.org/10.1016/j.soilbio.2013.04.004
Artz RR, Chapman SJ, Siegenthaler A, Mitchell EA, Buttler A, Bortoluzzi E, Gilbert D, Yli-Petays M, Vasander H, Francez A (2008) Functional microbial diversity in regenerating cutover peatlands responds to vegetation succession. Journal of Applied Ecology 45(6):1799–1809. https://doi.org/10.1111/j.1365-2664.2008.01573.x
Auestad I, Rydgren K, Halvorsen R (2020) Dataset properties affect the performance of the ordination regression-based approach (ORBA) in predicting time to recovery. Ecological Engineering 152(105875):1–10. https://doi.org/10.1016/j.ecoleng.2020.105875
Baird AJ, Beckwith CW, Waldron S, Waddington JM (2004) Ebullition of methane-containing gas bubbles from near-surface Sphagnum peat. Geophysical Research Letters 31(21):1–4. Art. Number L21505. https://doi.org/10.1029/2004GL021157
Basiliko N, Henry K, Gupta V, Moore T, Driscoll B, Dunfield P (2013) Controls on bacterial and archaeal community structure and greenhouse gas production in natural, mined, and restored Canadian peatlands. Frontiers in Microbiology 4(Art. 215):1–14, https://doi.org/10.3389/fmicb.2013.00215
Basiliko N, Yavitt J, Dees P, Merkel S (2003) Methane biogeochemistry and methanogen communities in two northern peatland ecosystems. New York State Geomicrobiology Journal 20(6):563–577. https://doi.org/10.1080/713851165
Belova SE, Baani M, Suzina NE, Bodelier PL, Liesack W, Dedysh SN (2011) Acetate utilization as a survival strategy of peat-inhabiting Methylocystis spp. Environmental Microbiology Reports 3(1):36–46. https://doi.org/10.1111/j.1758-2229.2010.00180.x
Bieniada A, Hug LA, Parsons CT, Strack M (2021) Experimental determination of methane production and consumption potential in unrestored, restored and natural peatlands. https://doi.org/10.5683/SP2/YXRBUT, Scholars Portal Dataverse, V1
Bieniada A, Hug LA, Parsons CT, Strack M (2020) Peat chemistry: Raw data and quality control, Seba Beach peat samples used for microbial analyses (a). Peat chemistry: Raw data and quality control, Seba Beach peat samples used for microcosms (b). https://doi.org/10.5683/SP2/R6FCJV, Scholars Portal Dataverse, V1
Bieniada A, Strack M (2021) Steady and ebullitive methane fluxes from active, restored and unrestored horticultural peatlands. Ecological Engineering 169(Art. 106324):1–14. https://doi.org/10.1016/j.ecoleng.2021.106324
Bisanz JE (2018) qiime2R: Importing QIIME2 artifacts and associated data into R sessions. v0.99. https://github.com/jbisanz/qiime2R. Accessed 17 March 2020
Blodau C, Moore TR (2003) Experimental response of peatland carbon dynamics to a water table fluctuation. Aquatic Sciences 65(1):47–62. https://doi.org/10.1007/s000270300004
Bonn A, Reed MS, Evans CD, Joosten H, Bain C, Farmer J, Emmer I, Couwenberg J, Moxey A, Artz R, Tannenberger F, von Unger M, Smyth M-A, Birnie D (2014) Investing in nature: Developing ecosystem service markets for peatland restoration. Ecosystem Services 9:54–65. https://doi.org/10.1016/j.ecoser.2014.06.011
Bossio DA, Fleck JA, Scow KM, Fujii R (2006) Alteration of soil microbial communities and water quality in restored wetlands. Soil Biology and Biochemistry 38(6):1223–1233. https://doi.org/10.1016/j.soilbio.2005.09.027
Bolyen E, Rideout JR, Dillon MR et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nature Biotechnology 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Boudreau S, Rochefort (2008) Plant establishment in restored peatlands: 10-years monitoring of sites restored from 1995 to 2003. Paper presented at the Proceedings of the 13th International Peat Congress: After Wise Use–The Future of Peatlands 1:362–366. https://peatlands.org/document/plant-establishment-in-restored-peatlands-10-years-monitoring-of-sites-restored-from-1995-to-2003/. Accessed 10 Apr 2019
Bridgham SD, Cadillo-Quiroz H, Keller JK, Zhuang Q (2013) Methane emissions from wetlands: Biogeochemical, microbial, and modeling perspectives from local to global scales. Global Change Biology 19(5):1325–1346. https://doi.org/10.1111/gcb.12131
Brudvig LA (2017) Toward prediction in the restoration of biodiversity. J Appl Ecol 54(4):1013–1017. https://doi.org/10.1111/1365-2664.12940
Brudvig LA, Barak RS, Bauer JT, Caughlin TT, Laughlin DC, Larios L, Matthews JW, Stuble KL, Turley NE, Zirbel CR (2017) Interpreting variation to advance predictive restoration science. Journal of Applied Ecology 54:1018–1027. https://doi.org/10.1111/1365-2664.12938
Cadillo-Quiroz H, Bräuer S, Yashiro E, Sun C, Yavitt J, Zinder S (2006) Vertical profiles of methanogenesis and methanogens in two contrasting acidic peatlands in central New York state, USA. Environmental Microbiology 8(8):1428–1440. https://doi.org/10.1111/j.1462-2920.2006.01036.x
Cadillo-Quiroz H, Yavitt JB, Zinder SH, Thies JE (2010) Diversity and community structure of Archaea inhabiting the rhizoplane of two contrasting plants from an acidic bog. Microbial Ecology 59(4):757–767. https://doi.org/10.1007/s00248-009-9628-3
