Abstract
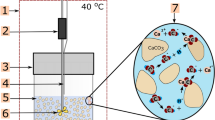
The carbonate–CO2–H2O interaction in a closed system under burial conditions was simulated in diamond anvil cell (DAC) with an in situ Raman spectroscopy. Experiments were performed in \({\text{H}}_{2} {\text{CO}}_{3}\) solution at acidic pH (≈ 5.6), 20–260 °C, and atmospheric pressure to about 400 MPa. Pressure was first solely increased to separately evaluate the pressure effect on carbonate dissolution/precipitation. Then temperature and pressure were increased together alike in the burial process, to monitor the dissolution/precipitation tendency of carbonate during burial. A continuous dissolution was observed for both limestone and dolostone when solely increasing pressure. A rapid dissolution and then continuous precipitation were observed when increasing temperatures and pressures simultaneously. These observations indicate that pressure has a positive effect (prograde solubility), while temperature has a negative effect (retrograde solubility) on CO2 solubility and carbonate dissolution. In case of low initial CO2 content as our experiments revealed, the increase in solid carbonate volume and a counterbalance decrease in porosity caused merely by increasing burial depth are minor (< 0.23%). The porosity decrease of dolostone was likely less than that of limestone at smaller burial depth (< 1.5 km), and notable at bigger burial depth. This work may help to improve our understanding on carbonate reservoir porosity evolution during burial.











Similar content being viewed by others
References
Alkattan M, Oelkers EH, Dandurand JL (1998) An experimental study of calcite and limestone dissolution rates as a function of pH from 1 to 3 and temperature from 25 to 80 C. Chem Geol 151(1–4):199–214. https://doi.org/10.1016/S0009-2541(98)00080-1
AlKattan M, Oelkers E, Dandurand J, Schott J (2002) An experimental study of calcite dissolution rates at acidic conditions and 25 C in the presence of NaPO3 and MgCl2. Chem Geol 190(1–4):291–302. https://doi.org/10.1016/S0009-2541(02)00121-3
Al-Khaldi MH, Nasr-El-Din HA, Mehta S, Al-Aamri AD (2007) Reaction of citric acid with calcite. Chim Eng Sci 62(21):5880–5896. https://doi.org/10.1016/j.ces.2007.06.021
Arvidson RS, Luttge A (2010) Mineral dissolution kinetics as a function of distance from equilibrium—new experimental results. Chem Geol 269:79–88. https://doi.org/10.1016/j.chemgeo.2009.06.009
Azbej T, Severs MJ, Rusk BG, Bodnar RJ (2007) In situ quantitative analysis of individual H2O–CO2 fluid inclusions by laser Raman spectroscopy. Chem Geol 237(3–4):255–263. https://doi.org/10.1016/j.chemgeo.2006.06.025
Bassett WA, Anderson AJ, Mayanovic RA, Chou IM (2000) Hydrothermal diamond anvil cell for XAFS studies of first-row transition elements in aqueous solution up to supercritical conditions. Chem Geol 167:3–10. https://doi.org/10.1016/S0009-2541(99)00196-5
Beavington-Penney SJ, Nadin P, Wright VP, Clarke E, McQquilken J, Bailey HW (2008) Reservoir quality variation in an Eocene carbonate ramp, El Garia formation, offshore Tunisia: structural control of burial corrosion and dolomitization. Sediment Geol 209:42–57. https://doi.org/10.1016/j.sedgeo.2008.06.006
Bénézeth P, Dandurand JL, Harrichoury JC (2009) Solubility product of siderite (FeCO3) as a function of temperature (25–250 °C). Chem Geol 265:3–12. https://doi.org/10.1016/j.chemgeo.2009.03.015
Bischoff JL, Rosenbauer RJ (1996) The alteration of rhyolite in CO2 charged water at 200 and 350 °C: the unreactivity of CO2 at higher temperature. Geochim Cosmochim Act 60(20):3859–3867. https://doi.org/10.1016/0016-7037(96)00208-6
Bjørlykke K, Jahren J (2012) Open or closed geochemical systems during diagenesis in sedimentary basins: constraints on mass transfer during diagenesis and the prediction of porosity in sandstone and carbonate reservoirs. Am Assoc Pet Geol Bull 96(12):2193–2214. https://doi.org/10.1306/04301211139
Davies GR, Smith JLB (2006) Structurally controlled hydrothermal dolomite reservoir facies: an overview. Am Assoc Pet Geol Bull 90(11):1641–1690. https://doi.org/10.1306/05220605164
