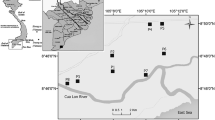
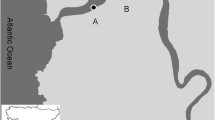
Abstract
Maturity index (MI), based on nematode life strategies, has been proposed in 1990 to assess the possible variations of the terrestrial and freshwater nematode assemblages induced by anthropogenic activities. It was subsequently applied also to marine ecosystems and, even if comparatively not yet very popular, it offers a good method to assess the ecological quality in relation to a wide range of anthropogenic drivers. However, few data are available on its response to physical stress, a key factor especially in the coastal areas. In this study, marine nematode genera from two study cases carried out in Maldives are used to test both MI and life strategy traits (i.e., c-p classes) for detecting the effects of physical disturbance. The results confirm that nematodes are well adapted to physical stress showing a general high rate of recovery. C-p scaling and MI did not seem to be appropriate for revealing this disturbance type probably because there are no empirical evidences on the life strategy of several genera, and a possible differential response to various disturbance types may be hypothesized.
Similar content being viewed by others
References
Altaff K, Sugumaran J, Naveed M S. 2005. Impact of tsunami on meiofauna of Marina beach, Chennai, India. Curr Sci, 89(1): 34–38
Balsamo M, Albertelli G, Ceccherelli V U, et al. 2010. Meiofauna of the Adriatic Sea: current state of knowledge and future perspectives. Chem Ecol, 26(1): 45–63
Balsamo M, Semprucci F, Frontalini F, et al. 2012. Chapter 4: Meiofauna as a tool for marine ecosystem biomonitoring. In: Cruzado A, ed. Marine Ecosystems. Rijeka: InTech Publisher, 77–104
Bongers T. 1990. The maturity index: an ecological measure of environmental disturbance based on nematode species composition. Oecologia, 83(1): 14–19
Bongers T, Alkemade R, Yeates G W. 1991. Interpretation of disturbanceinduced maturity decrease in marine nematode assemblages by means of the Maturity Index. Mar Ecol Prog Ser, 76(2): 135–142
Bongers T, Ferris H. 1999. Nematode community structure as a bioindicator in environmental monitoring. Trends in Ecology & Evolution, 14(6): 224–228
Boufahja F, Semprucci F. 2015. Stress-induced selection of a single species from an entire meiobenthic nematode assemblage: is this possible using iron enrichment and does pre-exposure affect the ease of the process?. Environ Sci Pollut Res, 22(3): 1979–1998
Buchanan J B. 1984. Sediment analysis. In: Holme N A, McIntyre A D, eds. Methods for the Study of Marine Benthos. 2nd ed. Oxford Boston: Blackwell Scientific Publications, 41–65
Clarke K R, Gorley R N. 2001. PRIMER v5: User Manual/Tutorial. Plymouth Marine Laboratory. Plymouth, UK: Primer-E Limited
Clarke K R, Warwick R M. 2001. Change in marine communities: an approach to statistical analysis and interpretation. 2nd ed. Plymouth Marine Laboratory, Plymouth, UK: Primer-E Limited
Essink K, Keidel H. 1998. Changes in estuarine nematode communities following a decrease of organic pollution. Aquat Ecol, 32(3): 195–202
Folk L R, Ward W C. 1957. Brazos river bar [Texas]; a study in the significance of grain size parameters. J Sediment Res, 27(1): 3–26
Fonsêca-Genevois V, Somerfield P J, Neves M H B, et al. 2006. Colonization and early succession on artificial hard substrata by meiofauna. Mar Biol, 148(5): 1039–1050
Fraschetti S, Gambi C, Giangrande A, et al. 2006. Structural and functional response of meiofauna rocky assemblages to sewage pollution. Mar Pollut Bull, 52(5): 540–548
Gingold R, Ibarra-Obando S E, Rocha-Olivares A. 2011. Spatial aggregation patterns of free-living marine nematodes in contrasting sandy beach micro-habitats. J Mar Biol Assoc UK, 91(3): 615–622
Grzelak K, Kotwicki L, Szczucinski W. 2009. Monitoring of sandy beach meiofaunal assemblages and sediments after the 2004 tsunami in Thailand. Pol J Environ St, 18(1): 43–51