Chroňáková A, Bárta J, Kaštovská E, Urbanová Z, Picek T (2019) Spatial heterogeneity of belowground microbial communities linked to peatland microhabitats with different plant dominants. FEMS Microbiology Ecology 95(9):1–13. Art. fiz130. https://doi.org/10.1093/femsec/fiz130
Cleary J, Roulet NT, Moore TR (2005) Greenhouse Gas Emissions from Canadian Peat Extraction, 1990–2000: A Life-cycle Analysis. Ambio 34(6):456–461. https://doi.org/10.1579/0044-7447-34.6.456
Clymo RS, Bryant CL (2008) Diffusion and mass flow of dissolved carbon dioxide, methane, and dissolved organic carbon in a 7-m deep raised peat bog. Geochimica Et Cosmochimica Acta 72(8):2048–2066. https://doi.org/10.1016/j.gca.2008.01.032
Conrad R (1999) Contribution of hydrogen to methane production and control of hydrogen concentrations in methanogenic soils and sediments. FEMS Microbiology Ecology 28(3):193–202. https://doi.org/10.1111/j.1574-6941.1999.tb00575.x
Conrad R, Klose M, Claus P (2000) Phosphate inhibits acetotrophic methanogenesis on rice roots. Applied and Environmental Microbiology 66(2):828–831. https://doi.org/10.1128/AEM.66.2.828-831.2000
Cooper MD, Evans CD, Zielinski P, Levy PE, Gray A, Peacock M, Norris D, Fenner N, Freeman C (2014) Infilled ditches are hotspots of landscape methane flux following peatland re-wetting. Ecosystems 17(7):1227–1241. https://doi.org/10.1007/s10021-014-9791-3
Couwenberg J (2009) Methane Emissions from Peat Soils (Organic Soils, Histosols): Facts, MRV-Ability, Emission Factors. Bonn: Greifswald University Wetlands International. http://www.imcg.net/media/download_gallery/climate/couwenberg_2009b.pdf. Accessed 17 March 2020
Dedysh SN (2009) Exploring methanotroph diversity in acidic northern wetlands: Molecular and cultivation-based studies. Microbiology 78(6):655–669. https://doi.org/10.1134/S0026261709060010
Dedysh SN, Berestovskaya YY, Vasylieva LV, Belova SE, Khmelenina VN, Suzina NE, Trotsenko YA, Liesack W, and Zavarzin GA (2004) Methylocella tundrae sp. nov., a novel methanotrophic bacterium from acidic tundra peatlands. International Journal of Systematic and Evolutionary Microbiology 54(1):151–156. https://doi.org/10.1099/ijs.0.02805-0
Dedysh S, Knief C, Dunfield P (2005) Methylocella species are facultatively methanotrophic. Journal of Bacteriology 187(13):4665–4670. https://doi.org/10.1128/JB.187.13.4665-4670.2005
Dettling MD, Yavitt JB, Cadillo-Quiroz H, Sun C, Zinder SH (2007) Soil-Methanogen Interactions in Two Peatlands (Bog, Fen) in Central New York State. Geomicrobiology Journal 24(3–4):247–259. https://doi.org/10.1080/01490450701456651
Environment Canada (2023) National inventory report 1990–2021: greenhouse gas sources and sinks in Canada. Government of Canada. https://publications.gc.ca/collections/collection_2023/eccc/En81-4-2021-1-eng.pdf. Accessed 31 May 2023
Esson KC, Lin X, Kumaresan D, Chanton JP, Murrell JC, Kostka JE (2016) Alpha- and gammaproteobacterial methanotrophs codominate the active methane-oxidizing communities in an acidic boreal peat bog. Applied and Environmental Microbiology 82(8):2363–2371. https://doi.org/10.1128/AEM.03640-15
Estop-Aragonés C, Knorr K, Blodau C (2013) Belowground in situ redox dynamics and methanogenesis recovery in a degraded fen during dry-wet cycles and flooding. Biogeosciences 10(1):421–436. https://doi.org/10.5194/bg-10-421-2013
Etto R, Cruz L, Jesus E, Galvão C, Galvão F, Souza E, Pedrosa FO, Steffens M (2012) Prokaryotic communities of acidic peatlands from the southern Brazilian Atlantic forest. Brazilian Journal of Microbiology 43(2):661–674. https://doi.org/10.1590/S1517-83822012000200031
Faith DP (1992) Conservation evaluation and phylogenetic diversity. Biological Conservation 61(1):1–10. https://doi.org/10.1016/0006-3207(92)91201-3
Frolking S, Roulet N, Fuglestvedt J (2006). How northern peatlands influence the Earth's radiative budget: Sustained methane emission versus sustained carbon sequestration. Journal of Geophysical Research: Biogeosciences 111(G1). https://doi.org/10.1029/2005JG000091
Galand PE, Fritze H, Conrad R, Yrjala K (2005) Pathways for methanogenesis and diversity of methanogenic archaea in three boreal peatland ecosystems. Applied and Environmental Microbiology 71(4):2195–2198. https://doi.org/10.1128/AEM.71.4.2195-2198.2005
Galand P, Saarnio S, Fritze H, Yrjala K (2002) Depth related diversity of methanogen Archaea in Finnish oligotrophic fen. FEMS Microbiology Ecology 42(3):441–449. https://doi.org/10.1111/j.1574-6941.2002.tb01033.x
Gao C, Sander M, Agethen S, Knorr K (2019) Electron accepting capacity of dissolved and particulate organic matter control CO2 and CH4 formation in peat soils. Geochimica Et Cosmochimica Acta 245:266–277. https://doi.org/10.1016/j.gca.2018.11.004
Gao D, Sheng R, Whiteley AS, Moreira-Grez B, Qin H, Zhang W, Zhan Y, Wei W (2020) Effect of phosphorus amendments on rice rhizospheric methanogens and methanotrophs in a phosphorus deficient soil. Geoderma 368(114312). https://doi.org/10.1016/j.geoderma.2020.114312.
