Abstract
Wetland impoundments are constructed for recreational and conservational purposes. Here, the water level can be carefully controlled, producing ideal conditions for aquatic plant growth to support migratory birds or other management goals. These wetlands also perform a critical function of nutrient assimilation, with the capability to protect downstream waters from eutrophication. Understanding how the structural characteristics of wetlands are related to this functional capacity within shallow impoundments will help inform management practices to improve overall wetland function. We characterized 18 waterfowl impoundments surrounding the Great Salt Lake, Utah, USA. Wetland assimilation of nitrogen (N) and phosphorus (P) was estimated at each wetland by controlled nutrient addition within mesocosms. In addition, wetland condition was assessed using a multimetric index (MMI), an indicator of the biological quality of the wetlands. We found that N assimilation was inversely correlated with water depth and positively correlated with soil % clay and total iron. Phosphorus assimilation was related to dissolved oxygen, aluminum, and N and P concentrations within the water column and soil. Nutrient assimilation did not differ among wetlands rated as poor, fair, and good by the MMI.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Great Salt Lake (GSL) is the largest terminal saline lake in the Western Hemisphere and the largest inland body of water along the Pacific flyway, a major pathway for migratory birds in the Americas (Cohenour and Thompson 1966; Kijowski et al. 2020). In addition, the lake is surrounded by approximately 145,000 ha of wetlands, many of which are waterfowl impoundments that are hydrologically managed to support submerged aquatic vegetation, an important food source for the 4–6 million waterfowl and other migratory birds that frequent the region (Downard and Endter-Wada 2013; Downard et al. 2014). Because of their ecological importance, efforts are underway by the Utah Division of Water Quality (UDWQ) to protect the integrity of these wetlands.
Wetlands perform many vital ecosystem functions, including storage of surface water, streamflow maintenance, sediment retention, fish and wildlife habitat, and nutrient transformation and/or retention (Tiner 2005). Wetland transformation and/or retention of nitrogen (N) and phosphorus (P) is essential because it protects downstream waters from eutrophication. Baron and others (2013) found that 64% of reactive N removal in freshwater ecosystems of the United States occurs in wetlands. In addition, research conducted over 12 years in tile-drained agricultural lands shows that wetlands removed 28 to 52% of mass nitrate (NO3-) and 71 to 85% of mass orthophosphate loadings (Lemke et al. 2022). Wetland N removal occurs through denitrification, biological uptake by plants and microorganisms, and sedimentation of organic particulates (Saunders and Kalff 2001). The primary removal pathways for P are slightly different. Phosphorus remains in the phosphate (PO43-) form through assimilation and decomposition, and the primary phosphorus removal processes in wetlands are chemical sorption, precipitation, biological uptake, and soil accretion (Vymazal 2007).
Despite their importance in nutrient removal, wetlands are understudied at the regional, landscape, and global scales of nutrient cycling, partly because nutrient removal rates for processes like denitrification are difficult to quantify due to high spatial and temporal variation (Groffman et al. 2006). In addition, instead of direct measurements, nutrient removal rates in wetlands are frequently estimated from the inputs and outputs of total N and P (Cheng and Basu 2017). As a result, the wetland characteristics that influence nutrient retention are not clearly defined. Crumpton and others (2020) found that mass NO3- removal is a function of hydraulic loading rate, temperature, and NO3- concentration in restored corn belt wetlands. In waterfowl impoundments, seasonal water management and the soil physicochemical environment have been shown to significantly affect N cycling rates, with bird waste leading to high ammonium (NH4+) concentrations, and environmental conditions causing inhibited nitrification (Hinckley et al. 2020). In addition, managed waterfowl impoundments can also act as a source of nutrients; during seasonal drawdown, impoundments have been shown to export similar loads of N as fertilized agricultural fields (Winton et al. 2016). Seasonal water management can also decrease nitrogen removal in impounded wetlands, and there are currently no best practices for nitrogen removal in managed wetland systems (Jones et al. 2022).
Mesocosms may provide an effective method to directly measure ecosystem functions like nutrient assimilation in waterfowl impoundments. Mesocosms are intermediately-sized environmental enclosures commonly used to research aquatic ecosystems (Odum 1984). Additionally, mesocosms make it possible to conduct controlled manipulative experiments in the natural environment. For example, Wang and others (2006) used experimental mesocosms to study the effect of nutrient additions of phytoplankton growth in addition to estimating nutrient uptake kinetic parameters. Tavşanoğlu and others (2017) established mesocosms within a shallow lake to show how nutrient and water level variability effects zooplankton community taxonomic structure. In our study, we used wetland mesocosms to measure nutrient assimilation kinetics.