Grzelak K, Szczucinski W, Kotwicki L, et al. 2014. Ecological status of sandy beaches after tsunami events: insights from meiofauna investigations after the 2011 Tohoku-oki Tsunami, Sendai Bay, Japan. In: Kontar Y A, Santiago-Fandiño V, Takahashi T, eds. Tsunami Events and Lessons Learned-Environmental and Societal Significance. Advances in Natural and Technological Hazards Research. Netherlands: Springer, 177–191
Kench P S, Brander R W, Parnell K E, et al. 2006. Wave energy gradients across a Maldivian atoll: implications for island geomorphology. Geomorphology, 81(1–2): 1–17
Kendall M A, Aryuthaka C, Chimonides J, et al. 2009. Post-tsunami recovery of shallow water biota and habitats on Thailand’s Andaman coast. Pol J Environ St, 18(1): 69–75
Kotwicki L, Szczucinski W. 2006. Meiofauna assemblages and sediment characteristic of sandy beaches on the west coast of Thailand after the 2004 tsunami event. Phuket Mar Biol Cent Res Bull, 67: 39–47
Lee H J, Vanhove S, Peck L S, et al. 2001. Recolonisation of meiofauna after catastrophic iceberg scouring in shallow Antarctic sediments. Polar Biol, 24(12): 918–925
Losi V, Montefalcone M, Moreno M, et al. 2012. Nematodes as indicators of environmental quality in seagrass (Posidonia oceanica) meadows of the NW Mediterranean Sea. Adv Oceanogra Limnol, 3(1): 69–91
Mirto S, La Rosa T, Gambi C, et al. 2002. Nematode community response to fish-farm impact in the western Mediterranean. Environ Pollut, 116(2): 203–214
Moreno M, Semprucci F, Vezzulli L, et al. 2011. The use of nematodes in assessing ecological quality status in the Mediterranean coastal ecosystems. Ecol Indic, 11(2): 328–336
Moreno M, Vezzulli L, Marin V, et al. 2008. The use of meiofauna diversity as an indicator of pollution in harbours. ICES J Mar Sci, 65(8): 1428–1435
Netto S A, Attrill M J, Warwick R M. 1999. The effect of a natural watermovement related disturbance on the structure of meiofauna and macrofauna communities in the intertidal sand flat of Rocas Atoll (NE, Brazil). J Sea Res, 42(4): 291–302
Nicholas W L, Hodda M. 1999. The free-living nematodes of a temperate, high energy, sandy beach: faunal composition and variation over space and time. Hydrobiologia, 394: 113–127
Perry C T, Kench P S, Smithers S G, et al. 2013. Time scales and modes of reef lagoon infilling in the Maldives and controls on the onset of reef island formation. Geology, 41(10): 1111–1114
Pfannkuche O, Thiel H. 1988. Sample processing. In: Higgins R P, Thiel H, eds. Introduction to the Study of Meiofauna. Washington: Smithsonian Inst, 134–145
Platt H M, Warwick R M. 1983. Free Living Marine Nematodes. Part I. British Enoplids. Synopses of the British Fauna (New Series) No. 28. Cambridge: Cambridge University Press
Platt H M, Warwick R M. 1988. Free living marine nematodes. Part II. British Chromadorids. Synopses of the British Fauna (New Series) No. 38. Leiden, Netherlands: E J Brill/W Backhuys
Raes M, De Troch M, Ndaro S G M, et al. 2007. The structuring role of microhabitat type in coral degradation zones: a case study with marine nematodes from Kenya and Zanzibar. Coral Reefs, 26(1): 113–126
Raes M, Vanreusel A. 2006. Microhabitat type determines the composition of nematode communities associated with sedimentclogged cold-water coral framework in the Porcupine Seabight (NE Atlantic). Deep Sea Research Part I, 53(12): 1880–1894
Sandulli R, Semprucci F, Balsamo M. 2014. Taxonomic and functional biodiversity variations of meiobenthic and nematode assemblages across an extreme environment: a study case in a Blue Hole cave. Ital J Zool, 81(4): 508–516
Schratzberger M, Bolam S G, Whomersley P, et al. 2004a. Development of a meiobenthic nematode community following the intertidal placement of various types of sediment. J Exp Mar Biol Ecol, 303(1): 79–96
Schratzberger M, Bolam S, Whomersley P, et al. 2006. Differential response o nematode colonist communities to the intertidal placement of dredged material. J Exp Mar Biol Ecol, 334(2): 244–255
Schratzberger M, Lampadariou N, Somerfield P J, et al. 2009. The impact of seabed disturbance on nematode communities: linking field and laboratory observations. Mar Biol, 156(4): 709–724
Schratzberger M, Rees H L, Boyd S E. 2000. Effects of simulated deposition of dredged material on structure of nematode assemblagesthe role of burial. Mar Biol, 136(3): 519–530
