Abstract
The aim of this study was to understand the variation in traits relevant for desiccation avoidance among bryophyte species dominant in rich fens and to assess whether these traits explain the formation of a hummock-hollow gradient within peatlands. In samples of 10 species (Aulacomnium palustre, Calliergonella cuspidata, Climacium dendroides, Hamatocaulis vernicosus, Helodium blandowii, Marchantia polymorpha, Plagiomnium ellipticum, Sphagnum teres, S. warnstorfii, Tomentypnum nitens) collected in rich fens of NE Poland, we calculated: canopy bulk density of wet (CDW) and dry (CDD) colonies, maximum water content of bryophyte colonies (WCmax), desiccation rate (K), shoot area index (SAI), canopy dry mass per surface-projected area (CMA), and specific leaf area of a whole living bryophyte part (SLA). The hummock-forming frequency was quantified for each species in the field. Sphagna had the highest WCmax, SAI and CDW, T. nitens and C. dendroides had the lowest WCmax and SLA, P. ellipticum had the highest K, the lowest CMA and CDD. Hummock-forming frequency was positively correlated with CMA and generally negatively related to K, with exception of H. vernicosus showing a high water-retaining ability (low K) despite a hollow or lawn form of growth.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The last two decades have seen a lot of research effort being put into the quantification of interspecific variation in functional traits of vascular plants. This has resulted in the inference of important trade-offs and relationships between morphology and physiology of vascular plants (e.g. Grime et al. 1997; Craine et al. 2001; Wright et al. 2004; Diaz et al. 2016) and of the relationships between the community-weighted composition of plant traits and functioning of ecosystems (e.g. Garnier et al. 2004; Violle et al. 2007; Bardgett et al. 2014). However, these advances in plant trait research have largely ignored bryophytes (but see: Lang et al. 2009; Soudzilovskaia et al. 2013; Lett et al. 2017). This is despite the fact that bryophytes are abundant in all biomes worldwide (e.g. Werger and During 1989; Longton 1997; Gradstein et al. 2001; Bates et al. 2005), and are very important as determinants of wetlands functioning throughout the world (Gorham 1991). A better trait-based understanding of the functioning of these organisms and their role in ecosystems is urgent.
Although some research efforts have been taken to develop the concept of bryophyte traits (Cornelissen et al. 2007; Waite and Sack 2010; Deane-Coe and Stanton 2017), the functional trait framework remains difficult to apply to bryophytes. Most bryophytes have clonal or colonial life-forms (Bates 1998, for instance, classifies them into tufts, cushions, dendroids, mats, wefts, fans and pendants), which constitute their actual ecologically functional units – more than individual shoots (e.g. Bates 1998; Elumeeva et al. 2011; Michel et al. 2012; Rice et al. 2014). Traits of colonies, rather than traits of shoots, are more relevant predictors for bryophyte strategies related to habitat water availability, because of the large dependence of bryophytes on transient external water supply (Bates 1998; Proctor 2000). For instance, denser cushions are able to retain more water in the capillary spaces created between shoots (e.g. Rice 2012). In addition, the laminar boundary layer saturated with water vapour that is formed above the relatively smooth surfaces of dense cushions allows lowering the rate of evaporation (i.e. minimise water loss) as compared to rougher, irregular colonies (Proctor 1981, 2000; Bates 1998).
In bryophyte-dominated peatlands, the pattern of hummocks, hollows and lawns formed by different bryophyte species creates a diversity of microhabitats affecting the overall biodiversity and ecosystem processes. Also, key feedback mechanisms in the functioning of many mire ecosystems are mediated by bryophytes – here especially the process of hummock formation by Sphagna has received much attention (e.g. Granath et al. 2010). All Sphagnum species have morphological adaptations which help them to avoid desiccation (Vitt et al. 2014), but the level of desiccation avoidance is higher in hummock than in hollow Sphagnum species due to the higher density of stems, branches and leaves (e.g. Li et al. 1992; Rydin et al. 2006; Hájek and Beckett 2008; Hájek 2014; Hájek and Vicherová 2014; Weston et al. 2015). Hummock bryophytes tend to destabilize open fen mires, enhancing their shifts to bogs and woodlands – this process has been attributed to the cation exchange mechanism resulting in enhanced acidification but also to fostering the establishment of shrubs and trees, which results in increased evapotranspiration and subsequent drainage of the mire (Rydin and Jeglum 2013). On the other hand, hummock bryophytes, due to their high ability to avoid desiccation and low decomposability, may stabilize bogs or poor fens by keeping the upper peat layer moist and slowing down its decomposition (Turetsky et al. 2012). Hummocks may also preserve microhabitats for rare plant species in open fens (e.g. Carex dioica, Liparis loeselii, Parnassia palustris), providing for them a shelter against light competition and flooding (Kotowski et al. 2013).
While functional traits of Sphagna have been relatively well studied in relation to mire functioning (Hájek 2014 and references therein), far less is known in this respect about brown mosses, many of which form hummocks as well and can be hypothesised to trigger ecosystem shifts in rich fens. Soudzilovskaia et al. (2010) shed light on how brown mosses feature a high cation exchange capacity, similar to Sphagna, whereas Soudzilovskaia et al. (2013) demonstrated that thermal conductivity and heat capacity in brown mosses is related to their moisture content, the latter being able therewith to impact peat temperature and decomposition. Furthermore, Goetz and Price (2015) proved that both hummocks of Sphagna and Tomenthyphnum nitens enhance the capillary rise of water, resulting in desiccation avoidance.