Our study goal was to determine which wetland characteristics are most related to nitrogen and phosphorus assimilation in wetland impoundments. Specifically, our objective was to relate wetland function (nutrient assimilation) to measures of wetland structure across environmental gradients. Our second objective was to evaluate whether wetland biological condition (e.g., good, fair, poor) predicts nutrient assimilation functioning. We hypothesized that sites identified as poor may have impaired nutrient retention function. Finally, understanding the relationship between wetland condition and function can help to inform appropriate water quality standards that should be kept to maintain wetland quality.
Methods
We collaborated with the Utah Division of Water Quality (UDWQ) to select 18 waterfowl impoundments for the study. These sites were selected because they captured the broad range of environmental conditions we hypothesized to be important drivers of wetland sensitivity to cultural eutrophication. In addition, the sites were previously assessed through the UDWQ in 2012, so comparisons could be made in their quality over time. The impoundments were studied during the summer, with water depths ranging from approximately 15–70 cm (Table 1). Most sites were from Bear River, Ogden, and Farmington Bay wetland complexes surrounding the Great Salt Lake, Utah (Fig. 1). These engineered impoundments are fed by surface flows from the Wasatch Mountains, through the Bear, Weber, and Jordan Rivers. They are diked, and the water level is carefully controlled to promote the growth of desirable submerged aquatic plants like Stuckenia spp. to support waterfowl (Downard et al. 2014).
At each site, a mesocosm was installed to provide an environmental enclosure where nutrient concentrations could be manipulated within wetland ponds. The mesocosms used in this study were made of clear plastic (polycarbonate resin thermoplastic). They were cylindrical, with a diameter of 1.94 m and a height of 1 m. They are open to the soil at the bottom, allowing interactions between the soil and water. At each site, the mesocosm was installed approximately 30 cm deep in the soil, enclosing soil and water from the impoundment. The experiments were performed between June and August (working with one site at a time). The mesocosm was deployed at each location two days before sampling and assessment to allow time for the soil and water column to equilibrate from disturbance.
To estimate nutrient assimilative capacity, we performed an experimental pulse release of nutrients (Tank et al., 2008). A single dose of nutrients was released concurrently as nitrate (NaNO3) and phosphate (KH2PO4) at 0.1 mg N, P L-1 doses and mixed into the water column. Water samples were collected from each mesocosm before the nutrient addition and at 15, 30, 45, 60, 75, 90, 105, 120, 150, 180, 210, and 240 min after addition to capture the decline in water column nutrients. The samples were filtered in the field (Whatman glass fiber filter size GF/F, Maidstone, UK) and stored on ice until arrival at the lab, where they were frozen until analysis for NO3-N and PO4-P concentrations.
Additionally, wetland condition was assessed through sampling across a 100 m transect, with the first point of the transect being next to the mesocosm. Measurements were taken at 0, 25, 50, 75, and 100 m along the transect. We measured pH, specific conductance, and temperature on site using a handheld meter (Pro1030, YSI Inc., Yellow Springs, OH, USA). Dissolved oxygen was measured on-site using an optical meter (ProODO, YSI Inc., Yellow Springs, OH, USA). Water samples were taken to measure dissolved organic carbon (DOC), NO3-N, NH4-N, SRP, Total N and P, and dissolved elements. As mentioned above, the samples (except for total N and P) were immediately filtered, stored on ice, and frozen until analysis. Water sample analyses were conducted at the Utah State University (USU) Aquatic Biogeochemistry Laboratory and the Brigham Young University (BYU) Environmental Analytical Laboratory. DOC was quantified using catalytic oxidation (TOC/TN-L, Shimadzu Scientific, Kyoto, Kyoto Prefecture, Japan). Micro-segmented flow analysis was used to determine NO3-N, NH4-N, and orthophosphate-P (Detection limits 0.001, 0.030, and 0.005, respectively; Astoria Analyzer, Astoria-Pacific, Clackamas, OR). The concentration of NO3-N was determined using the cadmium reduction method (U.S. Environmental Protection Agency 1993b), and the concentration of NH4-N was determined by the phenol-sodium nitroprusside method (U.S. Environmental Protection Agency 1993a). Orthophosphate was measured using the molybdenum blue method (U.S. Environmental Protection Agency 1993c). Total N and P were measured by persulfate digestion (Ameel et al. 1993) and quantified using micro-segmented flow as described above (Astoria Analyzer, Astoria-Pacific, Clackamas, OR). Dissolved elements (Al, Ca, Fe) were determined by ICP-OES (iCAP 7400, Thermo Scientific, Waltham, MA, USA).
