Abstract
Aim
To investigate the association of matrix metalloproteinase-3 (MMP-3) polymorphism with susceptibility to coronary artery stenosis (CAS) and the number of diseased vessels in patients with type 2 diabetes mellitus (T2DM).
Methods
The study population comprised 618 unrelated Iranian individual subjects, including 305 angiographically documented CAS patients with T2DM and 313 control subjects with T2DM. MMP3 genotypes were determined by polymerase chain reaction and restriction fragment length polymorphism.
Results
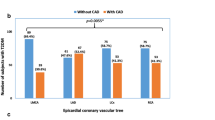
Significant differences between cases and controls were observed for MMP3 genotype frequencies (p < 0.01). The 6A allele was high frequently seen in the disease group, compared with the control group (64.75 vs. 56.24%, 6A/6A + 5A/6A vs. 5A/5A, p < 0.05). The association of this polymorphism with the severity of stenosis were also evaluated which according to results distribution of MMP3 genotypes were not significantly different as compared with the severity of stenosis (p > 0.05).
Conclusions
Frequency of the 6A allele of the human MMP3 gene is an independent risk factor for CAS in the Iranian T2DM studied.
Similar content being viewed by others
References
Beyzade S, Zhang S, Wong YK, Day IN, Eriksson P, Ye S (2003) Influences of matrix metalloproteinase-3 gene variation on extent of coronary atherosclerosis and risk of myocardial infarction. J Am Coll Cardiol 41:2130–2137
Bini A, Mann KG, Kudryk BJ, Schoen FJ (1999) Noncollagenous bone matrix proteins, calcification, and thrombosis in carotid artery atherosclerosis. Arterioscler Thromb Vasc Biol 19:1852–1861
Borghaei RC, Sullivan C, Mochan E (1999) Identification of a cytokineinduced repressor of interleukin-1 stimulated expression of stromelysin 1 (MMP-3). J Biol Chem 274:2126–2131
de Maat MP, Jukema JW, Ye S et al (1999) Effect of the stromelysin-1 promoter on efficacy of pravastatin in coronary atherosclerosis and restenosis. Am J Cardiol 83:852–856
Friedewald WT, Levi IR, Fredrickson DS (1972) Estimation of low density lipoprotein cholesterol levels in plasma without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Galis ZS, Khatri JJ (2002) Matrix metalloproteinases in vascular remodeling and atherogenesis: the good, the bad, and the ugly. Circ Res 90:251–262
Galis ZS, Sukhova GK, Lark MW, Libby P (1994) Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J Clin Invest 94:2493–2503
Gensini GF, Comeglio M, Colella A (1998) Classical risk factors and emerging elements in the risk profile for coronary artery disease. Eur Heart J 19:53–61
Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ (1987) Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med 316:1371–1375
Henney AM, Wakeley PR, Davies MJ et al (1991) Localization of stromelysin gene expression in atherosclerotic plaques by in situ hybridization. Proc Natl Acad Sci 88:8154–8158
Hirashiki A, Yamada Y, Murase Y, Suzuki Y, Kataoka H, Morimoto Y, Tajika T, Murohara T, Yokota M (2003) Association of gene polymorphisms with coronary artery disease in low- or high-risk subjects defined by conventional risk factors. J Am Coll Cardiol 42:1429–1437
Humphries SE, Luong LA, Talmud PJ et al (1998) The 5A/6A polymorphism in the promoter of the stromelysin-1 (MMP-3) gene predicts progression of angiographically determined coronary artery disease in men in the LOCAT gemfibrozil study. Lopid Coronary Angiography Trial. Atherosclerosis 139:49–56
Irace C, Cortese C, Migale M et al (2008) Stromelysin gene promoter polymorphism and common carotid geometry in diabetic subjects. Int Angiol 27(5):413–418
Kim JS, Park HY, Kwon JH, Im EK, Choi D, Jang Y, Cho SY (2002) The roles of stromelysin-1 and the gelatinase B gene polymorphism in stable angina. Yonsei Med J 43:473–481
Kirstein M, Sanz L, Quinones S, Moscat J, Diaz-Meco MT, Saus J (1996) Cross-talk between different enhancer elements during mitogenic induction of the human stromelysin-1 gene. J Biol Chem 271:18231–18236
Liu PY, Chen JH, Li YH, Wu HL, Shi GY (2003) Synergistic effect of stromelysin-1 (matrix metallo-proteinase-3) promoter 5A/6A polymorphism with smoking on the onset of young acute myocardial infarction. Thromb Haemost 90:132–139
Marenberg ME, Risch N, Berkman LF, Floderus B, de Faire U (1994) Genetic susceptibility to death from coronary heart disease in a study of twins. N Engl J Med 330:1041–1046
Matrisian LM (1990) Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet 6:121–125
Medley TL, Kingwell BA, Gatzka CD, Pillay P, Cole TJ (2003) Matrix metalloproteinase-3 genotype contributes to age-related aortic stiffening through modulation of gene and protein expression. Circ Res 92:1254–1261