The overall aim of our study was to enhance the inclusion of bryophytes in functional diversity analyses of mire ecosystems and to measure variability of water-related traits of the analysed dominant rich-fen bryophyte species. Our second, more specific, objective was to match the diversity in traits of the species with their hummock formation frequency. We hypothesised that the traits having a key relationship with the avoidance of desiccation necessary for hummock formation (and therefore evident in species that frequently form hummocks) are: (1) maximum water content, determining the ability to retain water in the colony, (2) canopy mass per area, describing how compact the colony is and (3) traits related to the large total leaf area in the colony, indicating the size of the water storage space outside the moss tissues.
Materials and Methods
Analysed Species and Sampling
We analysed traits of ten fen bryophytes: Aulacomnium palustre (Hedw.) Schwägr., Calliergonella cuspidata (Hedw.) Loeske, Climacium dendroides (Hedw.) F.Weber & D.Mohr, Helodium blandowii (F.Weber & D.Mohr) Warnst., Hamatocaulis vernicosus (Mitt.) Hedenäs, Marchantia polymorpha L., Plagiomnium ellipticum (Brid.) T.J.Kop., Sphagnum teres (Schimp.) Ångstr., Sphagnum warnstorfii Russow, Tomentypnum nitens (Hedw.) Loeske. The species nomenclature follows Hill et al. (2007). The selected species are dominant in the bryophyte community of open rich fens in NE Poland. Samples were collected by hand from relatively monospecific (containing < 5% of non-target species) stands in the region of Augustów Forest (NE Poland): Rospuda valley (E 22.954°, N 52.904°), Borsuki (E 23.312°, N 53.900°) and upper Biebrza valley (E 23.330°, N 53.733°). All the three sites host well-preserved low-productive brown moss-sedge vegetation with species like Carex lasiocarpa, C. rostrata, Equisetum fluviatile, Menyanthes trifoliata dominating the herb layer. We cut intact patches (c. 10 cm x 10 cm) from five colonies of each species (from different sites or from distant locations within one site; samples of each species were collected at two of the three sites mentioned above). The collected bryophyte samples included both fresh green shoots and ca. 1 cm of their brown basal parts. Consequently, the sample height varied and corresponded to the thickness of the living bryophyte layer (Fig. A1 in Appendix 1).
The bryophyte samples analysed in this study are deposited in the herbarium of the Faculty of Biology at the University of Warsaw. The samples received accession numbers from WA71259 to WA71308.
Assessing Hummock-Forming Frequency
For each species, we counted the share of colonies forming hummocks which were defined as mounds elevated at least 10 cm above the level of a neighbouring hollow or a moss-lawn surface (Fig. A2 in Appendix 2). We laid a circular path within a radius of 2 m around each of the five colonies of a species sampled for ex-situ measurements (the length of each path was c. 12.57 m). We noted whether the species (sampled in the middle) was present and whether it formed a hummock at every step (c. 50 cm) along a circular path. The hummock-forming frequency was calculated as the percentage of hummocks formed by the species in all records of the species within five circles (Fig. A3 in Appendix 2).
Measurements of Traits of Bryophytes
In the collected samples from bryophyte colonies we carefully removed all shoots of non-target species to reduce their abundance to < 1% while retaining an intact structure of a colony as far as possible. We placed the samples in plastic pots and immersed in tap water. After allowing the surplus water to leak down, we measured the following in each water-saturated sample: the volume, the surface-projected area and the mass. All samples were then placed in an open greenhouse and their mass was measured twice a day until the weight of the sample was changing for less than 100 mg per day (about 0.2% of the original saturated mass). After 14 days, the dry mass of samples was measured after drying them in an oven until constant mass. The treatment of moss samples used for the subsequent measurements followed the protocol proposed by Elumeeva et al. (2011) for moss colonies (but see Table A1 in Appendix 3 for differences). Three species (A. palustre, P. ellipticum, T. nitens) were analysed in both studies. From the above-described measurements we calculated maximum water content of bryophyte colonies (i.e. including both internal and external tissue water, WCmax), desiccation rate (K), canopy bulk density – wet (CDW), canopy bulk density – dry (CDD), canopy mass per area (CMA) (Table 1).
Additionally, we measured projected area of separate shoots (taken from each of the sampled bryophyte colonies) and their oven-dried mass. Shoot-projected area was measured following Bond-Lamberty and Gower (2007) with some modifications. We took green parts of several (10–25) dry shoots from each sample, watered them to full turgor, scanned them at 300 DPI using a flatbed scanner (CanoScan LiDE 110), then dried them in the oven at 70 °C until constant mass and then weighed them. Sphagnum branches were not separated before scanning. The TIFF images obtained from scanning were proceeded in Paint Shop Pro as follows: the blue channel (which allowed to best distinguish bryophytes from the background) has been extracted from the colour images and the histogram was extended in such a way that black (0) was set at 50, whereas white (255) was set at 190. We then established a threshold at level 155, checked the images for artefacts and if present, cleaned them on screen by hand. Finally, we automatically counted the number of pixels < 155. Projected shoot area was computed by dividing the final image pixel count by the scan resolution in pixels per unit area. Following Bond-Lamberty and Gower (2007), we calculated specific leaf area (SLA), called so for consistency with other studies; however, it is, in fact, the specific shoot area (projected area of green (alive) parts of the shoots/dry mass of them) (Table 1). With use of SLA values, we calculated also shoot area index (SAI) (Table 1).
We compared species trait means with one-way ANOVA followed by the Tukey test, as well as using 95% confidence intervals (CI) of the means (fractions for hummock-forming frequency), which allowed for both qualitative and quantitative comparisons. Relations among traits were visualised using Principal Components Analysis in Canoco 5 (Šmilauer and Lepš 2014) and checked for correlations using Pearson’s test.