Glatzel S, Basiliko N, Moore T (2004) Carbon dioxide and methane production potentials of peats from natural, harvested, and restored sites, eastern Quebec, Canada. Wetlands 24(2):261–267. https://doi.org/10.1672/0277-5212(2004)024[0261:CDAMPP]2.0.CO;2
Godin A, McLaughlin JW, Webster KL, Packalen M, Basiliko N (2012) Methane and methanogen community dynamics across a boreal peatland nutrient gradient. Soil Biology and Biochemistry 48:96–105. https://doi.org/10.1016/j.soilbio.2012.01.018
González E, Rochefort L (2014) Drivers of success in 53 cutover bogs restored by a moss layer transfer technique. Ecological Engineering 68:279–290. https://doi.org/10.1016/j.ecoleng.2014.03.051
González E, Rochefort L, Boudreau S, Hugron S, Poulin M (2013) Can indicator species predict restoration outcomes early in the monitoring process? A case study with peatlands. Ecological Indicators 32:232–238. https://doi.org/10.1016/j.ecolind.2013.03.019
Gorham E (1991) Northern Peatlands: Role in the Carbon Cycle and Probable Responses to Climatic Warming. Ecological Applications 1:182–195. https://doi.org/10.2307/1941811
Government of Canada (2013) Agriculture and Agri-Food Canada. Von Post. http://sis.agr.gc.ca/cansis/nsdb/soil/v2/slt/vonpost.html. Accessed 17 March 2020
Grman E, Bassett T, Brudvig LA (2013) EDITOR’S CHOICE: Confronting contingency in restoration: Management and site history determine outcomes of assembling prairies, but site characteristics and landscape context have little effect. Journal of Applied Ecology 50(5):1234–1243. https://doi.org/10.1111/1365-2664.12135
Gupta V, Smemo KA, Yavitt JB, Fowle D, Branfireun B, Basiliko N (2013) Stable isotopes reveal widespread anaerobic methane oxidation across latitude and peatland type. Environmental Science & Technology 47(15):8273–8279. https://doi.org/10.1021/es400484t
Hao X, Ball B, Culley J, Carter M, Parkin G (2008) Soil density and porosity. In: CRC Press (ed) Soil Sampling and Methods of Analysis, 2nd edn. Boca Raton, FL, pp 743–760
Hausmann B, Knorr K, Schreck K, Tringe SG, del Rio TG, Loy A, Pester M (2016) Consortia of low-abundance bacteria drive sulfate reduction-dependent degradation of fermentation products in peat soil microcosms. The ISME Journal 10(10):2365–2375. https://doi.org/10.1038/ismej.2016.42
Ho A, Kerckhof F, Luke C, Reim A, Krause S, Boon N, Bodelier PL (2013) Conceptualizing functional traits and ecological characteristics of methane-oxidizing bacteria as life strategies. Environmental Microbiology Reports 5(3):335–345. https://doi.org/10.1111/j.1758-2229.2012.00370.x
Hodgkins SB, Tfaily MM, McCalley CK, Logan TA, Crill PM, Saleska SR, Rich IR, Chanton JP (2014) Changes in peat chemistry associated with permafrost thaw increase greenhouse gas production. Proceedings of the National Academy of Sciences of the United States of America 111(16):5819–5824. https://doi.org/10.1073/pnas.1314641111
Høj L, Olsen RA, Torsvik VL (2008) Effects of temperature on the diversity and community structure of known methanogenic groups and other Archaea in high Arctic peat. Isme Journal 2(1):37–48. https://doi.org/10.1038/ismej.2007.84
Hope R (2013) Rmisc: Ryan Miscellaneous. R package version 1.5. https://CRAN.R-project.org/package=Rmisc. Accessed 17 March 2020
Horn M, Matthies C, Kusel K, Schramm A, Drake H (2003) Hydrogenotrophic methanogenesis by moderately acid-tolerant methanogens of a methane-emitting acidic peat. Applied and Environmental Microbiology 69(1):74–83. https://doi.org/10.1128/AEM.69.1.74-83.2003
Hu BL, Shen LD, Lian X, Zhu Q, Liu S, Huang Q, He ZF, Geng S, Cheng SQ, Lou LP, Xu XY, Zheng P, He YF (2014) Evidence for nitrite-dependent anaerobic methane oxidation as a previously overlooked microbial methane sink in wetlands. Proceedings of the National Academy of Sciences of the United States of America 111(12):4495–4500. https://doi.org/10.1073/pnas.1318393111
Hunger S, Schmidt O, Gößner AS, Drake HL (2016) Formate-derived H2, a driver of hydrogenotrophic processes in the root-zone of a methane-emitting fen. Environmental Microbiology 18(9):3106–3119. https://doi.org/10.1111/1462-2920.13301
Hunger S, Schmidt O, Hilgarth M, Horn MA, Kolb S, Conrad R, Drake HL (2011) Competing formate- and carbon dioxide-utilizing prokaryotes in an anoxic methane-emitting fen soil. Applied and Environmental Microbiology 77(11):3773–3785. https://doi.org/10.1128/AEM.00282-11
Jaatinen K, Fritze H, Laine J, Laiho R (2007) Effects of short-and long-term water-level drawdown on the populations and activity of aerobic decomposers in a boreal peatland. Global Change Biology 13(2):491–510. https://doi.org/10.1111/j.1365-2486.2006.01312.x
