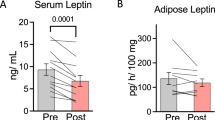
Abstract
The mechanisms underlying the relationships between nutritional status and immunity remain to be fully characterized. The present study was undertaken to analyze by flow cytometry, in the context of diet-induced obesity, the status of immune cells in subcutaneous, and epididymal fat depots in wild-type and immunodeficient Rag2−/− mice submitted to nutritional challenge, i.e., 48-h fasting and 1-week refeeding. In parallel, the responsiveness of mature adipocytes and immune cells in bone marrow, lymph node, and liver were also analyzed. The results show that fasting in obese wild-type mice induces a prominent lipolysis in epididymal AT and immunosuppression restricted to both subcutaneous and epididymal AT, characterized by reduced number of CD4+ T and B lymphocytes and M1/M2 macrophages associated with reduced leptin and increased FGF21 expression in mature adipocytes. One-week refeeding was sufficient to reverse the fasting-induced effects. Obese immunodeficient mice under nutritional challenge exhibited no changes in adipocyte leptin expression and no marked trafficking of AT macrophages or NK cells, while the fasted-induced upregulation of FGF21 expression was maintained as well as the lipolytic responses. The present results demonstrate that, in a context of diet-induced obesity, fasting-induced immunosuppression is restricted to fat depots in immunocompetent mice. Lack of adipocyte leptin regulation and fasting-induced immunosuppression in obese immunodeficient mice strongly suggests that lymphocytes are involved in the modulation of adipocyte leptin expression on one hand and on the other that leptin is involved in the immune changes in AT according to nutritional status.





Similar content being viewed by others
References
Ahima RS, Prabakaran D, Mantzoros C, Qu D, Lowell B, Maratos-Flier E, Flier JS (1996) Role of leptin in the neuroendocrine response to fasting. Nature 382:250–252
Ahren B, Mansson S, Gingerich RL, Havel PJ (1997) Regulation of plasma leptin in mice: influence of age, high-fat diet, and fasting. Am J Physiol 273:R113–R120
Asterholm IW, McDonald J, Blanchard PG, Sinha M, Xiao Q, Mistry J, Rutkowski JM, Deshaies Y, Brekken RA, Scherer PE (2012) Lack of “immunological fitness” during fasting in metabolically challenged animals. J Lipid Res 53:1254–1267
Ballak DB, Stienstra R, Hijmans A, Joosten LA, Netea MG, Tack CJ (2013) Combined B- and T-cell deficiency does not protect against obesity-induced glucose intolerance and inflammation. Cytokine+ 62:96–103
Behan JW, Ehsanipour EA, Sheng X, Pramanik R, Wang X, Hsieh YT, Kim YM, Mittelman SD (2013) Activation of adipose tissue macrophages in obese mice does not require lymphocytes. Obesity (Silver Spring) 21:1380–1388
de Jong AJ, Kloppenburg M, Toes RE, Ioan-Facsinay A (2014) Fatty acids, lipid mediators, and T-cell function. Front Immunol 5:483
Duffaut C, Galitzky J, Lafontan M, Bouloumie A (2009) Unexpected trafficking of immune cells within the adipose tissue during the onset of obesity. Biochem Biophys Res Commun 384:482–485
Duffaut C, Zakaroff-Girard A, Bourlier V, Decaunes P, Maumus M, Chiotasso P, Sengenes C, Lafontan M, Galitzky J, Bouloumie A (2009) Interplay between human adipocytes and T lymphocytes in obesity. CCL20 as an adipochemokine and T lymphocytes as lipogenic modulators. Arterioscler, Thromb, Vasc Biol 29:1608–1614
Faggioni R, Feingold KR, Grunfeld C (2001) Leptin regulation of the immune response and the immunodeficiency of malnutrition. Faseb J 15:2565–2571
