Abstract
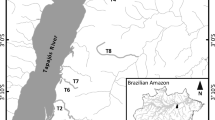
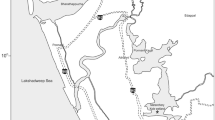
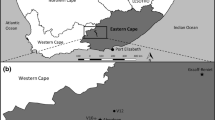
The relationship between organisms’ traits and the characteristics of their habitats has been a central theme in ecology. Using two complementary approaches to analyze three-matrices of species composition, functional trait, and environment data (RLQ and fourth-corner analyses), we tested if environmental gradient determines aquatic plant traits in ponds in the Pantanal wetland. Furthermore, we tried to understand if plant composition is spatially structured and if environmental gradient is correlated with taxonomic and functional community properties. We sampled aquatic plants coverage in 20 ponds and measured pond size, depth and pH to represent the environmental gradient. We found significant turnover of several traits of aquatic plants along the environmental gradient, being niche partitioning the most probable process structuring plant communities. There was no evidence of spatial autocorrelation determining species composition in the scale of the study, which may indicate that the annual flooding can act as homogenizing factor that promotes species co-occurrence along the ponds. Taxonomic abundance and functional richness were positively related with environmental gradient. Larger and deeper ponds provide higher resource availability thus supporting more functional attributes and vegetation cover. Our results support the hypothesis that niche process is the main driver of aquatic plants in the Pantanal.

Similar content being viewed by others
References
Akasaka M, Takamura N (2011) The relative importance of dispersal and the local environment for species richness in two aquatic plant growth forms. Oikos 120:38–46
Alahuhta J, Kanninen A, Hellsten S, Vuorif KM, Kuoppala M, Hämäläinen H (2013) Environmental and spatial correlates of community composition, richness and status of boreal lake macrophytes. Ecological Indicators 32:172–181
Alahuhta J, Virtala A, Hjort J, Ecke F, Johnson LB, Sass L, Heino J (2017) Average niche breadths of species in lake macrophyte communities respond to ecological gradients variably in four regions on two continents. Oecologia 184(1):219–235
Arber A (1920) Water plants; a study of aquatic angiosperms. Cambridge the University press, Cambridge, p 462
Arthaud F, Vallod D, Robin J, Bornette G (2012) Eutrophication and drought disturbance shape functional diversity and life-history traits of aquatic plants in shallow lakes. Aquatic Sciences 74:471–481
Arthaud F, D. Vallod D, Robin J, Wezel A, Bornette G (2013) Short-term succession of aquatic plant species richness along ecosystem productivity and dispersal gradients in shallow lakes. Journal of Vegetation Science 24: 148–156
Baattrup-Pedersen A, Göthe E, Riis T, O'Hare MT (2016) Functional trait composition of aquatic plants can serve to disentangle multiple interacting stressors in lowland streams. Science of the Total Environment 543:230–238
Bergier I, Assine LM (2015) Dynamics of the Pantanal wetland in South America. Springer International Publishing, Cham
Bornette G, Puijalon S (2009) Macrophytes: Ecology of Aquatic Plants. In: Encyclopedia of Life Sciences (ELS). John Wiley and Sons, Ltd, Chichester
Bozelli RL, Thomaz SM, Padial AA, Lopes PM, Bini LM (2015) Floods decrease zooplankton beta diversity and environmental heterogeneity in an Amazonian floodplain system. Hydrobiologia 753:233–241
Catian G, Silva DM, Súarez YR, Scremin-Dias E (2018) Effects of flood pulse dynamics on functional diversity of macrophyte communities in the Pantanal wetland. Wetlands 38:975–991
Colwell RK (2013) EstimateS: statistical estimation of species richness and shared species from samples (version 9 and earlier. User’s guide and application. Storrs, CT: published by the author). Accessed online 10 June 2014 at: http://purl.oclc.org/estimates
Correa SB, Costa-Pereira R, Fleming T, Goulding M, Anderson JT (2015) Neotropical fish–fruit interactions: eco-evolutionary dynamics and conservation. Biological Reviews 90:1263–1278
Costa M, Telmer KH, Evans TL, Almeida TIR, Diakun MT (2015) The lakes of the Pantanal: inventory, distribution, geochemistry, and surrounding landscape. Wetlands Ecology and Management 23:19–39
Cunha NL, Delatorre M, Rodrigues RB, Vidotto C, Gonçalves F, Scremin-Dias E, Damasceno-Júnior G, Pott VJ, Pott A (2012) Structure of aquatic vegetation of a large lake, western border of the Brazilian Pantanal. Brazilian Journal of Biology 72(3):519–531