Results
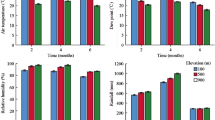
The mean maximum water content of bryophyte colonies (WCmax) varied among the 10 analysed species from c. 760% for C. dendroides to c. 1500% for S. warnstorfii (Table A2 in Appendix 5). The mean CMA varied from c. 530 g m− 2 for P. ellipticum to c. 1300 g m− 2 for S. teres (Table A2 in Appendix 5). SLA was the highest in P. ellipticum and in both Sphagnum species and both Sphagnum species had higher mean SAI than all non-Sphagnum species (Figs. 1 and 2). Looking at the results in terms of species trait characteristics, we found that Sphagnum species were related to high WCmax, SAI and CDW values, T. nitens and C. dendroides to low WCmax and SLA values, whereas P. ellipticum was distinguished by high values of K and low of CMA and CDD (Figs. 1 and 2).
Mean values and 95% confidence intervals for means of functional traits (n = 5) of bryophytes (for hummock-forming frequency – fractions and 95% confidence intervals for fractions). Mean values of traits differed in Tukey multiple comparisons (p < 0.05) for species not sharing the same letter (a, b, c …). AP – Aulacomnium palustre, CC – Calliergonella cuspidata, CD – Climacium dendroides, HB – Helodium blandowii, HV – Hamatocaulis vernicosus, MP – Marchantia polymorpha, PE – Plagiomnium ellipticum, ST – Sphagnum teres, SW – Sphagnum warnstorfii, TN – Tomentypnum nitens
PCA of species and their mean values of water balance traits. Refer Table 1 for traits abbreviations. PCA centred and standardized by traits, 1st and 2nd axes shown, explained variation: 1st axis – 47.7, 2nd axis – 35.7, 3rd axis – 11.1, 4th axis – 3.8. Size of circles indicates hummock-forming frequency of the species which was projected as supplementary variable in the analysis. For species abbreviations refer to Fig. 1. The dashed lines surround the groups characterised in Fig. 3
Hummock-forming frequency was positively correlated with CMA (R = 0.77, p < 0.05; Table 2) whereas CMA was negatively correlated with desiccation rate (R = 0.70, p < 0.05). Surprisingly, we detected no correlation between hummock-forming frequency and WCmax. H. vernicosus rarely formed hummocks despite similar functional traits, i.e. relatively high CWA and CDD and low K, to hummock-forming species (Figs. 1 and 2).
Discussion
We found considerable interspecific traits variability in water-related traits, even though we focused on bryophytes dominant in one type of a fen mire and sampled only the lawn-hummock part of the gradient, avoiding typical submerged species. Species which effectively form hummocks have generally low desiccation rate (K), probably owing it to high canopy mass per area (CMA). H. vernicosus is an exception to this rule. WCmax and SLA were not directly related to the hummock formation, whereas high SAI described only one type of hummocks i.e. Sphagnum hummocks. Below we discuss in detail the findings of our study.
Variation in Traits
Our estimations of WCmax were by at least 500% points (pp) of dry weight lower (Fig. A4 in Appendix 3, the difference between nearer limits of 95% CI for the means) than those measured by Elumeeva et al. (2011) for three species common to both studies (A. palustre, P. ellipticum, T. nitens).This variation may result from differences in sample preparation. The relative instability of WCmax might explain why Elumeeva et al. (2011) and Michel et al. (2012) obtained quite different values for the time of 50% water loss for colonies of A. palustre (114, SE = 13 vs. 40, SE = 5) and T. nitens (137, SE = 9 vs. 24, SE = 5), in spite of collecting samples in the same localities and using similar methodologies. In our opinion, reproducible WCmax measurements for entire colonies with preserved natural structure (including all external water retained) are hardly possible due to inevitable disruption of the structure during sample cleaning (removing fragments of vascular plants and non-target moss shoots), performed with different precision by each researcher. WCmax correlated positively with CDW (Table 2; Fig. 2), due to the high proportion of water in the weight of a moist sample. Using dry mass for calculating canopy bulk density (CDD), gives results that are not affected by the amount of water in the moist sample. However, CDD hardly differentiated our species, except for the exceptionally low value in P. ellipticum. This species has a distinct morphology, with relatively large and thin leaves loosely placed at the stem. This is consistent with the findings of Waite and Sack (2010), that bryophyte leaf area is negatively correlated with canopy bulk density (our CDD trait).
The desiccation rate (K) is relatively unaffected by initial differences in WCmax. Unlike WCmax, the K index calculated from our data for A. palustre and T. nitens was similar to that calculated from data of Elumeeva et al. (2011), which was denoted by overlapping 95% confidence intervals (Fig. A4 in Appendix 3). We found differences for P. ellipticum, which may be due to a different morphology of this species in our sites (more shaded and more nutrient rich) and in northern mires (poorer and more exposed). Plagiomnium species express some phenotypic plasticity (Wigh 1972) and especially P. ellipticum is a species with a highly variable morphology (Smith 2004). Polish P. ellipticum had visually much looser colonies and larger individual shoots and leaves, as compared to this species growing in Scandinavia (our observations).
The distinct morphology of Sphagna was reflected by their higher shoot area index (SAI) as compared to all other species (Figs. 1 and 2). We recorded similar SAI values for C. cuspidata as van der Hoeven et al. (1993) (although they used a different method of photo-electric planimeters) and much higher values for T. nitens, A. palustre and S. warnstorfii, compared to those of Bond-Lamberty and Gower (2007). A different approach to the separation between dead and living moss tissue is one possible reason for this discrepancy (Niinemetes and Tobias 2014).