Jeffrey LC, Maher DT, Johnston SG, Maguire K, Steven AD, Tait DR (2019) Rhizosphere to the atmosphere: contrasting methane pathways, fluxes, and geochemical drivers across the terrestrial–aquatic wetland boundary. Biogeosciences 16(8):1799–1815. https://doi.org/10.5194/bg-16-1799-2019
Juottonen H (2020) Disentangling the effects of methanogen community and environment on peatland greenhouse gas production by a reciprocal transplant experiment. Functional Ecology 34:1268–1279. https://doi.org/10.1111/1365-2435.13536
Juottonen H, Galand PE, Tuittila ES, Laine J, Fritze H, Yrjälä K (2005) Methanogen communities and Bacteria along an ecohydrological gradient in a northern raised bog complex. Environmental Microbiology 7(10):1547–1557. https://doi.org/10.1111/j.1462-2920.2005.00838.x
Juottonen H, Hynninen A, Nieminen M, Tuomivirta TT, Tuittila ES, Nousiainen H, Kell DK, Yrjälä K, Tervahauta A, Fritze H (2012) Methane-cycling microbial communities and methane emission in natural and restored peatlands. Applied and Environmental Microbiology 78(17):6386–6389. https://doi.org/10.1128/AEM.00261-12
Kassambara A (2020) ggpubr: ‘ggplot2’ Based publication ready plots. R package version 0.4.0, https://CRAN.R-project.org/package=ggpubr. Accessed 15 Sept 2020
Knorr K, Lischeid G, Blodau C (2009) Dynamics of redox processes in a minerotrophic fen exposed to a water table manipulation. Geoderma 153(3–4):379–392. https://doi.org/10.1016/j.geoderma.2009.08.023
Köppen W, Volken E, Brönnimann S (2011) The thermal zones of the earth according to the duration of hot, moderate and cold periods and to the impact of heat on the organic world (translated from: Die wärmezonen der erde, nach der dauer der heissen, gemässigten und kalten zeit und nach der wirkung der wärme auf die organische welt betrachtet, Meteorol Z 1884, 1, 215–226). Meteorologische Zeitschrift 20(3):351–360. https://doi.org/10.1127/0941-2948/2011/105
Kreyling J, Tanneberger F, Jansen F et al (2021) Rewetting does not return drained fen peatlands to their old selves. Nature Communication 12, Art. 5693. https://doi.org/10.1038/s41467-021-25619-y
Küsel K, Blöthe M, Schulz D, Reiche M, Drake H (2008) Microbial reduction of iron and porewater biogeochemistry in acidic peatlands. Biogeosciences Discussions 5(3):2165–2196. https://doi.org/10.5194/bg-5-1537-2008
Laughlin DC, Strahan RT, Moore MM, Fulé PZ, Huffman DW, Covington WW (2017) The hierarchy of predictability in ecological restoration: are vegetation structure and functional diversity more predictable than community composition? J Appl Ecol 54:1058–1069. https://doi.org/10.1111/1365-2664.12935
Lin X, Green S, Tfaily MM, Prakash O, Konstantinidi KT, Corbett JE, Chanton JP, Cooper WT, Kostka JE (2012) Microbial community structure and activity linked to contrasting biogeochemical gradients in bog and fen environments of the Glacial Lake Agassiz Peatland. Applied and Environmental Microbiology 78(19):7023–7031. https://doi.org/10.1128/AEM.01750-12
Lin X, Tfaily MM, Steinweg JM, Chanton P, Esson K, Yang ZK, Chanton JP, Cooper W, Schadt CW, Kostka JE (2014) Microbial community stratification linked to utilization of carbohydrates and phosphorus limitation in a boreal peatland at Marcell experimental forest, Minnesota, USA. Applied and Environmental Microbiology 80(11):3518–3530. https://doi.org/10.1128/AEM.00205-14
Lovley DR, Coates JD, Blunt-Harris EL, Phillips EJ, Woodward JC (1996) Humic substances as electron acceptors for microbial respiration. Nature 382:445–448. https://doi.org/10.1038/382445a0
Lovley DR, Phillips EJ (1986) Organic matter mineralization with reduction of ferric iron in anaerobic sediments. Applied and Environmental Microbiology 51(4):683–689. https://doi.org/10.1128/aem.51.4.683-689.1986
Lozupone CA, Hamady M, Kelley ST, Knight R (2007) Quantitative and qualitative beta diversity measures lead to different insights into factors that structure microbial communities. Applied and Environmental Microbiology 73(5):1576–1585. https://doi.org/10.1128/AEM.01996-06
Lozupone C, Knight R (2005) UniFrac: A new phylogenetic method for comparing microbial communities. Applied and Environmental Microbiology 71(12):8228–8235. https://doi.org/10.1128/AEM.71.12.8228-8235.2005
Lucchese M, Waddington J, Poulin M, Pouliot R, Rochefort L, Strack M (2010) Organic matter accumulation in a restored peatland: Evaluating restoration success. Ecological Engineering 36(4):482–488. https://doi.org/10.1016/j.ecoleng.2009.11.017
Matheson CD, Gurney C, Esau N, Lehto R (2010) Assessing PCR inhibition from humic substances. The Open Enzyme Inhibition Journal 3(1):38–45. https://doi.org/10.2174/1874940201003010038