Cunha NL, Fischer E, Lorenz-Lemke AP, Barrett SCH (2014) Floral variation and environmental heterogeneity in a tristylous clonal aquatic of the Pantanal wetlands of Brazil. Annals of Botany 114:1637–1649
Cronk JK, Fennessy MS (2001) Wetland plants: biology and ecology. Lewis Publishers, Boca Raton
Datry T, Bonada N, Heino J (2016) Towards understanding the organization of metacommunities in highly dynamic ecological systems. Oikos 125:149–159
De Bie T, De Meester L, Brendonck L, Martens K, Goddeeris B et al (2012) Body size and dispersal mode as key traits determining metacommunity structure of aquatic organisms. Ecology Letters 15:740–747
Delatorre M, Cunha N, Raizer J, Ferreira VL (2015) Evidence of stochasticity driving anuran metacommunity structure in the Pantanal wetlands. Freshwater Biology 60:2197–2207
Dolédec S, Chessel D, terBraak CJF, Champely S (1996) Matching species traits to environmental variables: a new three-table ordination method. Environmental and Ecological Statistics 3:143–166
Dray S, Legendre P, Peres-Neto PR (2006) Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecological Modelling 196:483–493
Dray S, Pélissier R, Couteron P, Fortin MJ, Legendre P, Peres-Neto PR, Bellier E, Bivand R, Blanchet FG, de Cáceres M, Dufour AB, Heegaard E, Jombart T, Munoz F, Oksanen J, Thioulouse J, Wagner HH (2012) Community ecology in the age of multivariate multiscale spatial analysis. Ecological Monographs 82:257–275
Dray S, Legendre P (2008) Testing the species traits environment relationships: the fourth-corner problem revisited. Ecology 89:3400–3412
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. Journal of Statistical Software 22(4):1–20
Dray S, Chessel D, Thioulouse J (2003) Co-inertia analysis and the linking of ecological data tables. Ecology 84:3078–3089
Fernandes IM, Henriques-Silva R, Penha J, Zuanon J, Peres-Neto PR (2014) Spatiotemporal dynamics in a seasonal metacommunity structure is predictable: the case of floodplain-fish communities. Ecography 37:464–475
Fu H, Yuan G, Zhong J, Cao T, Ni L, Xie P (2013) Environmental and ontogenetic effects on intraspecific trait variation of a macrophyte species across five ecological scales. PLoS One 8:e62794
Fu H, Zhong J, Yuan G, Ni L, Xie P, Cao T (2014) Functional traits composition predict macrophytes community productivity along a water depth gradient in a freshwater lake. Ecology and Evolution 4:1516–1523
Fukami T (2015) Historical contingency in community assembly: integrating niches, species pools, and priority effects. Annual Review of Ecology, Evolution, and Systematics 46:1–23
Grime JP (2001) Plant strategies, vegetation processes, and ecosystem properties. Second edition. Chichester (United Kingdom) and. John Wiley and Sons, New York
Grime JP (2006) Trait convergence and trait divergence in herbaceous plant communities: mechanisms and consequences. Journal of Vegetation Science 17:255–260
Hill MO, Smith AJE (1976) Principal component analysis of taxonomic data with multi-state discrete characters. Taxon 25:249–255
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biometrical Journal 50:346–363
Junk WJ, Cunha CN, Wantzen KM, Petermann P, Strüssmann C, Marques MI, Adis J (2006) Biodiversity and its conservation in the Pantanal of Mato Grosso, Brazil. Aquatic Sciences 68:278–309
Krebs CJ (1999) Ecological methodology. Addison-Welsey Longman, New York, pp 1–620
Laliberté E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305
Legendre P, Legendre L (2012) Numerical Ecology. Elsevier Science BV, Amsterdam
Legendre P, Borcard D, Blanchet G, Dray S (2010) PCNM: PCNM spatial Eigenfunction and principal coordinate analyses (R package version 2.1/r82). Accessed online 10 June 2014 at: http://r-forge.r-project.org/projects/sedar/
Legendre P, Fortin MJ, Borcard D (2015) Should the mantel test be used in spatial analysis? Methods in Ecology and Evolution 6:1239–1247
Lehmann A, Castella E, Lachavanne JB (1997) Morphological traits and spatial heterogeneity of aquatic plants along sediment and depth gradients, Lake Geneva, Switzerland. Aquatic Botany 55:281–299
Mason NWH, de Bello F, Doležal J, Lepš J (2011) Niche overlap reveals the effects of competition, disturbance and contrasting assembly processes in experimental grassland communities. Journal of Ecology 99:788–796