Canopy dry mass per canopy surface-projected area (CMA) roughly followed the variation in CDD (Fig. 1). Both CDD and CMA depend on tissue thickness and canopy structure (loose or densely packed shoots), whereas CMA includes also information on the height of the green part of the moss. The values of CMA obtained in our study are higher than values of SMA (shoot mass per area) found by Rice et al. (2008, 2011) and Wang et al. (2014), which could be a result of different methodology, as well as those found by Waite and Sack (2010), which is likely due to a different canopy structure of the Hawaiian and Central-European mosses.
Bond-Lamberty and Gower (2007) found that Sphagna have higher SLA values than other mosses, but found no significant differences in SLA among non-Sphagnum moss species. We confirmed differences between Sphagna and some (but not all analysed) non-Sphagnum species, whereas we found also differences between P. ellipticum and other non-Sphagnum species (Figs. 1 and 2), SLA is the only trait for which we did not confirm the difference between Sphagna and P. ellipticum. SLA was positively correlated with WCmax (Fig. 2; Table 2), pointing at the role of leaves and branches in the water holding.
Functional Background of Desiccation Avoidance
The vital trade-off for bryophytes is between desiccation tolerance and desiccation avoidance. According to Vitt et al. (2014), Sphagnum and Marchantia species are typical desiccation avoiders, while Tomentypnum, Aulacomnium and Helodium have an evident prevalence of neither desiccation avoidance nor desiccation tolerance strategy, showing some features of both. Goetz and Price (2015) found that the trade-off between desiccation tolerance and avoidance is likely to result in a moss species having both adaptation types, but the proportion of them can vary among species. They found also that Sphagnum and T. nitens enhance capillary rise of water in their canopies to avoid desiccation, but this capillary rise is more limited at low water tables in Tomentypnum due to the larger pore sizes (predominately external pores, occurring between overlapping leaves and branches and in the rhizoid tomentum). In Sphagna, the continuous network of small pores (including hyaline cells) enhances capillary rise of the water table to the capitula even during relatively low water tables (Goetz and Price 2015; Glime 2015 – height of capillary rise for different bryophyte structures).
Desiccation rate (K) was the highest for P. ellipticum. The ineffective avoidance of desiccation in this species as compared to both Sphagnum species is probably due to less effective holding of external capillary water. P. ellipticum is therefore less effective in external water transport (ectohydry), while investing more in transmitting water through the stem (endohydry) (Glime 2015). However, K coefficient of C. dendroides, the other species with an internal water transport, did not differ significantly from that of Sphagna. The rigid stem of Plagiomnium and Climacium, enabling endohydry (Glime 2015), helps them probably to efficiently redistribute water to leaves after desiccation.
Van der Hoeven and During (1997) found that three pleurocarpous mosses (including Calliergonella cuspidata) could rapidly return to an original shoot density after 50% thinning, which was attributed to the regulation of stem density by some intrinsic mechanisms. Densely packed shoots in bryophyte colonies give them an advantage of reduced water loss, which may outweigh the cost of reduced light availability (Bates 1988). Denser colonies (higher CDD), such as in A. palustre, C. dendroides, H. vernicosus, M. polymorpha or S. warnstorfii, ensure a lower desiccation (Figs. 1 and 2). Avoiding desiccation is quite well represented by canopy mass per projected area (CMA), as it increases with biomass allocation to structural components, such as rhizoid tomentum (A. palustre, T. nitens) or hyaline cells of Sphagnum (Rice et al. 2008; Waite and Sack 2010; Wang et al. 2014).
The group of species with relatively low SLA included T. nitens, C. dendroides and M. polymorpha, while the group of species with relatively high SLA includes: P. ellipticum and both Sphagnum species (Fig. 2). As shown above, Sphagna are typical desiccation avoiders, while P. ellipticum is clearly not, so SLA alone cannot be directly connected with desiccation avoidance ability of a species.
Desiccation Avoidance and Hummock Formation
Four species in our analysis, A. palustre, C. dendroides, S. warnstorfii and T. nitens have been identified as hummock-forming in previous studies (e.g. Vitt 1990; Wassen and Joosten 1996; Bauer et al. 2007; Vitt 2007; Manukjanová et al. 2014). However, T. nitens was reported from some areas as a species having a preference to low-lying (wet) microsites (Bauer et al. 2007). Previous studies assessed hummock-forming ability as an arbitrary binary trait, whereas we quantified species potential to form hummocks, which enabled us to correlate it with water balance and canopy structure traits.
Hummock formation is closely related to the desiccation rate (K) and canopy mass per area (CMA). The other traits considered turned out to be less useful or biased by measuring methods. WCmax and CDW depend on the way the samples are soaked. SLA may indicate diverse properties of the species. CDD varied too little between the studied species for a good predictor of ecological functions. SAI was high only for Sphagna, so it cannot predict other types of hummock formation.
Our connotation that a dense packing of biomass per surface area (high CMA) is a precondition for building hummocks that effectively hold water is consistent with Laing et al. (2014), who showed a positive relationship between water content of bryophytes and the dry mass of their stems as well as the density of stems per unit area. High CMA of hummocks may result from a high annual productivity and/or slow decomposition. In bogs, hummock Sphagna have lower or similar productivity and lower decomposition rates than hollow species (Rochefort et al. 1990; Johnson and Damman 1991; Turetsky et al. 2008; Hájek 2009; Bengtsson et al. 2016). In turn, slow decomposition of Sphagnum mosses is the a process preconditioning peat accumulation in bogs, whereas in rich fens peat accumulates rather due to high net primary production and despite fast decomposition (Vitt et al. 2009; Mettrop et al. 2014).