We sampled submerged aquatic vegetation (SAV), soil, chlorophyll a, algae (if present), zooplankton, and benthic macroinvertebrates at 0, 25, 50, 75, and 100 m across transects at each site. The percent cover of SAV and algae was recorded at each location. SAV samples were analyzed for total N, total P, and percent ash. Total N in SAV and total C and N in soil samples were determined by combustion (McGeehan & Naylor, 1988; TruSpec CN Determinator, LECO Instruments, St. Joseph, MI, USA). Total P in SAV and total P, Ca, Fe, and Al in soil samples was determined by nitric-hydrogen peroxide microwave digestion (Ethos EZ, Milestone, Shelton, CT, USA) followed by quantification with ICP-OES (iCAP 7400, Thermo Scientific, Waltham, MA, USA). Soil % organic matter was determined by dichromate oxidation (Walkley and Black 1934), and soil texture was analyzed by hydrometer (Gee and Or 2002). Chlorophyll a was extracted from filters using hot ethanol and analyzed using a fluorometer (Hauer and Lamberti 2006). Benthic macroinvertebrates were sampled with five x 1 m sweeps using a 500 μm D-net. Zooplankton was collected with five x 5 m tows (radial) using a Wisconsin net (153 μm mesh size). The BLM/USU National Aquatic Monitoring Center Bug Lab at Utah State University conducted the identification and enumeration of species.
We assessed wetland quality through a multimetric index (MMI) developed by the Utah Division of Water Quality (CH2M Hill 2009). While beneficial to prioritize management needs, assessment tools like MMIs are not comprehensive descriptors of wetland quality. For example, it is unknown whether assessments of wetland quality based on structural attributes predict wetland functions like nutrient removal. The MMI used in the study is based on three areas related to habitat quality for wildlife. According to this metric, a high-quality wetland would have extensive SAV, a low incidence of surface algal mats, and diverse macroinvertebrate communities. The specific characteristics used were percent SAV cover, percent algae cover, and the composition of benthic macroinvertebrate communities. The benthic macroinvertebrate composition includes Simpson’s Index (SI) of invertebrates and the relative abundance of invertebrates anticipated to be strongly correlated with SAV. The invertebrates strongly correlated with SAV include: Ephemeroptera (mayflies), Odonata (dragonflies and damselflies, excluding the genus Aeshna), Hesperocorixa spp., Ylodes sp., and Gyraulus (Gray 2009, 2012, 2013).
For each characteristic, the relative value across sites was calculated by dividing the mean (of five transect observations) obtained at the site by the geometric mean across all measured sites. Next, a metric was created by rescaling the data that ranged from 100 (representing high water quality) to 0 (representing poor water quality). For variables with values expected to decrease with decreasing water quality (SAV cover and macroinvertebrate diversity), the site value was divided by the maximum concentration across all sites and then multiplied by 100. For variables that increase with decreasing water quality (algae cover), the same process was followed, except the final value was subtracted from 100 so that lower scores indicate poorer water quality (CH2M Hill 2009). The metrics were then rescaled using the following equation so that the site with the best water quality received a score of 100.
Finally, all three MMI parameters (SAV cover, algae cover, macroinvertebrates) were averaged to obtain the overall MMI metric. Classes were assigned based on the relative quality of wetland impoundments found during an extensive, randomized impounded wetland assessment by the Utah Division of Water Quality in 2012 (UDWQ 2014). Sites with an MMI score greater than or equal to the 75th percentile were classified as ‘Good’ quality, below the 25th percentile were classified as ‘Poor ‘quality, and all others were classified as ‘Fair.’
Statistical Analysis
The decline in NO3-N and PO4-P over time within mesocosms was modeled as first-order decay, which has been shown to accurately describe nutrient removal in wetland mesocosms (Messer et al. 2017). We plotted the natural log concentration of NO3-N and PO4-P vs. time and determined the first-order rate constant (i.e., the nutrient removal rate constant) from the slope of the regression line. The rate constant was used as a measure of nutrient assimilation.
We tested the hypotheses of relationships between the measured biological, geochemical, and physical characteristics with NO3 and PO4 assimilation rate constants and the MMI. We used Spearman’s rank correlation coefficient since it is a nonparametric measure and is not sensitive to outliers (Zar 1972). We set alpha to 0.10 and conducted the analysis using IBM SPSS Statistics version 28.0 (IBM Corp 2021).
