Abstract
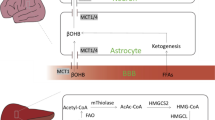
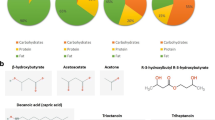
Ketogenesis is the branch of mammalian metabolism concerned with the synthesis of ketone bodies. In this process, the small, water-soluble compounds acetoacetate, D-3-β-hydroxybutyrate and propanone are produced by the liver in response to reduced glucose availability. Although ketone bodies are always present at a low level in healthy individuals, dietary manipulation and certain pathological conditions can increase the levels of these compounds in vivo. In some instances, such as in refractory epilepsy, high levels of ketone bodies can be beneficial—in this instance, by exerting an anticonvulsant effect. Conversely, if the levels of ketones rise to supraphysiological levels, as can occur in diabetes mellitus, a state of ketoacidosis can occur, which has serious consequences for cellular function. More recently, research has identified a possible link between ketogenesis and free radical-mediated pathologies, highlighting the potential application of ketogenic diets to the treatment of conditions such as Alzheimer's disease. Overall, an understanding of ketone body metabolism and its links to human disease may prove to be vital in developing new regimens for the treatment of human disease.





Similar content being viewed by others
Notes
Decarboxylation is also possible via. the cytosolic enzyme acetoacetate decarboxylase.
Glutathione (γ-glutamylcysteinylglycine) has the largest negative reduction potential found in vivo (E°′ = −1.5 V) making it the most potent naturally occuring antioxidant in biological systems. See Ref. [4] for an excellent treatment of the thermodynamics of reduction by glutathione.
References
Abete I, Astrup A, Martínez JA, Thorsdottir I, Zulet MA (2010) Obesity and the metabolic syndrome: role of different dietary macronutrient distribution patterns and specific nutritional components on weight loss and maintenance. Nutr Rev 68:214–231
Bough KJ, Rho JM (2007) Anticonvulsant mechanisms of the ketogenic diet. Epilepsia 48:43–58
Bravata DM, Sanders L, Huang J, Krumholz HM, Olkin I, Gardner CD (2003) Efficacy and safety of low-carbohydrate diets: a systematic review. JAMA 289:1837–1850
Buettner GR (1993) The pecking order of free radicals and antioxidants: lipid peroxidation, α-tocopherol, and ascorbate. Arch Biochem Biophys 300:535–543
Cahill CF (1976) Starvation in man. Clin Endocrinol Metab 5:398–417
Chen TY, Smith W, Rosenstock JL, Lessnau KD (2006) A life-threatening complication of Atkins diet. Lancet 367:958
Crujeiras AB, Parra D, Goyenechea E, Abete I, González-Muniesa P, Martínez JA (2008) Energy restriction in obese subjects impact differently two mitochondrial function markers. J Physiol Biochem 64:211–219
Dashti HM, Al-Zaid NS, Mathew TC, Al-Mousawi M, Talib H, Asfar SK, Behbahani AI (2006) Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol Cell Biochem 286:1–9
Denke MA (2001) Metabolic effects of high-protein, low-carbohydrate diets. Am J Cardiol 88:59–61
Devlin TM (1997) Textbook of biochemistry with clinical correlations. Wiley, New York
Eastmond PJ, Graham IA (2001) Re-examining the role of the glyoxylate cycle in oilseeds. Trends Plant Sci 6:72–77
Eaton S, Bartlett K, Pourfarzam M (1996) Mammalian mitochondrial β-oxidation. Biochem J 320:345–357
Erlanson-Albertsson C, Mei J (2005) The effect of low carbohydrate on energy metabolism. Int J Obes (Lond) 29(Suppl 2):S26–S30