Moisture levels and traits that regulate water availability may also impact the rate of photosynthesis and affect overall productivity. Species that form hummocks tend to have low desiccation rate (Figs. 2 and 3). Bryophytes have lower light saturation points than vascular plants (Marshall and Proctor 2004; Glime 2007) and their photosynthesis is strongly regulated by water availability – due to thick external water films, CO2 diffusion resistance limits CO2 fixation (Hájek 2014). The extent of external water films is proportional to the specific leaf area (SLA), which has, therefore, a different adaptive function in bryophytes than in vascular plants (trade-off between maximisation of light capture and stress tolerance). Photosynthetic assimilation of CO2 by bryophytes is hindered by low, as well as by too high, water content (Schipperges and Rydin 1998; Jauhiainen and Silvola 1999; Ueno and Kanda 2006; Hájek 2014). Species retaining more external water (e.g. Sphagna) can stay photosynthetically active for a longer time but this adaptation is on the cost of slower CO2 diffusion due to thick water films of external water (see Hájek (2014) for a detailed review). In permanently-hydrated peatland mosses (mainly Sphagna) growing in full sunlight, the rate of chlorophyll excitation may greatly exceed the capacity for CO2 fixation, so investments in photoprotection are necessary. Being desiccation avoiders, Sphagna cannot escape the light stress by drying out, rolling up their shoots or leaves and becoming inactive like other bryophytes in drier habitats; they must remain metabolically active (Hájek et al. 2009). Nevertheless, Sphagna can afford a less efficient photosynthesis and slow growth under the reduced competition in open mires (Hájek et al. 2009).
Summary of traits characteristics for all species analysed. For species abbreviations refer to Fig. 1 and for traits abbreviations – to Table 1. Species are divided into five groups (separated with black vertical lines in the head of the table) regarding their traits characteristics. Trait values are generalized into three relative categories: high (H), medium (M) and low (L) based on Fig. 2
Avoiding desiccation is advantageous when forming hummocks, however, the main reason why species form hummocks could be to avoid toxicity of calcium (Ca2+), iron (Fe2+) or manganese (Mn2+) (cf. Vicherová et al. 2015), which are often very abundant in fen water. Similarly, Granath et al. (2010) showed for a typical hummock species, Sphagnum fuscum, that flooding alone did not reduce the photosynthesis rate in contrast to flooding with calcareous rich-fen water. Therefore, hummock formation is less advantageous in species that are able to tolerate high concentrations of these elements. For example, calcium-tolerant H. vernicosus (Vicherová et al. 2015) forms hummocks rarely despite low K and relatively high CMA values, typical for the those of the species in our analysis that form hummocks more often (Figs. 2 and 3). Our results contradict Bauer et al. (2007) stating that H. vernicosus lacks efficient adaptations to uptake and retain water. This species apparently has such adaptations but it uses them to build dense lawns just above the groundwater level – not mounding to higher hummocks.
Summing-up and Conclusions
The analysed bryophyte species can be divided into five groups (Figs. 2 and 3). The first group, containing both Sphagnum species, is characterized by high values of all studied traits except for CDD (average value) and K (low value). These species frequently form hummocks and are most effective in avoiding desiccation. The second group (C. dendroides and T. nitens) differs from the first one mainly with regard to lower WCmax and SLA values and slightly lower CDW and SAI. These species also frequently form hummocks and effectively avoid drying out, but they do this with help of different structures than Sphagna, the external water is held in these species by rhizoid tomentum rather than between leaves. The third group includes morphologically and phylogenetically quite different species (H. blandowii, C. cuspidata, A. palustre and M. polymorpha) which however share intermediate values of all studied traits. P. ellipticum (the only member of the fourth group) is characterized by high K and SLA values as well as low CDW, CDD, CMA and hummock-forming frequency, the remaining features have an intermediate value here. Finally, H. vernicosus (the only member of the fifth group) has low K and a low hummock-forming frequency, scoring intermediate in the remaining traits. The hummock-forming frequency shows a tendency to decrease from the first to the fifth group, while the desiccation rate shows an opposite pattern increasing from the first to the fourth group. H. vernicosus (the fifth group) is an exception from this trade-off, combining a low desiccation rate (i.e. a high capability of retaining water) with a low hummock-forming frequency.
Our study is among the first to quantify and assess the ecological significance of water-related functional traits in rich fen bryophytes. Further research should in our opinion link these traits to productivity and growth rates of species, as well as their decomposition rates and recalcitrance of biomass, which is a necessary step to analyse the role of bryophyte functional diversity for peat accumulation.