Markelova E, Parsons CT, Couture R, Smeaton CM, Madé B, Charlet L, Van Cappellen P (2018) Deconstructing the redox cascade: What role do microbial exudates (flavins) play? Environmental Chemistry 14(8):515–524. https://doi.org/10.1071/EN17158
Martí M, Juottonen H, Robroek BJM, Yrjala K, Danielsson A, Lindgren P, Svensson BH (2015) Nitrogen and methanogen community composition within and among three Sphagnum dominated peatlands in Scandinavia. Soil Biology and Biochemistry 81:204–211. https://doi.org/10.1016/j.soilbio.2014.11.016
McDonald JH (2014) Handbook of Biological Statistics, 3rd edn. Sparky House Publishing, Baltimore, Maryland
McMurdie PJ, Holmes S (2013) Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PloS One 8(4):1–11. Art. e61217. https://doi.org/10.1371/journal.pone.0061217
Metje M, Frenzel P (2005) Effect of temperature on anaerobic ethanol oxidation and methanogenesis in acidic peat from a northern wetland. Applied and Environmental Microbiology 71(12):8191–8200. https://doi.org/10.1128/AEM.71.12.8191-8200.2005
Miller KE, Lai C, Dahlgren RA, Lipson DA (2019) Anaerobic methane oxidation in high-Arctic Alaskan peatlands as a significant control on net CH4 fluxes. Soil Systems 3(1):1–21. Art. 7. https://doi.org/10.3390/soilsystems3010007
Min H, Zinder S (1990). Isolation and characterization of a thermophilic sulfate-reducing bacterium Desulfotomaculum thermoacetoxidans sp. nov. Archives of Microbiology 153(4):399–404. https://doi.org/10.1007/BF00249012
Narrowe AB, Angle JC, Daly RA, Stefanik KC, Wrighton KC, Miller CS (2017) High-resolution sequencing reveals unexplored archaeal diversity in freshwater wetland soils. Environmental Microbiology 19(6):2192–2209. https://doi.org/10.1111/1462-2920.13703
Nugent KA, Strachan IB, Strack M, Roulet NT, Rochefort L (2018) Multi-year net ecosystem carbon balance of a restored peatland reveals a return to carbon sink. Global Change Biology 24(12):5751–5768. https://doi.org/10.1111/gcb.14449
Oksanen J (2012) Constrained ordination: tutorial with R and vegan. http://cc.oulu.fi/~jarioksa/opetus/metodi/sessio2.pdf. Accessed 15 July 2019
Oksanen J, Guillaume Blanchet F, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagnervegan H (2019) Vegan: Community Ecology Package, R package version 2.5–5, https://CRAN.R-project.org/package=vegan. Accessed 1 June 2019
Pallud C, Kausch M, Fendorf S, Meile C (2010) Spatial patterns and modeling of reductive ferrihydrite transformation observed in artificial soil aggregates. Environmental Science & Technology 44(1):74–79. https://doi.org/10.1021/es901736t
Pan X, Angelidaki I, Alvarado-Morales M, Liu H, Liu Y, Huang X, Zhu G (2016) Methane production from formate, acetate and H2/CO2; focusing on kinetics and microbial characterization. Bioresource Technology 218:796–806. https://doi.org/10.1016/j.biortech.2016.07.032
Paulo P, Dos Santos AB, Ide C, Lettinga G (2005) Phosphate inhibition on thermophilic acetoclastic methanogens: A warning. Water Science and Technology 52(1–2):331–336. https://doi.org/10.2166/wst.2005.0535
Popp TJ, Chanton JP, Whiting GJ, Grant N (2000) Evaluation of methane oxidation in the rhizosphere of a Carex dominated fen in northcentral Alberta, Canada. Biogeochemistry 51(3):259–281. https://doi.org/10.1023/A:1006452609284
Poulin M, Andersen R, Rochefort L (2013) A new approach for tracking vegetation change after restoration: A case study with peatlands. Restoration Ecology 21(3):363–371. https://doi.org/10.1111/j.1526-100X.2012.00889.x
Pouliot R, Hugron S, Rochefort L, Godbout S, Palacios JH, Groeneveld E, Jarry I (2015) Manure derived biochar can successfully replace phosphate rock amendment in peatland restoration. J Environ Manag 157:118–126. https://doi.org/10.1016/j.jenvman.2015.04.019
Prietzel J, Thieme J, Salomé M (2010) Assessment of sulfur and iron speciation in a soil aggregate by combined S and Ee micro-XANES: Microspatial patterns and relationships. Journal of Synchrotron Radiation 17(2):166–172. https://doi.org/10.1107/S0909049509049917
Putkinen A, Tuittila E, Siljanen HM, Bodrossy L, Fritze H (2018) Recovery of methane turnover and the associated microbial communities in restored cutover peatlands is strongly linked with increasing sphagnum abundance. Soil Biology and Biochemistry 116:110–119. https://doi.org/10.1016/j.soilbio.2017.10.005
Quinty F, Rochefort L (2003) Peatland Restoration Guide. Québec: Canadian Sphagnum Peat Moss Association and New Brunswick Department of Natural Resources and Energy. https://www.gret-perg.ulaval.ca/fileadmin/Fichiers/centre_recherche/Peatland_Restoration_guide_2ndEd.pdf. Accessed 4 January 2023
Raghoebarsing AA, Pol A, van de Pas-Schoonen KT, Smolders AJ, Ettwig KF, Rijpstra WIC, Schouten S, Sinninghe JS, Op den Camp HJM, Jetten MS, Strous M (2006) A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 440:918–921. https://doi.org/10.1038/nature04617