Duan Z, Sun R (2003) An improved model calculating CO2 solubility in pure water and aqueous NaCl solutions from 273 to 533 K and from 0 to 2000 bar. Chem Geol 193(3–4):257–271. https://doi.org/10.1016/S0009-2541(02)00263-2
Ehrenberg SN (2006) Porosity destruction in carbonate platforms. J Petrol Geol 29:41–52. https://doi.org/10.1111/j.1747-5457.2006.00041.x
Ehrenberg SN, Nadeau PH (2005) Sandstone vs. carbonate petroleum reservoirs: a global perspective on porosity–depth and porosity–permeability relationships. Am Assoc Pet Geol Bull 89(4):435–445. https://doi.org/10.1306/11230404071
Ehrenberg SN, Nadeau PH, Steen Ø (2009) Petroleum reservoir porosity versus depth: influence of geological age. Am Assoc Pet Geol Bull 93(10):1281–1296. https://doi.org/10.1306/06120908163
Ehrenberg SN, Walderhaug O, Bjorlykke K (2012) Carbonate porosity creation by mesogenetic dissolution: reality or illusion? Am Assoc Pet Geol Bull 96(2):217–233. https://doi.org/10.1306/05031110187
Ehrenberg SN, Walderhaug O, Bjorlykke K (2013) Carbonate porosity creation by mesogenetic dissolution: reality or illusion? Reply. Am Assoc Pet Geol Bull 97(2):347–349. https://doi.org/10.1306/07111212089
Emerson S, Jahnke R, Bender M, Froelich P, Klinkhammer G, Bowser C, Setlock G (1980) Early diagenesis in sediments from the eastern equatorial Pacific, I. Pore water nutrient and carbonate results. Earth Planet Sci Lett 49(1):57–80. https://doi.org/10.1016/0012-821X(80)90150-8
Evelein KA, Moore RG, Heidemann RA (1976) Correlation of the phase behavior in the systems hydrogen sulfide–water and carbon dioxide–water. Ind Eng Chem Process Des Dev 15(3):423–428. https://doi.org/10.1021/i260059a013
Fein JB, Walther JV (1987) Calcite solubility in supercritical CO2–H2O fluids. Geochim Cosmochim Act 51(6):1665–1673. https://doi.org/10.1016/0016-7037(87)90346-2
Finneran DW, Morse JW (2009) Calcite dissolution kinetics in saline waters. Chem Geol 268(1–2):137–146. https://doi.org/10.1016/j.chemgeo.2009.08.006
Fredd CN, Fogler HS (1998) The kinetics of calcite dissolution in acetic acid solutions. Chem Eng Sci 53(22):3863–3874. https://doi.org/10.1016/S0009-2509(98)00192-4
Garcia-Rios M, Cama J, Luquot L, Soler JM (2014) Interaction between CO2-rich sulfate solutions and carbonate reservoir rocks from atmospheric to supercritical CO2 conditions: experiments and modeling. Chem Geol 383:107–122. https://doi.org/10.1016/j.chemgeo.2014.06.004
Gautelier M, Oelkers EH, Schott J (1999) An experimental study of dolomite dissolution rates as a function of pH from − 0.5 to 5 and temperature from 25 to 80 °C. Chem Geol 157:13–26. https://doi.org/10.1016/S0009-2541(98)00193-4
Gautelier M, Schott J, Oelkers EH (2007) An experimental study of dolomite dissolution rates at 80 °C as a function of chemical affinity and solution composition. Chem Geol 242(3–4):509–517. https://doi.org/10.1016/j.chemgeo.2007.05.008
Gledhill DK, Morse JW (2006) Calcite dissolution kinetics in Na–Ca–Mg–Cl brines. Geochim Cosmochim Act 70(23):5802–5813. https://doi.org/10.1016/j.gca.2006.03.024
Golubev SV, Bénézeth P, Schott J, Dandurand JL, Castillo A (2009) Siderite dissolution kinetics in acidic aqueous solutions from 25 to 100 °C and 0 to 50 atm pCO2. Chem Geol 265:13–19. https://doi.org/10.1016/j.chemgeo.2008.12.031
Goncharov AF, Crowhurst JC (2005) Pulsed laser Raman spectroscopy in the laser heated diamond anvil cell. Rev Sci Instrum 76(063905):1–5. https://doi.org/10.1063/1.1931205
Halley RB, Schmoker JW (1983) High-porosity cenozoic carbonate rocks of south Florida: progressive loss of porosity with depth. Am Assoc Pet Geol Bull 67(2):191–200
Huang S, Huang K, Zhang X, Liu L, Zhong Q (2009) Chemical thermodynamics foundation of retrograde solubility for carbonate: solution media related to CO2. J Chengdu Univ Tech 36(5):457–464
Lai X, Zhu F, Wu Y, Huang R, Wu X, Zhang Q, Yang K, Qin S (2014) New high-pressure polymorph of In2S3 with defect Th3P4-type structure. J Solid State Chem 210:155–159. https://doi.org/10.1016/j.jssc.2013.11.015