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215–1219
Murphy G, Reynolds JJ, Royce PM, Steinman B (1993) Extracellular matrix degradation. In: Connective tissue and its heritable disorders. Wiley-Liss, New York, pp 287–316
Murphy G, Ward R, Gavrilovic J, Atkinson S (1992) Physiological mechanism for metalloproteinase activation. Matrix suppl 1:224–230
Nojiri T, Morita H, Imai Y et al (2003) Genetic variations of matrix metalloproteinase-1 and-3 promoter regions and their associations with susceptibility to myocardial infarction in Japanese. Int J Cardiol 92:181–186
Oceandy D, Yusoff R, Baudoin FM, Neyses L, Ray SG (2007) Promoter polymorphism of the matrix metalloproteinase 3 gene is associated with regurgitation and left ventricular remodeling in mitral valve prolapse patients. European Journal of Heart Failure 9:1010–1017
Polak JF, Kronmal RA, Tell GS, O’Leary DH, Savage PJ, Gardin JM, Rutan GH, Borhani NO, on behalf of the Cardiovascular Health Study (1996) Compensatory increase in common carotid artery diameter: relation to blood pressure and artery intima-media thickness in older adults. Stroke 27:2012–2015
Pollanen PJ, Lehtimaki T, Ilveskoski E et al (2002) Coronary artery calcification is related to functional polymorphism of matrix metalloproteinase: 3. The Helsinki Sudden Death Study. Atherosclerosis 164:329–335
Quinones S, Buttice G, Kurkinen M (1994) Promoter elements in the transcriptional activation of the human stromelysin-1 gene by the inflammatory cytokine, interleukin 1. Biochem J 302(Pt 2):471–477
Quinones S, Saus J, Otani Y, Harris ED Jr, Kurkinen M (1989) Transcriptional regulation of human stromelysin. J Biol Chem 264:8339–8344
Schwarz A, Haberbosch W, Tillmanns H, Gardemann A (2002) The stromelysin-1 5A/6A promoter polymorphism is a disease marker for the extent of coronary heart disease. Dis Markers 18:121–128
Sirum-Connolly K, Brinckerhoff CE (1991) Interleukin-1 or phorbol induction of the stromelysin promoter requires an element that cooperates with AP-1. Nucleic Acids Res 19:335–341
Steinke W, Els T, Hennerici M (1994) Compensatory carotid artery dilation in atherosclerosis. Circulation 89:2578–2581
Terashima M, Akita H, Kanazawa K et al (1999) Stromelysin promoter 5A/6A polymorphism is associated with acute myocardial infarction. Circulation 99:2717–2719
Woessner JF Jr (1991) Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J 5:2145–2154
Ye S, Eriksson P, Hamsten A, Kurkinen M, Humphries SE, Henney AM (1996) Progression of coronary atherosclerosis is associated with a common genetic variant of the human stromelysin-1 promoter, which results in reduced gene expression. J Biol Chem 271:13055–13060
Ye S, Watts GF, Mandalia S, Humphries SE, Henney AM (1995) Preliminary report: genetic variation in the human stromelysin promoter is associated with progression of coronary atherosclerosis. Br Heart J 73:209–215
ZhuC OJ, HamstenA EP (2006) Allele-specific MMP-3 transcription under in vivo conditions. Biochem Biophys Res Commun 348:1150–1156
Acknowledgments
This study was supported in part by a grant from the Iran University of Medical Sciences. The authors wish to thank Gifted and Talented Students Center of University for funding of this project and thank Shahid Rajaee and Shahid Madani Hospitals in the north of Tehran and Tabriz, respectively, for providing the samples.
Study limitation
There were several limitations for our study. Firstly, this was a small study, and these observations must be confirmed in a larger sample of patients. Nevertheless, a large-scale prospective study should be launched to confirm the associative effect of this polymorphism with CAS T2DM patients. Secondly, we analyzed only one polymorphism in most of the genes, which could have missed an association that was to a specific polymorphism. It will be interesting also to see if this polymorphism has a role in the manifestation and development of other coronary artery disease such as myocardial infarction, coronary aneurysm, etc...in patients with T2DM.
Conflict of interest statement
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Grant support
This study was supported in part by a grant from the Medical Sciences of Iran University.
Rights and permissions
About this article
Cite this article
Fallah, S., Seifi, M. & Samadikuchaksaraei, A. Risk of coronary artery stenosis in Iranian type 2 diabetics: is there a role for matrix metalloproteinase-3 gene (-1612 5A/6A) polymorphism?. J Physiol Biochem 66, 359–364 (2010). https://doi.org/10.1007/s13105-010-0042-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-010-0042-6