Reumer M, Harnisz M, Lee HJ, Reim A, Grunert O, Putkinen A, Fritze H, Bodelier PLE, Ho A (2018) Impact of peat mining and restoration on methane turnover potential and methane-cycling microorganisms in a northern bog. Applied and Environmental Microbiology 84(3). Art. e02218–17. https://doi.org/10.1128/AEM.02218-17
Rezanezhad F, Kleimeier C, Milojevic T, Liu H, Weber TKD, Van Cappellen P, Lennartz B (2017) The role of pore structure on nitrate reduction in peat soil: A physical characterization of pore distribution and solute transport. Wetlands 37:951–960. https://doi.org/10.1007/s13157-017-0930-4
Robroek BJ, Jassey VE, Kox MA, Berendsen RL, Mills RT, Cécillon L, Puissant J, Meima-Franke M, Bakker PAHM, Bodelier PLE (2015) Peatland vascular plant functional types affect methane dynamics by altering microbial community structure. Journal of Ecology 103(4):925–934. https://doi.org/10.1111/1365-2745.12413
Rochefort L, Quinty F, Campeau S, Johnson K, Malterer T (2003) North American approach to the restoration of Sphagnum dominated peatlands. Wetlands Ecology and Management 11:3–20. https://doi.org/10.1023/A:1022011027946
Roslev P, King GM (1996) Regulation of methane oxidation in a freshwater wetland by water table changes and anoxia. FEMS Microbiology Ecology 19(2):105–115. https://doi.org/10.1111/j.1574-6941.1996.tb00203.x
Ruiz-Jaen MC, Aide T Mitchell (2005) Restoration success: How is it being measured? Restoration Ecology 13(3):569–577. https://doi.org/10.1111/j.1526-100X.2005.00072.x
Rydgren K, Auestad I, Halvorsen R, Hamre LN, Jongejans E, Töpper JP, Sulavik J (2020) Assessing restoration success by predicting time to recovery—But by which metric? Journal of Applied Ecology 57(2):390–401. https://doi.org/10.1111/1365-2664.13526
Rydgren K, Halvorsen R, Töpper JP, Auestad I, Hamre LN, Jongejans E, Sulavik J (2019) Advancing restoration ecology: A new approach to predict time to recovery. Journal of Applied Ecology 56(1):225–234. https://doi.org/10.1111/1365-2664.13254
Saunois M, Stavert AR, Poulter B et al (2020) The global methane budget 2000–2017. Earth System Science Data 12(3):1561–1623. https://doi.org/10.5194/essd-12-1561-2020
Segarra K, Schubotz F, Samarkin V, Yoshinaga M, Hinrichs K, Joye S (2015) High rates of anaerobic methane oxidation in freshwater wetlands reduce potential atmospheric methane emissions. Nature Communications 6(1):1–8. https://doi.org/10.1038/ncomms8477
Segers R (1998) Methane production and methane consumption: a review of processes underlying wetland methane fluxes. Biogeochemistry 41(1):23–51. https://doi.org/10.1023/A:1005929032764
Schmidt O, Hink L, Horn MA, Drake HL (2016) Peat: Home to novel syntrophic species that feed acetate-and hydrogen-scavenging methanogens. The ISME Journal 10(8):1954–1966. https://doi.org/10.1038/ismej.2015.256
Shannon CE, Weaver W (1949) The Mathematical Theory of Communication. The University of Illinois Press, Urbana, pp 1–117
Simpson GL (2019) ggvegan: ‘ggplot2’ Plots for the ‘vegan’ Package. R package version 0.1–0. https://gavinsimpson.github.io/ggvegan/. Accessed 17 Mar 2020
Slowikowski K (2019) Ggrepel: automatically position non-overlapping text labels with ‘ggplot2’. R package version 0.8.1. https://CRAN.R-project.org/package=ggrepel. Accessed 15 Sept 2020
Smeaton CM, Van Cappellen P (2018) Gibbs energy dynamic yield method (GEDYM): Predicting microbial growth yields under energy-limiting conditions. Geochimica Et Cosmochimica Acta 241:1–16. https://doi.org/10.1016/j.gca.2018.08.023
Smemo KA, Yavitt JB (2007) Evidence for anaerobic CH4 oxidation in freshwater peatlands. Geomicrobiology Journal 24(7–8):583–597. https://doi.org/10.1080/01490450701672083
Smith GJ, Angle JC, Solden LM, Borton MA, Morin TH, Daly RA, Johnston MD, Stefanik KC, Wolfe R, Gil B, Wrighton KC (2018) Members of the genus Methylobacter are inferred to account for the majority of aerobic methane oxidation in oxic soils from a freshwater wetland. mBio 9(6). Art. e00815–18. https://doi.org/10.1128/mBio.00815-18
Söllinger A, Schwab C, Weinmaier T, Loy A, Tveit AT, Schleper C, Urich T (2015) Phylogenetic and genomic analysis of Methanomassiliicoccales in wetlands and animal intestinal tracts reveals clade-specific habitat preferences. FEMS Microbiology Ecology. 92(1). Art. fiv149. https://doi.org/10.1093/femsec/fiv149
Stookey LL (1970) Ferrozine - a new spectrophotometric reagent for iron. Analytical Chemistry 42(7):779–781. https://doi.org/10.1021/ac60289a016