Data Availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Bates JW (1988) The effect of shoot spacing on the growth and branch development of the moss Rhytidiadelphus triquetrus. New Phytol 109:499–504. https://doi.org/10.1111/j.1469-8137.1988.tb03726.x
Bates JW (1998) Is “life-form” a useful concept in bryophyte ecology? Oikos 82:223–237. https://doi.org/10.2307/3546962
Bates JW, Thompson K, Grime JP (2005) Effects of simulated long-term climatic change on the bryophytes of a limestone grassland community. Glob Change Biol 11:757–769. https://doi.org/10.1111/j.1365-2486.2005.00953.x
Bauer IE, Tirlea D, Bhatti JS, Errington RC (2007) Environmental and biotic controls on bryophyte productivity along forest to peatlands ecotones. Can J Bot 85:463–475. https://doi.org/10.1139/B07-045
Bengtsson F, Granath G, Rydin H (2016) Photosynthesis, growth, and decay traits in Sphagnum – a multispecies comparison. Ecol Evol 6:3325–3341. https://doi.org/10.1002/ece3.2119
Bardgett RD, Mommer L, De Vries FT (2014) Going underground: root traits as drivers of ecosystem processes. Trends Ecol Evol 29:692–699. https://doi.org/10.1016/j.tree.2014.10.006
Bond-Lamberty B, Gower ST (2007) Estimation of stand-level leaf area for boreal bryophytes. Oecologia 151:584–592. https://doi.org/10.1007/s00442-006-0619-5
Cornelissen JHC, Lang SI, Soudzilovskaia NA, During HJ (2007) Comparative cryptogam ecology: a review of bryophyte and lichen traits that drive biogeochemistry. Ann Bot 99:987–1001. https://doi.org/10.1093/aob/mcm030
Craine JM, Froehle J, Tilman DG, Wedin DA, Chapin FS (2001) The relationships among root and leaf traits of 76 grassland species and relative abundance along fertility and disturbance gradients. Oikos 93:274–285. https://doi.org/10.1034/j.1600-0706.2001.930210.x
Deane-Coe KK, Stanton D (2017) Functional ecology of cryptogams: scaling from bryophyte, lichen, and soil crust traits to ecosystem processes. New Phytol 213:993–995. https://doi.org/10.1111/nph.14408
Diaz S, Kattge J, Cornelissen JHC, Wright IJ, Lavorel S et al (2016) The global spectrum of plant form and function. Nature 529:167–171. https://doi.org/10.1038/nature16489
Elumeeva TG, Soudzilovskaia NA, During HJ, Cornelissen JHC (2011) The importance of colony structure versus shoot morphology for the water balance of 22 subarctic bryophyte species: factors affecting bryophyte water balance. J Veg Sci 22:152–164. https://doi.org/10.1111/j.1654-1103.2010.01237.x
Garnier E, Cortez J, Billes G, Navas ML, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A, Neill C, Toussaint JP (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637. https://doi.org/10.1890/03-0799
Glime JM (2007) Light: the shade plants. – In: Bryophyte Ecology, vol. 1, Physiological Ecology, URL: https://digitalcommons.mtu.edu/bryophyte-ecology/ (accessed 15 June 2016)
Glime JM (2015) Water relations: movement. – In: Bryophyte Ecology, vol. 1, Physiological Ecology, URL: https://digitalcommons.mtu.edu/bryophyte-ecology/ (accessed 15 June 2016)
Goetz JD, Price JS (2015) Role of morphological structure and layering of Sphagnum and Tomenthypnum mosses on moss productivity and evaporation rates. Can J Soil Sci 95:109–124. https://doi.org/10.4141/cjss-2014-092
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to climatic warming. Ecol Appl 1:182–195. https://doi.org/10.2307/1941811
Gradstein SR, Churchill SP, Salazar-Allen N (2001) Guide to the bryophytes of tropical America. Memoirs of the New York Botanical Garden 86. NYBG Press, New York
Granath G, Strengbom J, Rydin H (2010) Rapid ecosystem shifts in peatlands: linking plant physiology and succession. Ecology 91:3047–3056. https://doi.org/10.1890/09-2267.1
Grime JP, Thompson K, Hunt R, Hodgson JG, Cornelissen JHC et al (1997) Integrated screening validates primary axes of specialisation in plants. Oikos 79:259–281. https://doi.org/10.2307/3546011
Hájek T (2009) Habitat and species controls on Sphagnum production and decomposition in a mountain raised bog. Boreal Environ Res 14:947–958
Hájek T (2014) Physiological ecology of peatland bryophytes. In: Hanson DT, Rice SK (eds) Photosynthesis in Bryophytes and Early Land plants. Springer, Dordrecht, pp 233–252
Hájek T, Beckett RP (2008) Effect of water content components on desiccation and recovery in Sphagnum mosses. Ann Bot 101:165–173. https://doi.org/10.1093/aob/mcm287
Hájek T, Tuittila E-S, Ilomets M, Laiho R (2009) Light responses of mire mosses - a key to survival after water-level drawdown? Oikos 118:240–250. https://doi.org/10.1111/j.1600-0706.2008.16528.x
Hájek T, Vicherová E (2014) Desiccation tolerance of Sphagnum revisited: a puzzle resolved. Plant Biol 16:765–773. https://doi.org/10.1111/plb.12126
Hill MO, Preston CD, Bosanquet SDS, Roy DB (2007) BRYOATT: attributes of british and irish mosses, liverworts and hornworts. Centre for Ecology and Hydrology, Huntingdon, Cambridgeshire
Jauhiainen J, Silvola J (1999) Photosynthesis of Sphagnum fuscum at long-term raised CO2 concentrations. Ann Bot Fenn 36:11–19
Johnson LC, Damman AWH (1991) Species-controlled Sphagnum decay on a south Swedish raised bog. Oikos 61:234–242. https://doi.org/10.2307/3545341