Finally, a Kruskal-Wallis test was performed to determine if there were differences among MMI classes (“Good,” “Fair,” “Poor”) and measured nutrient assimilation rate constants for NO3 and PO4. The test was performed in R using the base R function kruskal.test (R Core Team 2020).
Results
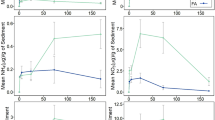
The impounded wetlands sampled represent a gradient in environmental characteristics. The surface areas ranged from 6.99 to 3209.85 ha, with depths from shallow (13.8 cm) to relatively deep ponds (73.8 cm; Table 1). The wetlands also ranged from freshwater to saline, with specific conductance values varying from 0.7 to 11.6 dS/m (Table 1). The ambient N and P concentrations measured within transects were generally high in all the measured impoundments, with 0.051 mg/L total water column P as the lowest measured and 0.175 mg/L as the median total P (Table 1). Total N in the water column ranged from 0.85 to 4.45 mg/L (Table 1). The NO3 assimilation rate constants ranged from 0.05 to 1.21 h-1 with a median value of 0.28 h-1. The PO4 assimilation rate constants were generally lower than NO3. They ranged from 0.005 to 0.95 h-1 with a median of 0.08 h-1. The plots showing nutrient decline over time can be viewed in Online Resource 1. The wetland sites were relatively evenly spread according to the determined MMI condition class with six “Good,” eight “Fair,” and four “Poor” (Table 2).
Through Spearman’s rank correlation coefficient, we found the following wetland characteristics to be related to the NO3 assimilation rate constant. Nitrate assimilation was strongly inversely correlated with water depth (Fig. 2; Table 3; ρ(18) = − 0.65, p < .01) and was positively associated with soil % clay (Table 3; ρ(18) = 0.55, p < .05) and total Fe (Table 3; ρ(18) = 0.41, p < .10).
The PO4 assimilation rate constant was positively correlated with % DO saturation in the water column (Table 3; ρ(18) = 0.41, p < .10) and dissolved Al (Table 3; ρ(18) = 0.44, p < .10). Phosphate assimilation was negatively correlated with many of the nutrient concentrations. For example, PO4 assimilation rate constant was inversely correlated with water column dissolved NO3-N (Table 3; ρ(18) = − 0.46, p < .10), soluble reactive P (Table 3; ρ(18) = − 0.61, p < .01), and total P (Table 3; ρ(18) = − 0.43, p < .10). It was also inversely correlated with soil total N (Table 3; ρ(18) = − 0.44, p < .10) and total P (Fig. 3; Table 3; ρ(18) = − 0.73, p < .01).
The MMI, an index calculated from macroinvertebrate diversity, aquatic plant cover, and algae cover, was positively associated with specific conductance (Table 3; ρ(18) = 0.51, p < .05). It was also significantly inversely associated with soil total Fe (Table 3; ρ(18) = − 0.59, p < .05), soil total P (Table 3; ρ(18) = − 0.42, p < .10), and water column total P (Table 3; ρ(18) = − 0.43, p < .10). Chlorophyll a was strongly, inversely correlated with % SAV cover (Table 3; ρ(18) = − 0.65, p < .01). In contrast to our hypothesis, NO3 assimilation did not differ by MMI condition class (Kruskal-Wallis H(2) = 1.26, p = .53; Fig. 4). Similarly, there were no demonstrated differences in PO4 assimilation over MMI class (Kruskal-Wallis H(2) = 0.81, p = .67; Fig. 5). Finally, most wetland impoundments (66%) had a higher MMI score in 2018 compared to the previous assessment by UDWQ in 2012 (Fig. 6).
Discussion
Our experimental goals were to (1) determine which wetland characteristics are associated with short-term NO3 and PO4 assimilation and (2) evaluate whether NO3 and PO4 assimilation differs among groups of sites determined to be good, fair, or poor in habitat quality for waterfowl. The nutrient assimilation rate constants we measured were comparable to others reported within natural wetlands. Cheng and Basu (2017) completed a meta-analysis with articles providing information on hydraulic residence time and input/output nutrient concentrations and mass loads to and from wetlands. They calculated an overall removal rate constant based on a continuously stirred tank reactor (CSTR) model (Vollenweider 1975; Spieles and Mitsch 1999; Cheng and Basu 2017). For natural wetlands, they reported 1.009 d-1 (0.042 h-1) for NO3 and 0.190 d-1 (0.008 h-1) for PO4. Our measured nutrient assimilation rate constants ranged from 0.05 to 1.21 h-1 for NO3 and 0.005 to 0.95 h-1 for PO4. Most of our measured values were higher than the removal rate constants reported in their study. However, the values reported by Cheng and Basu (2017) fall within the range or are close to the range we measured from wetland impoundments.
