Abstract
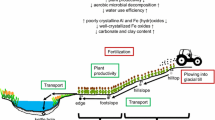
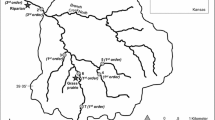
Changes in climate and land use pressures in the boreal region may influence peatland stream sediment organic carbon (C) dynamics. Fifteen to 50% of stream sediment organic C resides in recalcitrant pools, with the lipids accounting for up to 25% of the peatland C pool. Nevertheless, lipid mineralization within peatlands and their draining stream sediments is poorly understood. Potential esterase activity, which is known to be an important enzyme group in recalcitrant C decomposition, is presented as an indicator of lipid hydrolysis in three streams surrounding an intermediate fen peatland. Organic C concentration ranged from 5 to 250 g C kg−1 sediment during the sampling period, with the recalcitrant C pool accounting for 15 to 20%. Fourier Transform Infrared (FT-IR) spectra were typical of humic-like substances, such as aliphatic compounds (including lipids), phenolic and other aromatic structures, carboxyl groups, and polysaccharide structures. Esterase activity was positively correlated with organic C and total nitrogen (N) concentrations, and sediment pH. Esterase was most active in the presence of high organic C fractions and recalcitrant C, supporting the hypothesis that lipid hydrolysis in the peatland stream sediments may be an important and underestimated component of peatland C sequestration dynamics.






Similar content being viewed by others
References
Artz RRE, Chapman SJ, Robertson AHJ, Potts JM, Laggoun DeFarge F, Gogo S, Comont L, Disnar JR, Francez AJ (2008) FTIR spectroscopy can be used as a screening tool for organic matter quality in regenerating cutover peatlands. Soil Biol Biochem 40:515–527
Baker M, Dahm CN, Valett M (1999) Acetate retention and metabolism in the hyporheic zone of a mountain stream. Limnol Oceanogr 44:1530–1539
Battin T (1997) Assessment of fluorescein diacetate hydrolysis as a measure of total esterase activity in natural stream sediment biofilms. Sci Total Environ 198:51–60
Battin T (2000) Hydrodynamics is a major determinant of streambed biofilms activity: from the sediment to the reach scale. Limnol Oceanogr 45:1308–1319
Billett MF, Palmer SM, Hope D, Deacon C, Storeton-West R, Hargreaves KJ, Flechard C, Fowler D (2004) Linking land-atmosphere-stream carbon fluxes in a lowland peatland system. Glob Biogeochem Cycles. doi:10.1029/2003GB002058
Bott TL, Kaplan LA (1985) Bacterial biomass, metabolic state, and activity in stream sediments: relation to environmental variables and multiple assay comparisons. Appl Environ Microbiol 50:508–522
Chafiq M, Gibert J, Claret C (1999) Interactions among sediments, organic matter, and microbial activity in the hyporheic zone of an intermittent stream. Can J Fish Aquat Sci 56:487–495
Cocozza C, D’Orazio V, Miano TM, Shotyk W (2003) Characterization of solid and aqueous phases of a peat bog profile using molecular fluorescence spectroscopy, ESR and FT-IR, and comparison with physical properties. Org Geochem 34:49–60
Dawson J, Billet MF, Hope D, Palmer SM, Deacon CM (2004) Sources and sinks of aquatic carbon in a peatland stream continuum. Biogeochemistry 70:71–92
Feng X, Xu Y, Jaffe R, Schlesinger WH, Simpson MJ (2010) Turnover rates of hydrolysable aliphatic lipids in Duke Forest soils determined by compound specific 13C isotopic analysis. Org Geochem 41:573–579
Fenner N, Freeman C, Reynolds B (2005) Observations of a seasonally shifting thermal optimum in peatland carbon-cycling processes: implication for the global carbon cycle and soil enzyme methodologies. Soil Biol Biochem 37:1814–1821
Fernandes AN, Giovanela M, Esteves VI, de Souza-Sierra MM (2010) Elemental and spectral properties of peat and soil samples and their respective humic substances. J Mol Struct 971:33–38
Fontvieille DA, Outaguerouine A, Thevenot DR (1992) Fluorescein diacetate hydrolysis as a measure of microbial activity in aquatic systems: application to activated sludges. Environ Technol 13:531–540
Freeman C, Nevison GB (1999) Simultaneous analysis of multiple enzymes in environmental samples using methylumbelliferyl substrates and HPLC. J Environ Qual 28:1378–1380
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to climate warming. Ecol Application 1:182–195
Hajje N, Jaffe R (2006) Molecular characterization of Cladium peat from the Florida Everglades: biomarker association with humic fractions. Hydrobiologia 569:99–112
Intergovernmental Panel on Climate Change (2000) Land use, land use change, and forestry. A special report of the IPCC. ISBN 92-9169-114-3
Jolivet C, Arrouays D, Lévèque J, Andreux F, Chenu C (2003) Organic carbon dynamics in soil particle-size separates of sandy Spodosols when forest is cleared for maize cropping. Eur J Soil Sci 54:257–268
Lorenz K, Lal R, Preston CM, Nierop KGL (2007) Strengthening the soil organic pool by increasing contributions from recalcitrant aliphatic bio(macro)molecules. Geoderma 142:1–10