Strack M, Cagampan J, Hassanpour FG, Keith AM, Nugent K, Rankin T, Robinson C, Strachan IB, Waddington JM, Xu B (2016) Controls on plot-scale growing season CO2 and CH4 fluxes in restored peatlands: Do they differ from unrestored and natural sites? Mires and Peat 17:1–18, Art. 05. https://doi.org/10.19189/MaP.2015.OMB.216
Strack M, Waller M, Waddington J (2006) Sedge succession and peatland methane dynamics: A potential feedback to climate change. Ecosystems 9(2):278–287. https://doi.org/10.1007/s10021-005-0070-1
Ström L, Mastepanov M, Christensen TR (2005) Species-specific effects of vascular plants on carbon turnover and methane emissions from wetlands. Biogeochemistry 75(1):65–82. https://doi.org/10.1007/s10533-004-6124-1
Sun CL, Brauer SL, Cadillo-Quiroz H, Zinder SH, Yavitt JB (2012). Seasonal changes in methanogenesis and methanogenic community in three peatlands, New York state. Frontiers in Microbiology 3:1–8. Art. 8. https://doi.org/10.3389/fmicb.2012.00081
Sundh I, Nilsson M, Granberg G, Svensson B (1994) Depth distribution of microbial production and oxidation of methane in northern boreal peatlands. Microbial Ecology 27(3):253–265. https://doi.org/10.1007/BF00182409
Sundh I, Mikkelä C, Nilsson M, Svensson BH (1995) Potential aerobic methane oxidation in a sphagnum-dominated peatland - controlling factors and relation to methane emission. Soil Biology and Biochemistry 27(6):829–837. https://doi.org/10.1016/0038-0717(94)00222-M
Sundh I, Nilsson M, Mikkela C, Granberg G, Svensson B (2000) Fluxes of methane and carbon dioxide in peat-mining areas in Sweden. Ambio 29(8):499–503. https://doi.org/10.1579/0044-7447-29.8.499
Tuittila E, Komulainen V, Vasander H, Nykanen H, Martikainen P, Laine J (2000a) Methane dynamics of a restored cut-away peatland. Global Change Biology 6(5):569–581. https://doi.org/10.1046/j.1365-2486.2000.00341.x
Tuittila E, Vasander H, Laine J (2000b) Impact of rewetting on the vegetation of a cut-away peatland. Applied Vegetation Science 3(2):205–212. https://doi.org/10.2307/1478999
Turetsky M, Wieder K, Halsey L, Vit D (2002) Current disturbance and the diminishing peatland carbon sink. Geophysical Research Letters 29(11), Art. 1526. https://doi.org/10.1029/2001GL014000
Vasander H, Tuittila E, Lode E, Lundin L, Ilomets M, Sallantaus T, Heikkilä R, Pitkänen M-L, Laine J (2003) Status and restoration of peatlands in northern Europe. Wetlands Ecology and Management 11(1–2):51–63. https://doi.org/10.1023/A:1022061622602
Viollier E, Inglett P, Hunter K, Roychoudhury A, Van Cappellen P (2000) The ferrozine method revisited: Fe (II)/Fe (III) determination in natural waters. Applied Geochemistry 15(6):785–790. https://doi.org/10.1016/S0883-2927(99)00097-9
Waddington JM, Day SM (2007) Methane emissions from a peatland following restoration. Journal of Geophysical Research G: Biogeosciences 112:1–11. Art. G03018. https://doi.org/10.1029/2007JG000400
Waddington J, Price J (2000) Effect of peatland drainage, harvesting, and restoration on atmospheric water and carbon exchange. Physical Geography 21(5):433–451. https://doi.org/10.1080/02723646.2000.10642719
Waldén E, Lindborg R (2016) Long term positive effect of grassland restoration on plant diversity-success or not? PloS One 11(5). Art. e0155836. https://doi.org/10.1371/journal.pone.0155836
Weedon JT, Kowalchuk GA, Aerts R, Freriks S, Röling WFM, van Bodegom PM (2017) Compositional Stability of the Bacterial Community in a Climate-Sensitive Sub-Arctic Peatland. Frontiers in Microbiology 8:1–11. Art. 317. https://doi.org/10.3389/fmicb.2017.00317
Wickham H (2016) ggplot2: Elegant Graphics for Data Analysis, Springer-Verlag, New York, ISBN 978–3–319–24277–4. https://ggplot2.tidyverse.org
Wickham H (2017) tidyverse: Easily Install and Load the 'Tidyverse'. R package version 1.2.1. https://CRAN.R-project.org/package=tidyverse. Accessed 15 Sept 2020
Wind-Mulder HL, Rochefort L, Vitt DH (1996) Water and peat chemistry comparisons of natural and post-harvested peatlands across Canada and their relevance to peatland restoration. Ecological Engineering 7(3):161–181. https://doi.org/10.1016/0925-8574(96)00004-3
Wind-Mulder HL, Vitt DH (2000) Comparisons of water and peat chemistries of a post-harvested and undisturbed peatland with relevance to restoration. Wetlands 20(4):616–628. https://doi.org/10.1672/0277-5212(2000)020[0616:COWAPC]2.0.CO;2
Xiang X, Wang R, Wang H, Gong L, Man B, Xu Y (2017) Distribution of Bathyarchaeota communities across different terrestrial settings and their potential ecological functions. Scientific Reports 7:1–11. Art. 45028. https://doi.org/10.1038/srep45028
Xu J, Morris PJ, Liu J, Holden J (2018) PEATMAP: Refining estimates of global peatland distribution based on a meta-analysis. Catena 160:134–140. https://doi.org/10.1016/j.catena.2017.09.010