Farooqi IS, Matarese G, Lord GM, Keogh JM, Lawrence E, Agwu C, Sanna V, Jebb SA, Perna F, Fontana S, Lechler RI, DePaoli AM, O’Rahilly S (2002) Beneficial effects of leptin on obesity, T cell hyporesponsiveness, and neuroendocrine/metabolic dysfunction of human congenital leptin deficiency. J Clin Invest 110:1093–1103
Frederich RC, Hamann A, Anderson S, Lollmann B, Lowell BB, Flier JS (1995) Leptin levels reflect body lipid content in mice: evidence for diet-induced resistance to leptin action. Nat Med 1:1311–1314
Howard JK, Lord GM, Matarese G, Vendetti S, Ghatei MA, Ritter MA, Lechler RI, Bloom SR (1999) Leptin protects mice from starvation-induced lymphoid atrophy and increases thymic cellularity in ob/ob mice. J Clin Invest 104:1051–1059
Huttunen R, Syrjanen J (2013) Obesity and the risk and outcome of infection. Int J Obes (Lond) 37:333–340
Kliewer SA, Mangelsdorf DJ (2010) Fibroblast growth factor 21: from pharmacology to physiology. Am J Clin Nutr 91:254S–257S
Kosteli A, Sugaru E, Haemmerle G, Martin JF, Lei J, Zechner R, Ferrante AW Jr (2010) Weight loss and lipolysis promote a dynamic immune response in murine adipose tissue. J Clin Invest 120:3466–3479
Lam QL, Wang S, Ko OK, Kincade PW, Lu L (2010) Leptin signaling maintains B-cell homeostasis via induction of Bcl-2 and cyclin D1. Proc Natl Acad Sci U S A 107:13812–13817
Lee MJ, Fried SK (2009) Integration of hormonal and nutrient signals that regulate leptin synthesis and secretion. Am J Physiol Endocrinol Metab 296:E1230–E1238
Lolmede K, Duffaut C, Zakaroff-Girard A, Bouloumie A (2011) Immune cells in adipose tissue: key players in metabolic disorders. Diabetes Metab 37:283–290
Lord GM, Matarese G, Howard JK, Baker RJ, Bloom SR, Lechler RI (1998) Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 394:897–901
Papathanassoglou E, El-Haschimi K, Li XC, Matarese G, Strom T, Mantzoros C (2006) Leptin receptor expression and signaling in lymphocytes: kinetics during lymphocyte activation, role in lymphocyte survival, and response to high fat diet in mice. J Immunol 176:7745–7752
Park HK, Ahima RS (2015) Physiology of leptin: energy homeostasis, neuroendocrine function and metabolism. Metabolism 64:24–34
Saladin R, De Vos P, Guerre-Millo M, Leturque A, Girard J, Staels B, Auwerx J (1995) Transient increase in obese gene expression after food intake or insulin administration. Nature 377:527–529
Schaible UE, Kaufmann SH (2007) Malnutrition and infection: complex mechanisms and global impacts. PLoS Med 4:e115
Szkudelski T (2007) Intracellular mediators in regulation of leptin secretion from adipocytes. Physiol Res 56:503–512
Tian Z, Sun R, Wei H, Gao B (2002) Impaired natural killer (NK) cell activity in leptin receptor deficient mice: leptin as a critical regulator in NK cell development and activation. Biochem Biophys Res Commun 298:297–302
Zhang Y, Guo KY, Diaz PA, Heo M, Leibel RL (2002) Determinants of leptin gene expression in fat depots of lean mice. Am J Physiol Regul Integr Comp Physiol 282:R226–R234
Acknowledgments
This work was supported by grants from the Agence Nationale de la Recherche (ANR, Transflora). The authors gratefully thank the GenoToul Animal Care Facility of Rangueil Hospital (UMS US006/Inserm) and M. Girma-Vidal for the excellent assistance for adipocyte RNA extraction and real-time PCR experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lolmède, K., Zakaroff-Girard, A., Dray, C. et al. Interrelationship between lymphocytes and leptin in fat depots of obese mice revealed by changes in nutritional status. J Physiol Biochem 71, 497–507 (2015). https://doi.org/10.1007/s13105-015-0388-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-015-0388-x