Monção FS, Santos AM, Bini LM (2012) Aquatic macrophyte traits and habitat utilization in the upper Paraná River floodplain. Brazil. Aquatic Botany 102:50–55
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P et al. (2017) Vegan: Community ecology package. Version 2.4-4. Available at: http://CRAN.R-project.org/package=vegan
Padial AA, Carvalho P, Thomaz SM, Boschilia SM, Rodrigues RB, Kobayashi JT (2009) The role of an extreme flood disturbance on macrophyte assemblages in a Neotropical floodplain. Aquatic Sciences 71:389–398
Peres-Neto PR, Legendre P, Dray S, Borcard D (2006) Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87:2614–2625
Pott A, Silva JSV (2015) Terrestrial and Aquatic Vegetation Diversity of the Pantanal Wetland Volume 37 of the series The Handbook of Environmental Chemistry. In: Bergier I, Assine LM (Eds) Dynamics of the Pantanal Wetland in South America. Springer International Publishing, Cham, pp 111-131
Pott VJ, Pott A (2000) Plantas Aquáticas do Pantanal. Brasília: Embrapa
R Core Team 2017. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Ricklefs RE (1987) Community diversity: relative roles of local and regional processes. Science 235:167–171
Santamaría L (2002) Why are most aquatic plants widely distributed? Dispersal, clonal growth and small-scale heterogeneity in a stressful environment. Acta Oecologica 23:137–154
Schneider B, Cunha ER, Marchese M, Thomaz SM (2018) Associations between Macrophyte life forms and environmental and morphometric factors in a large sub-tropical floodplain. Frontiers in Plant Science 9:195
Sculthorpe CD (1967) The biology of aquatic vascular plants. Edward Arnold, London
Thomaz SM, Bini LM, Bozelli RL (2007) Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia 579:1–13
Van Geest GJ, Coops H, Roijackers RMM, Buijse AD, Scheffer M (2005) Succession of aquatic vegetation driven by reduced water-level fluctuations in floodplain lakes. Journal of Applied Ecology 42:251–260
Vellend M (2016) The theory of ecological communities. Princeton University Press
Vellend M, Srivastava DS, Anderson KM, Brown CD, Jankowski JE, Kleynhans EJ (2014) Assessing the relative importance of neutral stochasticity in ecological communities. Oikos 123:1420–1430
Vieira J Jr, Necchi O Jr (2006) Photosynthetic characteristics of a tropical population of Nitella cernua (Characeae, Chlorophyta). Brazilian Journal of Plant Physiology 18:379–388
Wetzel AJ, Likens GE (2000) Limnological analyses, 3rd edn. Springer-Verlag, New York
Acknowledgments
We thank T. Siqueira, A. A. Padial, S. M. Thomaz and F. O. Roque for comments on early versions. We thank Embrapa Pantanal and Centro de Pesquisa do Pantanal (CPP) for financial support; W.M. Tomas, S.M. Sales, G. Mourão and D.P. Eaton for logistic support. A.F. Beda for assistance with field sampling and V. Pott for species identification. Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) provided a master’s fellowship to M. Delatorre and R. B. Rodrigues and financial support to V. L. Ferreira (#309305/2018-7) and G. A. Damasceno-Júnior. Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) provided scholarships to N. L. Cunha and M. Delatorre. This study was partially financed by CAPES - Finance Code 001. V. L. Ferreira is researcher CNPq (#409003/2018-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Table S1
Ponds environmental variables used to correlate traits and environment on aquatic plants communities in the Pantanal wetlands, Brazil. (DOCX 16 kb)
Fig. S1
Sample based Jackknife rarefaction curve estimated by 1000 randomizations using the frequency of incidence of species, which represents an estimate of ponds aquatic macrophyte richness, during the end of the dry-season, in the Pantanal wetlands, Corumbá, Mato Grosso do Sul, Brazil. Error bars are the standard deviation. (DOCX 27 kb)
Fig. S2
Mantel correlogram showing lack of spatial auto-correlation between Hellinger transformed aquatic macrophytes and geographical coordinates in 20 ponds of the Pantanal wetlands. Dot filled in black represents significant auto-correlation. (DOCX 81 kb)
Rights and permissions
About this article
Cite this article
Delatorre, M., da Cunha, N.L., Rodrigues, R.B. et al. Trait-Environment Relationship of Aquatic Vegetation in a Tropical Pond Complex System. Wetlands 40, 299–310 (2020). https://doi.org/10.1007/s13157-019-01189-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-019-01189-0