Yavitt JB, Yashiro E, Cadillo-Quiroz H, Zinder SH (2012) Methanogen diversity and community composition in peatlands of the central to northern Appalachian mountain region, North America. Biogeochemistry 109(1–3):117–131. https://doi.org/10.1007/s10533-011-9644-5
Yrjälä K, Tuomivirta T, Juottonen H, Putkinen A, Lappi K, Tuittila E-S, Penttilä T, Minkkinen K, Laine J, Peltoniemi K, Peltoniemi K, Fritze H (2011) CH4 production and oxidation processes in a boreal fen ecosystem after long-term water table drawdown. Global Change Biology 17(3):1311–1320. https://doi.org/10.1111/j.1365-2486.2010.02290.x
Yu Z (2012) Northern peatland carbon stocks and dynamics: A review. Biogeosciences 9(10):4071–4085. https://doi.org/10.5194/bg-9-4071-2012
Yu Z, Loisel J, Brosseau DP, Beilman DW, Hunt SJ (2010) Global peatland dynamics since the last glacial maximum. Geophysical Research Letters 37(13):L13402. https://doi.org/10.1029/2010GL043584
Yu Z, Beilman DW, Frolking S, MacDonald GM, Roulet NT, Camill P, Charman DJ (2011) Peatlands and Their Role in the Global Carbon Cycle. Eos, Transactions of the American Geophysical Union 92(12):97–108. https://doi.org/10.1029/2011EO120001
Zalman C, Keller J, Tfaily M, Kolton M, Pfeifer-Meister L, Wilson R, Lin X, Chanton J, Kostka JE, Gill A, Finzi A, Hopple AM, Bohannan BJM, Bridgham SD (2018) Small differences in ombrotrophy control regional-scale variation in methane cycling among sphagnum-dominated peatlands. Biogeochemistry 139(2):155–177. https://doi.org/10.1007/s10533-018-0460-z
Zhao B, Zhuang Q (2023) Peatlands and their carbon dynamics in northern high latitudes from 1990 to 2300: a process-based biogeochemistry model analysis. Biogeosciences 20:251–270. https://doi.org/10.5194/bg-20-251-2023
Zhu B, van Dijk G, Fritz C, Smolders AJP, Pol A, Jetten MSM, Ettwig KF (2012) Anaerobic Oxidization of Methane in a Minerotrophic Peatland: Enrichment of Nitrite-Dependent Methane-Oxidizing Bacteria. Applied and Environmental Microbiology 78(24):8657–8665. https://doi.org/10.1128/AEM.02102-12
Zuñiga C, Morales M, Revah S (2013) Polyhydroxyalkanoates accumulation by Methylobacterium organophilum CZ-2 during methane degradation using citrate or propionate as cosubstrates. Bioresource Technology 129:686–689. https://doi.org/10.1016/j.biortech.2012.11.120
Acknowledgements
We thank Angus Hilts, Emilie Spasov, and Alexandra Sauk for brainstorming and advising on molecular data analysis, Dr. Martin Brummell and Erin McDonald for field work assistance, Joanna Chudyk and Rebecca Co from the Hug Lab UW, Marianne Vandergriendt and Amanda Nidercorn from the Ecohydrology Research Group UW, Anne Grant and Beverly Raimbault from the Ecology Lab UW, and Mathew Coulas, Alexandra Engering, and Rhiannon Hodgson from the Wetland Soils & Greenhouse Gas Exchange Lab UW for assistance in the laboratory analyses. Dr. N. Basiliko, Dr. S. J. Davidson, and Dr. M. Macrae provided valuable feedback on the manuscript. Sungro Horticulture provided site access and logistical support. The symbols used in the feature image are courtesy of the Integration and Application Network, University of Maryland Center for Environmental Science (ian.umces.edu/symbols/).
Funding
This work was supported by Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grants [342020 to MS, 2016–03686 to LAH]; NSERC Collaborative Research and Development grant with support from the Canadian Sphagnum Peat Moss Association and its members [437463 to MS]; the Canada Excellence Research Chair in Ecohydrology (grant number not applicable); Canada First Excellence Research Fund (CFREF): Global Water Futures (GWF) program (grant number not applicable); and Joint Genome Institute: Small Scale sequencing grant [CSP 2812, LAH]. LAH holds a Tier II Canada Research Chair in Environmental Microbiology and MS holds a Tier II Canada Research Chair in Ecosystem and Climate.
Author information
Authors and Affiliations
Contributions
Aneta Bieniada: Conceptualization, Formal Analysis, Investigation, Data Curation, Writing-Original Draft, Visualization, Project Administration. Laura A. Hug: Supervision, Resources, Funding Acquisition, Writing – Review & Editing. Christopher T. Parsons: Supervision, Resources, Writing – Review & Editing. Maria Strack: Supervision, Resources, Funding Acquisition, Writing – Review & Editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bieniada, A., Hug, L.A., Parsons, C.T. et al. Methane Cycling Microbial Community Characteristics: Comparing Natural, Actively Extracted, Restored and Unrestored Boreal Peatlands. Wetlands 43, 83 (2023). https://doi.org/10.1007/s13157-023-01726-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01726-y