Liu Z, Dreybrodt W (1997) Dissolution kinetics of calcium carbonate minerals in H2O–CO2 solutions in turbulent flow: The role of the diffusion boundary layer and the slow reaction H2O + CO2 = H + + \({\text{HCO}}_{3}^{ - }\). Geochim Cosmochim Act 61(14):2879–2889. https://doi.org/10.1016/S0016-7037(97)00143-9
Montes-Hernandez G, Renard F, Geoffroy N, Charlet L, Pironon J (2007) Calcite precipitation from CO2–H2O–Ca(OH)2 slurry under high pressure of CO2. J Cryst Growth 308:228–236. https://doi.org/10.1016/j.jcrysgro.2007.08.005
Morse JW, Arvidson RS (2002) The dissolution kinetics of major sedimentary carbonate minerals. Earth-Sci Rev 58:51–84. https://doi.org/10.1016/S0012-8252(01)00083-6
Morse JW, Arvidson RS, Lüttge A (2007) Calcium carbonate formation and dissolution. Chem Rev 107(2):342–381. https://doi.org/10.1021/cr050358j
Pham VTH, Lu P, Aagaard P, Zhu C, Hellevang H (2011) On the potential of CO2–water–rock interactions for CO2 storage using a modified kinetic model. Int J Greenh Gas Control 5(4):1002–1015. https://doi.org/10.1016/j.ijggc.2010.12.002
Plummer LN, Parkhurst DL, Wigley TML (1979) Critical review of the kinetics of calcite dissolution and precipitation. In: Jenne E (ed) Chemical modeling—speciation, sorption, solubility and kinetics in aqueous systems. American Chemical Society, Washington, DC, pp 537–573. https://doi.org/10.1021/bk-1979-0093.ch025
Pokrovsky OS, Golubev SV, Schott J (2005) Dissolution kinetics of calcite, dolomite and magnesite at 25°C and 0 to 50 atm \({\text{p}}_{{{\text{CO}}_{2} }}\). Chem Geol 217(3–4):239–255. https://doi.org/10.1016/j.chemgeo.2004.12.012
Pokrovsky OS, Golubev SV, Schott J, Castillo A (2009) Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150°C and 1 to 55 atm \({\text{p}}_{{{\text{CO}}_{2} }}\): New constraints on CO2 sequestration in sedimentary basins. Chem Geol 265:20–32. https://doi.org/10.1016/j.chemgeo.2009.01.013
Rosenbauer RJ, Koksalan T, Palandri JL (2005) Experimental investigation of CO2–brine–rock interactions at elevated temperature and pressure: implications for CO2 sequestration in deep-saline aquifers. Fuel Process Tech 86(14–15):1581–1597. https://doi.org/10.1016/j.fuproc.2005.01.011
Saldi GD, Schott J, Pokrovsky OS, Oelkers EH (2010) An experimental study of magnesite dissolution rates at neutral to alkaline conditions and 150 and 200 C as a function of pH, total dissolved carbonate concentration, and chemical affinity. Geochim Cosmochim Act 74(22):6344–6356. https://doi.org/10.1016/j.gca.2010.07.012
Sayles FL (1980) The solubility of CaCO3 in seawater at 2 degrees C based upon in situ sampled pore water composition. Mar Chem 9(4):223–235. https://doi.org/10.1016/0304-4203(80)90026-2
Schmidt C, Ziemann MA (2000) In-situ Raman spectroscopy of quartz: a pressure sensor for hydrothermal diamond-anvil cell experiments at elevated temperatures. Am Min 85(11–12):1725–1734. https://doi.org/10.2138/am-2000-11-1216
Schmoker JW, Halley RB (1982) Carbonate porosity versus depth: a predictable relation for south Florida. Am Assoc Pet Geol Bull 66(12):2561–2570
Sharp WE, Kennedy GC (1965) The system CaO–CO2–H2O in the two-phase region calcite + aqueous solution. J Geol 73(2):391–403
Smith RL Jr, Fang Z (2009) Techniques, applications and future prospects of diamond anvil cells for studying supercritical water systems. J Supercrit Fluids 47(3):431–446. https://doi.org/10.1016/j.supflu.2008.10.015
Span R, Wagner W (1996) A new equation of state for carbon dioxide covering the fluid region from the triple-point temperature to 1100 K at pressures up to 800 MPa. J Phys Chem Ref Data 25(6):1509–1596. https://doi.org/10.1063/1.555991
Sterpenich J, Sausse J, Pironon J, Géhin A, Hubert G, Perfetti E, Grgic D (2009) Experimental ageing of oolitic limestones under CO2 storage conditions: petrographical and chemical evidence. Chem Geol 265:99–112. https://doi.org/10.1016/j.chemgeo.2009.04.011
Sun Q, Qin C (2011) Raman O–H stretching band of water as an internal standard to determine carbonate concentrations. Chem Geol 283(3–4):274–278. https://doi.org/10.1016/j.chemgeo.2011.01.025
Taberner C (2013) How important is the impact of burial corrosion on carbonate reservoirs? Learning’s from case studies. In: AAPG annual conferrence 2013, #1555780 (abstr.)