Fehm HL, Kern W, Peters A (2006) The selfish brain: competition for energy resources. Prog Brain Res 153:129–140
Fenselau WK (1974) Acetoacetate substrate inhibition of CoA transferase from various rat tissues. Life Sci 15:811–818
Fernandez ML, West KL (2005) Mechanisms by which dietary fatty acids modulate plasma lipids. J Nutr 135:2075–2078
Garber J, Menzel PH, Boden G, Owen OE (1974) Hepatic ketogenesis and gluconeogenesis in humans. J Clin Invest 54:981–989
Haces ML, Hernandez-Fonseca K, Medina-Campos ON, Montiel T, Pedraza-Chaverri J, Massieu L (2008) Antioxidant capacity contributes to protection of ketone bodies against oxidative damage induced during hypoglycemic conditions. Exp Neurol 211:85–96
Halliwell B, Gutteridge JMC (1984) Lipid peroxidation, oxygen radicals, cell damage and antioxidant therapy. Lancet I:1396
Hardie DG (1992) Regulation of fatty acid and cholesterol metabolism by the AMP-activated protein kinase. Biochem et Biophys Acta 1123:231–238
Holleman MAF (1904) Notice sur l'action de l'eau oxygénée sur les acides α-cétoniques et sur les dicétones. Rec Trav Chim Pays Bas Belg 23:169–171
Huttenlocher PR, Wilbourn AJ, Signore JM (1971) Medium-chain triglycerides as a therapy for intractable childhood epilepsy. Neurology 21:1097–1103
Jain SK, Kannan K, Lim G (1998) Ketosis (acetoacetate) can generate oxygen radicals and cause increased lipid peroxidation and growth inhibition in human endothelial cells. Free Radic Biol Med 25:1083–1088
Jomova K, Vondrakova D, Lawson M, Valko M (2010) Metals, oxidative stress and neurodegenerative disorders. Mol Cell Biochem 345:91–104
Larsen TM, Dalskov SM, van Baak M, Jebb SA, Papadaki A, Pfeiffer AF, Martinez JA, Handjieva-Darlenska T, Kunešová M, Pihlsgård M, Stender S, Holst C, Saris WH, Astrup A (2010) Diets with high or low protein content and glycemic index for weight-loss maintenance. N Engl J Med 363:2102–2113
Lemastersg JJ, Grunwald R, Emaus RK (1984) Thermodynamic limits to the ATP/site stoichiometries of oxidative phosphorylation by rat liver mitochondria. JBC 259:3058–3063
Liu YM (2008) Medium-chain triglyceride (MCT) ketogenic therapy. Epilepsia 49:33–36
MacDonald A, Webber J (1995) Feeding, fasting and starvation: factors affecting fuel utilization. Proc Nutr Soc 54:267–274
Martínez JA, Parra MD, Manninen AH, Hilton PJ, McKinnon W (2006) Life-threatening complications of the Atkins diet? Lancet 368:23–24
Masino SA, Geiger JD (2008) Are purines mediators of the anticonvulsant/neuroprotective effects of ketogenic diets? Trends Neurosci 31:273–278
McCall AL (2004) Cerebral glucose metabolism in diabetes mellitus. Eur J Pharmacol 490:147–158
Mogilevskaya E, Demin O, Goryanin I (2006) Kinetic model of mitochondrial Krebs cycle: unraveling the mechanism of salicylate hepatotoxic effects. J Biol Phys 32:245–271
Pan A, Hu FB (2011) Effects of carbohydrates on satiety: differences between liquid and solid food. Curr Opin Clin Nutr Metab Care 14:385–390
Pifferi F, Tremblay S, Croteau E, Fortier M, Tremblay-Mercier J, Lecomte R, Cunnane SC (2011) Mild experimental ketosis increases brain uptake of 11C-acetoacetate and 18F-fluorodeoxyglucose: a dual-tracer PET imaging study in rats. Nutr Neurosci 14:51–58
Porta N, Vallee L, Boutry E, Fontaine M, Dessein AF, Joriot S, Cuisset JM, Cuvellier JC, Auvin S (2009) Comparison of seizure reduction and serum fatty acid levels after receiving the ketogenic and modified Atkins diet. Seizure 18:359–364