McLaughlin, J. W. (2004). Carbon assessment in boreal wetlands of Ontario. Ontario Ministry of Natural Resources, Ontario Forest Research Institute, Forest Research Information Paper 158, 79 p
McLaughlin JW (2009) Boreal mixed-wood watershed riparian zone cation cycling during two contrasting climatic years. Soil Sci Soc Am J 73:1408–1418
McLaughlin JW, Phillips SA (2006) Soil carbon, nitrogen, and base cations cycling 17 years after whole-tree harvesting in a low-elevation red spruce (Picea rubens)-balsam fir (Abies balsamea) forested watershed in central Maine, USA. For Ecol Manage 222:234–253
McLaughlin JW, Webster KL (2010) Alkalinity and acidity cycling and fluxes in an intermediate fen peatland in northern Ontario. Biogeochemistry 99:143–155
Prest VK, Donaldson JA, Mooers HD (2000) The omar story: the role of omars in assessing glacial history of west-central North America. Géogr Phys Quatern 54:257–270
Reiche M, Gleixner G, Kusel K (2010) Effect of peat quality on microbial greenhouse gas formation in an acidic fen. Biogeosciences 7:187–198
Sánchez-Monedero MA, Mondini C, Cayuela ML, Roig A, Contin M, De Nobili M (2008) Fluorescein diacetate hydrolysis, respiration, and microbial biomass in freshly amended soils. Biol Fertil Soils 44:885–890
Schindler DW, Bayley SE, Parker BR, Beaty KG, Cruikshank DR, Fee EJ, Schindler EU, Stainton MP (1996) The effects of climatic warming on the properties of boreal lakes and streams at the experimental lakes area, northwestern Ontario. Limnol Oceanogr 41:1004–1017
Schnurer J, Rosswall T (1982) Fluorescein diacetate hydrolysis as a measure of total microbial activity in soil and litter. Appl Environ Microbiol 43:1256–1261
Siegel DI, Glaser PH, Janecky DR (2006) The dynamic balance between organic acids and circumneutral groundwater in a large boreal peat basin. J Hydrol 320:421–431
Stubberfield LCF, Shaw PJA (1990) A comparison of tetrazolium reduction and FDA hydrolysis with other measures of microbial activity. J Microbiol Meth 12:151–162
Sun M-Y, Shi W, Lee RF (2000) Lipid-degrading enzyme activities with distribution and degradation of fatty acids in the mixing zone of Altamaha estuarine sediments. Org Geochem 31:199–209
Tipping E, Billett MF, Bryant CL, Buckingham S, Thacker SA (2010) Sources and ages of dissolved organic matter in peatland streams: evidence from chemistry mixture modeling and radiocarbon data. Biogeochemistry 100:121–137
Turunen J, Tomppo E, Tolonen K, Reinikainen A (2002) Estimating carbon accumulation rates of undrained mires in Finland– application to boreal and subarctic regions. Holocene 12:69–80
Weinbauer MG, Beckmann C, Hofle MG (1998) Utility of green fluorescent nucleic acid dyes and aluminum oxide membrane filters for rapid epifluorescence enumeration of soil and sediment bacteria. Appl Environ Microbiol 64:5000–5003
Weisenberg GLB, Gocke M, Kuzyakov Y (2009) Fast incorporation of root-derived lipids and fatty acids into soil- evidence from a short term multiple 14CO2 pulse labeling experiment. Org Geochem. doi:10.1016/j.orggeochem.2009.12.007
Williams CJ, Shingara EA, Yavitt JB (2000) Phenol oxidase activity in peatlands in New York State: response to summer drought and peat type. Wetlands 20:416–421
Zar JH (1984) Biostatistical analysis, second edition. Prentice-Hall, Inc, Englewood Cliffs
Acknowledgments
Support for this project was provided by the Ontario Forest Research Institute, Ontario Ministry of Natural Resources, Project CC-167 and Michigan Technological University. Special thanks to John Adler, Casey Huckins, Martin Jurgensen, and Tom Noland for their insightful comments. We also thank Lisa Buse for editing the draft manuscript and Ted Atkinson, Darren Falcioni, Ravi Kanipayor, John Ralston, Lesley Rich, Stephanie Hall, and Trudy Vaittinen for technical assistance. Finally, we are grateful to the editors and three anonymous reviewers for their constructive comments and suggestions, which greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Below is the link to the electronic supplementary material.
Online resource 1
Growing season (June-October 2004) mean ± SD (μeq l−1, unless otherwise stated) porewater (PW) and surface water (SW) constituents, geophysical characteristics, and sediment physical, chemical, and biological features for three streams draining an intermediate fen peatland (TP: total phosphorus; TN: total nitrogen; DIC: dissolved inorganic carbon; DOC: dissolved organic carbon). Capitalized superscripts indicate significant difference between porewater and surface water parameters; miniscule superscripts indicate significant difference among streams, per parameter (DOC 104 kb)
Rights and permissions
About this article
Cite this article
Packalen, M.S., Bagley, S.T. & McLaughlin, J.W. Peatland Stream Lipid Biogeochemistry Features in an Intermediate Fen Peatland, Ontario Canada. Wetlands 31, 353–365 (2011). https://doi.org/10.1007/s13157-010-0141-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-010-0141-8