As wetlands are continuously loaded with P, the capacity for P assimilation decreases (Reddy et al. 1995; Richardson et al. 1996), and wetlands more heavily loaded with P may provide close to zero removal of P (Kadlec 1999). In agreement with these findings, we found that higher SRP, water column total P, and soil total P were related to lower PO4 assimilation rate constants. In addition, we found PO4 assimilation rate constants to be lower than NO3, similar to our findings in a mesocosm experimental manipulation study (Wood et al., in preparation). Similar results were found in Louisiana, where nutrient assimilation was highest for NO3, intermediate for NH4, and lowest for PO4 in a forested wetland (Brinson et al. 1984). We also found that PO4 assimilation was positively correlated with the concentration of dissolved Al in the water column. This finding is not surprising as Al is commonly used to precipitate P in wastewater treatment and remediation efforts (de-Bashan and Bashan 2004; Liu et al. 2009). We also found a positive association of PO4 assimilation with % DO saturation, likely due to more P uptake occurring when primary producers are active. Hourly in situ measurements of SRP in a large river in Florida showed a strong correlation to DO variation, suggesting photosynthetic organisms control the SRP concentration directly through uptake or indirectly through geochemical reactions (Cohen et al. 2013).
Factors commonly attributed as being influential to nutrient reduction in wetlands include oxygen concentration, redox, and waterlogging of the soil, as well as vegetation processes and hydraulic loading and retention time (Fisher and Acreman 2004). We found NO3 assimilation to be negatively correlated with water depth. Similar findings have been reported in stream isotope tracer studies (Botter et al. 2010; Ye et al. 2012). This relationship may be found because shallow waters are especially effective at NO3 removal through coupled nitrification-denitrification (Penton et al. 2013; Racchetti et al. 2016; Xia et al. 2017). Furthermore, the result is not surprising given that volumetric reaction rates are inversely linked to water depth (Tanner et al. 1999). It may also be related to higher soil surface-to-water ratio and increased interactions with soil. NO3 assimilation was also positively associated with % clay, and clay soils may have more anoxic microsites for denitrification. Kunickis and others (2010) studied the relationship between soil texture and redox potential within riparian buffers. They found that clay-textured soils provided lower redox values within the range for denitrification to occur (Kunickis et al. 2010).
Phosphate and NO3 assimilation rate constants were found to be correlated with abiotic variables, including depth, soil texture, and nutrient concentrations. However, they were not associated with biotic variables like % aquatic plant cover, chlorophyll a, or macroinvertebrate diversity. Differences in plant community composition have been shown to affect N removal (Weller et al. 2016); however, we only assessed the % cover of aquatic plants as a factor, and the aquatic plants found within these impoundments were predominantly Stuckenia pectinata. NO3 and PO4 assimilation were also not different among our calculated MMI condition classes, contrasting our hypothesis that high-quality wetlands in terms of waterfowl habitat would also have a high nutrient removal capacity. Our previous mesocosm manipulation experiment within wetland impoundments showed that removing aquatic plants did not change NO3 and PO4 assimilation rate constants (Wood et al., in preparation), so the lack of correlation between % aquatic plant cover and NO3 and PO4 assimilation is in line with this finding.
Stapanian and others (2016) found that an index of vegetation biological integrity was lower in emergent wetlands with high concentrations of plant available P in the soil. Similarly, the MMI used in this study, an index representing % aquatic plant cover, % algae cover, and macroinvertebrate diversity, declined with increasing soil and water total P, supporting a well-documented phenomenon that increased P leads to reduced aquatic plant cover and eutrophication of water bodies.
A limitation of this study is that only one experimental pulse release of nutrients occurred at each site during sampling. It would have been more representative and protected against sampling bias to perform at least three nutrient assimilation experiments at random locations within each wetland. Due to logistical constraints, we were unable to bring multiple mesocosms to each site. However, in future research, having replication in these measurements would be ideal and allow calculation of a mean assimilation coefficient with a measure of variance for each wetland.