Kotowski W, Jabłońska E, Bartoszuk H (2013) Conservation management in fens: do large tracked mowers impact functional plant diversity? Biol Conserv 167:292–297. https://doi.org/10.1016/j.biocon.2013.08.021
Lang SI, Cornelissen JHC, Klahn T, Van Logtestijn RSP, Broekman R, Schweikert W, Aerts R (2009) An experimental comparison of chemical traits and litter decomposition rates in a diverse range of subarctic bryophyte, lichen and vascular plant species. J Ecol 97:886–900. https://doi.org/10.1111/j.1365-2745.2009.01538.x
Laing CG, Granath G, Belyea LR, Allton KE, Rydin H (2014) Tradeoffs and scaling of functional traits in Sphagnum as drivers of carbon cycling in peatlands. Oikos 123:817–828. https://doi.org/10.1111/oik.01061
Lett S, Nilsson M-C, Wardle DA, Dorrepaal E (2017) Bryophyte traits explain climate-warming effects on tree seedling establishment. J Ecol 105:496–506. https://doi.org/10.1111/1365-2745.12688
Li Y, Glime JM, Liao C (1992) Responses of two interacting Sphagnum species to water level. J Bryol 17:59–70. https://doi.org/10.1179/jbr.1992.17.1.59
Longton RE (1997) The role of bryophytes and lichens in polar ecosystems. In: Woodin SJ, Marquiss M (eds) Ecology of arctic environments. Blackwell, Oxford, pp 69–96
Manukjanová A, Štechová T, Kučera J (2014) Drought survival test of eight fen moss species. Cryptogam Bryol 35:397–403. https://doi.org/10.7872/cryb.v35.iss4.2014.397
Marshall M, Proctor MC (2004) Are bryophytes shade plants? Photosynthetic light responses and proportions of chlorophyll a, chlorophyll b and total carotenoids. Ann Bot 94:593–603. https://doi.org/10.1093/aob/mch178
Mettrop IS, Cusell C, Kooijman AM, Lamers LPM (2014) Nutrient and carbon dynamics in peat from rich fens and Sphagnum fens during different gradations of drought. Soil Biol Biochem 68:317–328. https://doi.org/10.1016/j.soilbio.2013.10.023
Michel P, Lee WG, During HJ, Cornelissen JHC (2012) Species traits and their non-additive interactions control the water economy of bryophyte cushions: bryophyte species mixtures and water economy. J Ecol 100:222–231. https://doi.org/10.1111/j.1365-2745.2011.01898.x
Niinemetes Ü, Tobias M (2014) Scaling light harvesting from moss “leaves” to canopies. In: Hanson DT, Rice SK (eds) Photosynthesis in Bryophytes and Early Land plants. Springer, Dordrecht, pp 151–172
Proctor MCF (1981) Diffusion resistance in bryophytes. In: Grace J, Ford ED, Jarvis PG (eds) Plants and their Atmospheric Environment. Blackwell Scientific, Oxford, pp 219–229
Proctor MCF (2000) The bryophyte paradox: tolerance of desiccation, evasion of drought. Plant Ecol 151:41–49. https://doi.org/10.1023/A:1026517920852
Rice SK (2012) The cost of capillary integration for bryophyte canopy water and carbon dynamics. Lindbergia 35:53–62
Rice SK, Aclander L, Hanson DT (2008) Do bryophyte shoot systems function like vascular plant leaves or canopies? Functional trait relationships in Sphagnum mosses (Sphagnaceae). Am J Bot 95:1366–1374
Rice SK, Hanson DT, Portman Z (2014) Structural and functional analyses of bryophyte canopies. In: Hanson DT, Rice SK (eds) Photosynthesis in Bryophytes and Early Land plants. Springer, Dordrecht, pp 173–186
Rice SK, Neal N, Mango J, Black K (2011) Relationships among shoot tissue, canopy and photosynthetic characteristics in the feathermoss Pleurozium schreberi. The Bryologist 114:367–378. https://doi.org/10.1639/0007-2745-114.2.367
Rochefort L, Vitt DH, Bayley SE (1990) Growth, production, and decomposition dynamics of Sphagnum under natural and experimentally acidified conditions. Ecology 71:1986–2000. https://doi.org/10.2307/1937607
Rydin H, Gunnarsson U, Sundberg S (2006) The role of Sphagnum in peatland development and persistence, in Vitt DH, Wieder RK (eds), Boreal Peatland Ecosystems. Ecological studies 188: 49–65, Springer, Berlin, Heidelberg
Rydin H, Jeglum JK (2013) The Biology of Peatlands, 2nd edn. Oxford University Press, Oxford
Schipperges B, Rydin H (1998) Response of photosynthesis of Sphagnum species from contrasting microhabitats to tissue water content and repeated desiccation. New Phytol 148:677–684. https://doi.org/10.1046/j.1469-8137.1998.00311.x
Šmilauer P, Lepš J (2014) Multivariate analysis of Ecological Data using CANOCO 5. Cambridge University Press, Cambridge
Smith AJE (2004) The Moss Flora of Britain and Ireland, 2nd edn. Cambridge University Press, Cambridge
Soudzilovskaia NA, Cornelissen JH, During HJ, van Logtestijn RS, Lang SI, Aerts R (2010) Similar cation exchange capacities among bryophyte species refute a presumed mechanism of peatland acidification. Ecology 91:2716–2726. https://doi.org/10.1890/09-2095.1
Soudzilovskaia NA, van Bodegom PM, Cornelissen JHC (2013) Dominant bryophyte control over high-latitude soil temperature fluctuations predicted by heat transfer traits, field moisture regime and laws of thermal insulation. Funct Ecol 27:1442–1454. https://doi.org/10.1111/1365-2435.12127
Turetsky MR, Bond-Lamberty B, Euskirchen E, Talbot J, Frolking S, McGuire AD, Tuittila E-S (2012) The resilience and functional role of moss in boreal and arctic ecosystems. New Phytol 196:49–67. https://doi.org/10.1111/j.1469-8137.2012.04254.x