Schratzberger M, Whomersley P, Warr K, et al. 2004b. Colonisation of various types of sediment by estuarine nematodes via lateral infaunal migration: a laboratory study. Mar Biol, 145(1): 69–78
Seinhorst J W. 1959. A rapid method for the transfer of nematodes from fixative to anhydrous glycerin. Nematologica, 4(1): 67–69
Semprucci F, Balsamo M, Frontalini F. 2014a. The nematode assemblage of a coastal lagoon (Lake Varano, Southern Italy): ecology and biodiversity patterns. Sci Mar, 78(4): 579–588
Semprucci F, Boi P, Manti A, et al. 2010a. Benthic communities along a littoral of the Central Adriatic Sea (Italy). Helgol Mar Res, 64(2): 101–115
Semprucci F, Colantoni P, Baldelli G, et al. 2010b. The distribution of meiofauna on back-reef sandy platforms in the Maldives (Indian Ocean). Mar Ecol, 31(4): 592–607
Semprucci F, Colantoni P, Baldelli G, et al. 2013a. Meiofauna associated with coral sediments in the Maldivian subtidal habitats (Indian Ocean). Mar Biodiv, 43(3): 189–198
Semprucci F, Colantoni P, Sbrocca C, et al. 2011. Meiofauna in sandy back-reef platforms differently exposed to the monsoons in the Maldives (Indian Ocean). J Mar Syst, 87(3–4): 208–215
Semprucci F, Colantoni P, Sbrocca C, et al. 2014b. Spatial patterns of distribution of meiofaunal and nematode assemblages in the Huvadhoo lagoon (Maldives, Indian Ocean). J Mar Biol Assoc UK, 94(7): 1377–1385
Semprucci F, Frontalini F, Sbrocca C, et al. 2015a. Meiobenthos and free-living nematodes as tools for biomonitoring environments affected by riverine impact. Environ Monit Assess, 187: 251
Semprucci F, Losi V, Moreno M. 2015b. A review of Italian research on free-living marine nematodes and the future perspectives in their use as Ecological Indicators (EcoInd). Medit Mar Sci, 16(2): 352–365
Semprucci F, Moreno M, Sbrocca S. et al. 2013b. The nematode assemblage as a tool for the assessment of marine ecological quality status: a case-study in the Central Adriatic Sea. Medit Mar Sci, 14(1): 48–57
Soetaert K E R, Muthumbi A, Heip C H R. 2002. Size and shape of ocean margin nematodes: morphological diversity and depthrelated patterns. Mar Ecol Prog Ser, 242: 179–193
Somerfield P J, Dashfield S L, Warwick R M. 2007. Three-dimensional spatial structure: nematodes in a sandy tidal flat. Mar Ecol Prog Ser, 336: 177–186
Stoddart D R, Steers J A. 1977. The nature and origin of coral reef islands. In: Jones O A, Endean R, eds. Biology and Geology of Coral Reefs, vol 4. New York: Academic Press, 59–105
UNEP. 2005. After the tsunami: rapid environmental assessment. Nairobi: UNEP
Vanaverbeke J, Soetaert K, Vincx M. 2004. Changes in morphometric characteristics of nematode communities during a spring phytoplankton bloom deposition. Mar Ecol Prog Ser, 273: 139–146
Vanaverbeke J, Bezerra T N, Braeckman U, et al. 2015. NeMys: World Database of Free-Living Marine Nematodes. http://nemys. ugent.beon [2015-09-01]
Vanhove S, Vermeeren H, Vanreusel A. 2004. Meiofauna towards the South Sandwich Trench (750–6300 m), focus on nematodes. Deep Sea Research Part II, 51(14–16): 1665–1687
Vanreusel A, Fonseca G, Danovaro R, et al. 2010. The contribution of deep-sea macrohabitat heterogeneity to global nematode diversity. Mar Ecol, 31(1): 6–20
Warwick R M, Platt H M, Somerfield P J. 1998. Free-living Marine Nematodes: Monhysterids Part III (Synopses of the British Fauna), (New Series) No. 53. Shrewsbury, UK: Field Studies Council
Woodroffe C D, McLean R F, Smithers S G, et al. 1999. Atoll reef-island formation and response to sea-level change: West Island, Cocos (Keeling) Islands. Mar Geol, 160(1–2): 85–104
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Semprucci, F., Colantoni, P. & Balsamo, M. Is maturity index an efficient tool to assess the effects of the physical disturbance on the marine nematode assemblages?—A critical interpretation of disturbance-induced maturity successions in some study cases in Maldives. Acta Oceanol. Sin. 35, 89–98 (2016). https://doi.org/10.1007/s13131-016-0832-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13131-016-0832-y