Conclusions
As excessive N and P increasingly lead to eutrophication of surface waters, it is important to understand the factors controlling N and P assimilation in wetlands. Surprisingly, we found that wetland impoundments with a high index of biological integrity did not assimilate more N and P than impoundments in poor biological condition. Further elucidation of patterns controlling nutrient assimilation or removal in wetland impoundments is needed in future research. Percent removal of N and P could be determined at multiple points in time and space, giving a clearer picture of overall patterns. For example, it has been shown that the majority of studies have reported wetlands as a sink for nutrients, except those conducted over a year or more or involving frequent sampling during high-flow events. Our results suggest that abiotic variables, including nutrient concentrations within the water column and soil, dissolved oxygen, water depth, and soil texture, should be measured as important factors related to nutrient assimilation.
Data Availability
The datasets generated during the current study are available from the corresponding author upon reasonable request.
References
Ameel J, Axler R, Owen C (1993) Persulfate digestion for determination of total nitrogen and phosphorus in low-nutrient waters. Am Environ Lab 10:1–11
Baron JS, Hall E, Nolan B, Finlay J, Bernhardt E, Harrison J, Chan F, Boyer E (2013) The interactive effects of excess reactive nitrogen and climate change on aquatic ecosystems and water resources of the United States. Biogeochemistry 114:71–92
Botter G, Basu N, Zanardo S, Rao P, Rinaldo A (2010) Stochastic modeling of nutrient losses in streams: interactions of climatic, hydrologic, and biogeochemical controls. Water Resour Res, 46
Brinson MM, Bradshaw HD, Kane ES (1984) Nutrient assimilative capacity of an alluvial floodplain swamp. J Appl Ecol :1041–1057
CH2M Hill (2009) Development of an assessment framework for the impounded wetlands of Great Salt Lake. Draft report to the Utah Div. Of Water Quality. Dept. of Environmental Quality
Cheng FY, Basu NB (2017) Biogeochemical hotspots: role of small water bodies in landscape nutrient processing. Water Resour Res 53:5038–5056
Cohen MJ, Kurz MJ, Heffernan JB, Martin JB, Douglass RL, Foster CR, Thomas RG (2013) Diel phosphorus variation and the stoichiometry of ecosystem metabolism in a large spring-fed river. Ecol Monogr 83:155–176
Cohenour R, Thompson K (1966) Geologic setting of Great Salt Lake
Crumpton WG, Stenback GA, Fisher SW, Stenback JZ, Green DIS (2020) Water quality performance of wetlands receiving nonpoint-source nitrogen loads: nitrate and total nitrogen removal efficiency and controlling factors. J Environ Qual 49:735–744. https://doi.org/10.1002/jeq2.20061
de-Bashan LE, Bashan Y (2004) Recent advances in removing phosphorus from wastewater and its future use as fertilizer (1997–2003). Water Res 38:4222–4246. https://doi.org/10.1016/j.watres.2004.07.014
Downard R, Endter-Wada J (2013) Keeping wetlands wet in the western United States: adaptations to drought in agriculture-dominated human-natural systems. J Environ Manage 131:394–406. https://doi.org/10.1016/j.jenvman.2013.10.008
Downard R, Endter-Wada J, Kettenring KM (2014) Adaptive wetland management in an uncertain and changing arid environment. Ecol Soc. 19https://doi.org/10.5751/es-06412-190223
Fisher J, Acreman M (2004) Wetland nutrient removal: a review of the evidence. Hydrol Earth Syst Sci 8:673–685
Gee GW, Or D (2002) 2.4 particle-size analysis. Methods of soil analysis Part 4:255–293
Gray LJ (2009) Macroinvertebrates in the Wetlands of the Great Salt Lake. Citeseer
Gray LJ (2012) Macroinvertebrates of the Willard Spur Wetlands: Literature Review and Results of Sampling in 2011. Prepared for the Willard Spur Steering Committee and Science Panel and the Utah Department of Environmental Quality, Division of Water Quality, Salt Lake City, Utah.(30 September 2012)
Gray LJ (2013) Macroinvertebrate and zooplankton communities of the Willard Spur Wetlands: results of 2012 sampling. Prepared for the Willard Spur Steering Committee and Science Panel and the Utah Department of Environmental Quality, Division of Water Quality, Salt Lake City, Utah.(28 February 2013).