Talman SJ, Wiwchar B, Gunter WD, Scarge CM (1990) Dissolution kinetics of calcite in the H2O–CO2 system along the steam saturation curve to 210 °C. In: Spencer RJ, Chou IM (eds) Fluid–Mineral interactions: a tribute to H.P. Eugster Geochem Soc Spec Publ Series, vol 2, pp 41–55
Taylor KC, Al-Ghamdi A, Nasr-El-Din HA (2004a) Effect of additives on the acid dissolution rates of calcium and magnesium carbonates. SPE J 19:122–127
Taylor KC, Al-Ghamdi A, Nasr-El-Din HA (2004b) Measurement of acid reaction rates of a deep dolomitic gas reservoir. J Can Pet Technol 43(10):1–8
Testemale D, Dufaud F, Martinez I, Bénézeth P, Hazemann J, Schott J, Guyot F (2009) An X-ray absorption study of the dissolution of siderite at 300 bar between 50 °C and 100 °C. Chem Geol 259:8–16. https://doi.org/10.1016/j.chemgeo.2008.08.019
Wang T, Li Z (2005) Dissolution kinetics of granular calcium carbonate in concentrated aqueous sodium dichromate solution at pH 6.0–7.0 and 110–130 °C. J Colloid Interf Sci 281:130–135. https://doi.org/10.1016/j.jcis.2004.06.094
Wright VP, Harris P (2013) Carbonate dissolution and porosity development in the burial (Mesogenetic) environment. In: AAPG annual conferrence 2013, #1541663 (abstr.)
Wu X, Qin S, Dubrovinsky L (2011) Investigation into high-pressure behavior of MnTiO3: x-ray diffraction and Raman spectroscopy with diamond anvil cells. Geosci Front 2:107–114. https://doi.org/10.1016/j.gsf.2010.09.003
Xu M, Hu X, Knauss KG, Higgins SR (2010) Dissolution kinetics of calcite at 50–70 C: an atomic force microscopic study under near-equilibrium conditions. Geochim Cosmochim Act 74(15):4285–4297. https://doi.org/10.1016/j.gca.2010.04.066
Yang Y, Liu B, Qin S, Luo P, Gao J, He Y, Zhang X (2013) Dissolution response mechanism of the carbonate mineral with the increase of depth and its reservoir significance. Acta Sci Nat Univ Pekin 49(5):859–866. https://doi.org/10.13209/j.0479-8023.2013.114
Yang Y, Liu B, Qin S, Luo P, Zhang S, Zhou M, Shi K, Tian Y (2014) Re-recognition of deep carbonate dissolution based on the observation of in situ simulation experiment. Acta Sci Nat Univ Pekin 50(2):316–322. https://doi.org/10.13209/j.0479-8023.2014.048
Zhang R, Hu S, Zhang X, Yu W (2007) Dissolution kinetics of dolomite in water at elevated temperatures. Aquat Geochem 13(4):309–338. https://doi.org/10.1007/s10498-007-9022-z
Zhang X, Shi K, Liu B, Yang Y, Wang J (2014) Retention processes and porosity preservation in deep carbonate reservoirs. Geol Sci Tech Inform 33(2):80–85
Zheng H, Duan T, Sun Q (2012) In situ observation of \({\text{CH}}_{4} {-}{\text{H}}_{2} {\text{O}}\) binary system at high temperature and high pressure by diamond anvil cell and Raman spectroscopy. Exp Therm Fluid Sci 38:262–265. https://doi.org/10.1016/j.expthermflusci.2011.11.005
Acknowledgements
We thank Prof. Sijing Huang and Dr. Keke Huang at Chengdu University of Technology, for their data support related to their published papers, and this manuscript benefited a lot from discussion with them. We appreciate the help from Xiang Wu (Peking University) during the experiments. Special thanks are presented to two anonymous reviewers for their comments which obviously improved the manuscript. This work was jointly funded by the National Science and Technology Major Project (No. 2017ZX05005-003-005), and the National Natural Science Foundations of China (No. 41672123, No. 41572117, and No. 41272137).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Ma, Y., Liu, B. et al. In situ diamond anvil cell (DAC) experiments on equilibrated carbonate–CO2–H2O interaction under elevated temperature and pressure. Carbonates Evaporites 34, 1785–1798 (2019). https://doi.org/10.1007/s13146-019-00526-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13146-019-00526-y