Turetsky MR, Crow SE, Evans RJ, Vitt DH, Wieder RK (2008) Trade-offs in resource allocation among moss species control decomposition in boreal peatlands. J Ecol 96:1297–1305. https://doi.org/10.1111/j.1365-2745.2008.01438.x
Ueno T, Kanda H (2006) Photosynthetic response of the arctic semi-aquatic moss Calliergon giganteum to water content. Aquat Bot 85:241–243. https://doi.org/10.1016/j.aquabot.2006.03.010
van der Hoeven EC, During HJ (1997) The effect of density on size frequency distributions in chalk grassland bryophyte populations. Oikos 80:533–539. https://doi.org/10.2307/3546626
van der Hoeven EC, Huynen CIJ, During HJ (1993) Vertical profiles of biomass, light intercepting area and light intensity in chalk grassland mosses. J Hattori Bot Lab 74:261–270
Vicherová E, Hájek M, Hájek T (2015) Calcium intolerance of fen mosses: physiological evidence, effects of nutrient availability and successional drivers. Perspect Plant Ecol Evol Syst 17:347–359. https://doi.org/10.1016/j.ppees.2015.06.005
Violle C, Navas M-L, Vile D, Kazakou E, Fortunel C, Hummel I, Garnier E (2007) Let the concept of trait be functional! Oikos 116:882–892. https://doi.org/10.1111/j.0030-1299.2007.15559.x
Vitt DH (1990) Growth and production dynamics of boreal mosses over climatic, chemical and topographic gradients. Bot J Linn Soc 104:35–59
Vitt DH (2007) Estimating moss and lichen ground layer net primary production in tundra, peatlands, and forests. In: Fahey TJ, Knapp AK (eds) Principles and Standards for Measuring Primary production. Oxford University Press, Oxford, pp 82–105
Vitt DH, Crandall-Stotler B, Wood AJ, Rajakaruna N, Boyd RS, Harris TB (2014) Bryophytes: survival in a dry world through tolerance and avoidance. In: Rajakaruna N, Boyd RS, Harris TB (eds) Plant Ecology and Evolution in Harsh environments. Nova Science Publishers, New York, pp 267–295
Vitt DH, Wieder RK, Scott KD, Faller S (2009) Decomposition and peat accumulation in rich fens of boreal Alberta, Canada. Ecosystems 12:360–373. https://doi.org/10.1007/s10021-009-9228-6
Waite M, Sack L (2010) How does moss photosynthesis relate to leaf and canopy structure? Trait relationships for 10 Hawaiian species of contrasting light habitats. New Phytol 185:156–172. https://doi.org/10.1111/j.1469-8137.2009.03061.x
Wang Z, Bao W, Feng D, Lin H (2014) Functional trait scaling relationships across 13 temperate mosses growing in wintertime. Ecol Res 29:629–639. https://doi.org/10.1007/s11284-014-1146-1
Wassen MJ, Joosten JHJ (1996) In search of a hydrological explanation for vegetation changes along a fen gradient in the Biebrza Upper Basin (Poland). Vegetatio 124:191–209. https://doi.org/10.1007/BF00045494
Werger MJA, During HJ (1989) A subterranean moss greenhouse in the Californian desert. The Bryologist 92:411–412
Weston DJ, Timm CM, Walker AP, Gu L, Muchero W, Schmutz J, Shaw AJ, Tuskan GA, Warren JM, Wullschleger SD (2015) Sphagnum physiology in the context of changing climate: emergent influences of genomics, modelling and host-microbiome interactions on understanding ecosystem function. Plant Cell Environ 38:1737–1751. https://doi.org/10.1111/pce.12458
Wigh K (1972) Cytotaxonomical and modification studies in some Scandinavian mosses. Lindbergia 1:130–152
Wright IJ, Reich PB, Westoby M, Ackerly DD, Baruch Z et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Acknowledgements
Bryophyte samples were collected following the permission WPN.6400.52.2014.AP of the Regional Director for Environmental Protection in Białystok. This work was supported by the Polish-Norwegian Research Programme operated by the National Centre for Research and Development under the Norwegian Financial Mechanism 2009–2014 in the frame of Project Contract No Pol-Nor/199522/86/2013 (project MIRACLE). We would like to thank Marina Abramchuk, Zygmunt Jabłoński and Marcin Kułak for help in moss samples analyses.
Funding
The research leading to these results has received funding from the Polish-Norwegian Research Programme operated by the National Centre for Research and Development under the Norwegian Financial Mechanism 2009–2014 in the frame of Project Contract No Pol-Nor/199522/86/2013 (project MIRACLE).
Author information
Authors and Affiliations
Contributions
EJ, NS and WK contributed to the study conception and design. Material preparation and data collection were performed by EJ and NS. Data analysis was performed by EJ. The first draft of the manuscript was written by EJ. All authors commented on previous versions of the manuscript, as well as read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Appendix 1. Height of bryophyte samples analysed
Appendix 2. Methods of sampling and assessing hummock-forming frequency
Appendix 3. Differences in methods used to determine water-related bryophyte traits in our study and by Elumeeva et al. (2011) and comparison of results
Appendix 4. Relation of bryophyte sample water content and time
Appendix 5. Means and 95% confidence intervals for the functional traits of bryophytes
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jabłońska, E., Kotowski, W. & Soudzilovskaia, N. Desiccation Avoidance and Hummock Formation Traits of rich fen Bryophytes. Wetlands 43, 21 (2023). https://doi.org/10.1007/s13157-023-01669-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01669-4