Groffman PM, Altabet MA, Bohlke JK, Butterbach-Bahl K, David MB, Firestone MK, Giblin AE, Kana TM, Nielsen LP, Voytek MA (2006) Methods for measuring denitrification: diverse approaches to a difficult problem. Ecol Appl 16:2091–2122. https://doi.org/10.1890/1051-0761(2006)016[2091:mfmdda]2.0.co;2
Hauer F, Lamberti G (2006) Methods in stream ecology. Academic Press
Hinckley BR, Etheridge JR, Peralta AL (2020) Wetland Conditions differentially influence Nitrogen Processing within Waterfowl impoundments. Wetlands 40:1117–1131. https://doi.org/10.1007/s13157-019-01246-8
IBM Corp (2021) IBM SPSS Statistics for Windows. IBM Corp, Armonk, NY
Jones SF, Schutte CA, Roberts BJ, Thorne KM (2022) Seasonal impoundment management reduces nitrogen cycling but not resilience to surface fire in a tidal wetland. J Environ Manage 303:114153
Kadlec R (1999) The limits of phosphorus removal in wetlands. Wetlands Ecol Manage 7:165–175
Kijowski AM, Neill J, Wickline A, Swift J, Butler JK, Kimberly DA, Van Leeuwen J, Luft J, Stone K (2020) American White Pelicans of Gunnison Island, Great Salt Lake, Utah. p. 311–344. Great Salt Lake Biology. Springer.
Kunickis S, Gilliam J, Evans R, Dukes M (2010) Soil characteristics and their role in developing conditions favorable for denitrification. p. 1–6. 19th World Congress of Soil Science. Soil Solutions for a Changing World
Lemke AM, Kirkham KG, Wallace MP, VanZomeren CM, Berkowitz JF, Kovacic DA (2022) Nitrogen and phosphorus removal using tile-treatment wetlands: a 12-year study from the midwestern United States. J Environ Qual 51:797–810. https://doi.org/10.1002/jeq2.20316
Liu G-r, Ye C-s, He J-h, Qian Q, Jiang H (2009) Lake sediment treatment with aluminum, iron, calcium and nitrate additives to reduce phosphorus release. J Zhejiang University-SCIENCE A 10:1367–1373
McGeehan SL, Naylor DV (1988) Automated instrumental analysis of carbon and nitrogen in plant and soil samples. Commun Soil Sci Plant Anal 19:493–505
Messer T, Burchell M, Bírgand F (2017) Comparison of four nitrate removal kinetic models in two distinct Wetland Restoration Mesocosm Systems. Water 9:517. https://doi.org/10.3390/w9070517
Odum EP (1984) The Mesocosm BioScience 34:558–562. https://doi.org/10.2307/1309598
Penton CR, Deenik JL, Popp BN, Bruland GL, Engstrom P, D. St. Louis and, Tiedje J (2013) Importance of sub-surface rhizosphere-mediated coupled nitrification–denitrification in a flooded agroecosystem in Hawaii. Soil Biology and Biochemistry, 57:362–373. https://doi.org/10.1016/j.soilbio.2012.10.018
Racchetti E, Longhi D, Ribaudo C, Soana E, Bartoli M (2016) Nitrogen uptake and coupled nitrification–denitrification in riverine sediments with benthic microalgae and rooted macrophytes. Aquat Sci 79:487–505. https://doi.org/10.1007/s00027-016-0512-1
R Core Team (2020) R: a Language Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria
Reddy KR, Diaz OA, Scinto LJ, Agami M (1995) Phosphorus dynamics in selected wetlands and streams of the lake Okeechobee Basin. Ecol Eng 5:183–207. https://doi.org/10.1016/0925-8574(95)00024-0
Richardson CJ, Qian S, Craft CB, Qualls RG (1996) Predictive models for phosphorus retention in wetlands. Wetlands Ecol Manage 4:159–175. https://doi.org/10.1007/bf01879235
Saunders D, Kalff J (2001) Nitrogen retention in wetlands, lakes and rivers. Hydrobiologia 443:205–212
Spieles DJ, Mitsch WJ (1999) The effects of season and hydrologic and chemical loading on nitrate retention in constructed wetlands: a comparison of low-and high-nutrient riverine systems. Ecol Eng 14:77–91
Stapanian MA, Schumacher W, Gara B, Monteith SE (2016) Negative effects of excessive soil phosphorus on floristic quality in Ohio wetlands. Sci Total Environ 551–552:556–562. https://doi.org/10.1016/j.scitotenv.2016.02.041
Tank JL, Rosi-Marshall EJ, Baker MA, Hall RO (2008) Are rivers just big streams? A pulse method to quantify nitrogen demand in a large river. Ecology 89:2935–2945
Tanner CC, D’Eugenio J, McBride GB, Sukias JPS, Thompson K (1999) Effect of water level fluctuation on nitrogen removal from constructed wetland mesocosms. Ecol Eng 12:67–92. https://doi.org/10.1016/S0925-8574(98)00055-X