Rutter GA (2000) Diabetes: the importance of the liver. Curr Biol 10:R736–R738
Serra D, Casals N, Asins G, Royo T, Ciudad CJ, Hegardt FG (1993) Regulation of mitochondrial 3-hydroxy-3-methylglutarylcoenzyme A synthase protein by starvation, fat feeding and diabetes. Arch Biochem Biophys 307:40–45
Simpson IA, Carruthers A, Vannucci SJ (2007) Supply and demand in cerebral energy metabolism: the role of nutrient transporters. J Cereb Blood Flow Metab 27:1766–1791
Sumithran P, Proietto J (2008) Ketogenic diets for weight loss: a review of their principles, safety and efficacy. Obes Res Clin Pract 2:1–13
Temkin O (1971) Falling sickness: history of epilepsy from the Greeks to the beginnings of modern neurology, 2nd edn. Johns Hopkins, USA
Uppu RM, Pryor WA (1996) Carbon dioxide catalysis of the reaction of peroxynitrite with ethyl acetoacetate: an example of aliphatic nitration by peroxynitrite. Biochem Biophys Res Comm 229:764–769
Van der Auwera I, Wera S, Van Leuven F, Henderson ST (2005) A ketogenic diet reduces amyloid beta 40 and 42 in a mouse model of Alzheimer's disease. Nutr Metab (Lond) 17:2–28
Volek JS, Sharman MJ (2004) Cardiovascular and hormonal aspects of very-low-carbohydrate ketogenic diets. Obes Res 12(Suppl 2):115S–123S
Wagenmakers AJ (1998) Protein and amino acid metabolism in human muscle. Adv Exp Med Biol 441:307–319
Wang S, Soni KG, Semache M, Casavant S, Fortier M, Pan L, Mitchell AG (2008) Lipolysis and the integrated physiology of lipid energy metabolism. Mol Genet Metab 95:117–126
Wasserman DH (2009) Four grams of glucose. Am J Physiol Endocrinol Metab 296:E11–E21
Watt MJ, Steinberg GR (2008) Regulation and function of triacylglycerol lipases in cellular metabolism. Biochem J 414:313–325
White AM, Johnston CS, Swan PD, Tjonn SL, Sears B (2007) Blood ketones are directly related to fatigue and perceived effort during exercise in overweight adults adhering to low-carbohydrate diets for weight loss: a pilot study. J Am Diet Assoc 107:1792–1796
Yao J, Chen S, Mao Z, Cadenas E, Brinton RD (2011) 2-Deoxy-d-glucose treatment induces ketogenesis, sustains mitochondrial function, and reduces pathology in female mouse model of Alzheimer's disease. PLoS One 6:e21788
Young C, Gean PW, Chiou LC, Shen YZ (2000) Docosahexaenoic acid inhibits synaptic transmission and epileptiform activity in the rat hippocampus. Synapse 37:90–94
Young IS, Woodside JV (2001) Antioxidants in health and disease. J Clin Pathol 54:176–186
Yudkoff M, Daikhin Y, Horyn O, Lazarow A, Luhovyy B, Wehrli S, Nissim I (2005) Response of brain amino acid metabolism to ketosis. Neurochem Int 47:119–128
Yudkoff M, Daikhin Y, Nissim I, Grunstein R (1997) Effects of ketone bodies on astrocyte amino acid metabolism. J Neurochem 69:685–692
Zammi V (1994) Regulation of ketone body metabolism. Diabetes Reviews 132–155
Acknowledgements
This work was partially supported by the Northern Ireland R&D Office (Extension to RRG 5.42). The authors would like to thank Dr DJ Timson, School of Biological Sciences, Queen's University Belfast, for useful discussions of mammalian metabolism.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McPherson, P.A.C., McEneny, J. The biochemistry of ketogenesis and its role in weight management, neurological disease and oxidative stress. J Physiol Biochem 68, 141–151 (2012). https://doi.org/10.1007/s13105-011-0112-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-011-0112-4