Tavşanoğlu ÜN, Šorf M, Stefanidis K, Brucet S, Türkan S, Agasild H, Baho DL, Scharfenberger U, Hejzlar J, Papastergiadou E (2017) Effects of nutrient and water level changes on the composition and size structure of zooplankton communities in shallow lakes under different climatic conditions: a pan-european mesocosm experiment. Aquat Ecol 51:257–273
Tiner RW (2005) Assessing cumulative loss of wetland functions in the Nanticoke River watershed using enhanced National Wetlands Inventory data. Wetlands 25:405–419. https://doi.org/10.1672/15
UDWQ (2014) Final Integrated Report: Water Quality. p. 1–22. https://deq.utah.gov/water-quality/2014-final-integrated-report-water-quality
U.S. Environmental Protection Agency (1993a) Method 350.1: Nitrogen, Ammonia (Colorimetric, Automated Phenate). Cincinnati, OH
U.S. Environmental Protection Agency (1993b) Method 353.2: Determination of nitrate-nitrite nitrogen by automated colorimetry. Cincinnati, OH
U.S. Environmental Protection Agency (1993c) Method 365.1: Determination of phosphorus by semi-automated colorimetry. Cincinnati, OH
Vollenweider RA (1975) Input-output models. Schweizerische Z für Hydrol 37:53–84
Vymazal J (2007) Removal of nutrients in various types of constructed wetlands. Sci Total Environ 380:48–65
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Wang X, Han X, Shi X, Zhu C, Sun X, Zhang C (2006) Kinetics of nutrient uptake and release by phytoplankton in East China Sea: model and mesocosm experiments. Hydrobiologia 563:297–311
Weller NA, Childers DL, Turnbull L, Upham RF (2016) Aridland constructed treatment wetlands I: Macrophyte productivity, community composition, and nitrogen uptake. Ecol Eng 97:649–657. https://doi.org/10.1016/j.ecoleng.2015.05.044
Winton RS, Moorman M, Richardson CJ (2016) Waterfowl Impoundments as sources of Nitrogen Pollution. Water Air Soil Pollut 227:390. https://doi.org/10.1007/s11270-016-3082-x
Xia X, Liu T, Yang Z, Michalski G, Liu S, Jia Z, Zhang S (2017) Enhanced nitrogen loss from rivers through coupled nitrification-denitrification caused by suspended sediment. Sci Total Environ 579:47–59. https://doi.org/10.1016/j.scitotenv.2016.10.181
Ye S, Covino TP, Sivapalan M, Basu NB, Li HY, Wang SW (2012) Dissolved nutrient retention dynamics in river networks: a modeling investigation of transient flows and scale effects. Water Resour Res, 48
Zar JH (1972) Significance testing of the Spearman rank correlation coefficient. J Am Stat Assoc 67:578–580
Acknowledgements
Thank you to Toby Hooker, Rebekah Downard, and Jeff Ostermiller from the Utah Division of Water Quality for assistance with site selection, project development, and data analysis protocols. We are grateful to the federal, state, and private land managers who granted access to wetland impoundments for sampling. We acknowledge the students and staff at the Aquatic Biogeochemistry Lab and National Aquatic Monitoring Center at Utah State University and the Environmental Analytical Lab at Brigham Young University for water and soil sample analysis. Thanks to Hannah Fouts, Andrew Luymes, Kerri Russell, Adam Norris, Morgan Parkinson, and Emily Gervais for their help in the field. Thank you to Susan Durham, Scott George, and Trevor Williams for assistance in data analysis.
Funding
This project was funded by the U.S. Environmental Protection Agency Region 8 via a Wetland Program Development Grant (CD-96851301) to Michelle Baker. This research was also supported by the U.S. National Science Foundation (PRFB 2109778, EAR 2011439, and EAR 2012123).
Author information
Authors and Affiliations
Contributions
Rachel Wood designed the experiments, collected and analyzed the data, and wrote the manuscript. Michelle Baker designed the experiments, collected data, and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
Authors Rachel Wood and Michelle Baker declare they have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wood, R.L., Baker, M.A. Assessing Nutrient Assimilation by Wetland Impoundments Across Environmental Gradients. Wetlands 43, 66 (2023). https://doi.org/10.1007/s13157-023-01711-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01711-5