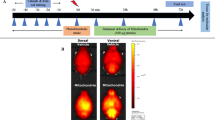
Abstract
Reactive oxygen species (ROS) exacerbate stroke-induced cell damage. We found that ShcA, a protein that regulates ROS, is highly expressed in a Rose Bengal photothrombosis model. We investigated whether ShcA is essential for mitophagy in ROS-induced cellular damage and determined whether ROS exacerbate mitochondrial dysfunction via ShcA protein expression. Ischemic stroke was generated by Rose Bengal photothrombosis in mice. To silence ShcA protein expression in the mouse brain, ShcA-targeting siRNA-encapsulated nanoparticles were intrathecally injected into the cisterna magna. Upon staining with antibodies against ShcA counterpart caspase-3 or NeuN, we found that the ShcA protein expression was increased in apoptotic neurons. In addition, mitochondrial dysfunction and excessive mitophagy were evident in photothrombotic stroke tissue. Infarct volumes were significantly reduced, and neurological deficits were diminished in the ShcA siRNA nanoparticle-treated group, compared with the negative control siRNA nanoparticle-treated group. We confirmed that the reduction of ShcA expression by nanoparticle treatment rescued the expression of genes, associated with mitochondrial dynamics and mitophagy mediation in a stroke model. This study suggests that the regulation of ShcA protein expression can be a therapeutic target for reducing brain damage with mitochondrial dysfunction caused by thrombotic infarction.






Similar content being viewed by others
References
Poli G, Leonarduzzi G, Biasi F, Chiarpotto E. Oxidative stress and cell signalling. Curr Med Chem. 2004;11(9):1163–82.
Chen K, Thomas SR, Keaney JF Jr. Beyond LDL oxidation: ROS in vascular signal transduction. Free Radic Biol Med. 2003;35(2):117–32.
Liu H, Colavitti R, Rovira II, Finkel T. Redox-dependent transcriptional regulation. Circ Res. 2005;97(10):967–74.
Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Phys Lung Cell Mol Phys. 2000;279(6):L1005–28.
Briones AM, Touyz RM. Oxidative stress and hypertension: current concepts. Curr Hypertens Rep. 2010;12(2):135–42.
Cai H, Harrison DG. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res. 2000;87(10):840–4.
Lyle AN, Griendling KK. Modulation of vascular smooth muscle signaling by reactive oxygen species. Physiology (Bethesda). 2006;21:269–80.
Crack PJ, Taylor JM. Reactive oxygen species and the modulation of stroke. Free Radic Biol Med. 2005;38(11):1433–44.
Gursoy-Ozdemir Y, Can A, Dalkara T. Reperfusion-induced oxidative/nitrative injury to neurovascular unit after focal cerebral ischemia. Stroke. 2004;35(6):1449–53.
Nelson CW, Wei EP, Povlishock JT, Kontos HA, Moskowitz MA. Oxygen radicals in cerebral ischemia. Am J Phys. 1992;263(5 Pt 2):H1356–62.
Fagiani E, Giardina G, Luzi L, Cesaroni M, Quarto M, Capra M, et al. RaLP, a new member of the Src homology and collagen family, regulates cell migration and tumor growth of metastatic melanomas. Cancer Res. 2007;67(7):3064–73.
Okada S, Kao AW, Ceresa BP, Blaikie P, Margolis B, Pessin JE. The 66-kDa Shc isoform is a negative regulator of the epidermal growth factor-stimulated mitogen-activated protein kinase pathway. J Biol Chem. 1997;272(44):28042–28,049.
Pelicci G, Lanfrancone L, Grignani F, McGlade J, Cavallo F, Forni G, et al. A novel transforming protein (SHC) with an SH2 domain is implicated in mitogenic signal transduction. Cell. 1992;70(1):93–104.
Jones N, Hardy WR, Friese MB, Jorgensen C, Smith MJ, Woody NM, et al. Analysis of a Shc family adaptor protein, ShcD/Shc4, that associates with muscle-specific kinase. Mol Cell Biol. 2007;27(13):4759–73.
Nakamura T, Muraoka S, Sanokawa R, Mori N. N-Shc and Sck, two neuronally expressed Shc adapter homologs. Their differential regional expression in the brain and roles in neurotrophin and Src signaling. J Biol Chem. 1998;273(12):6960–7.
Conti L, Sipione S, Magrassi L, Bonfanti L, Rigamonti D, Pettirossi V, et al. Shc signaling in differentiating neural progenitor cells. Nat Neurosci. 2001;4(6):579–86.
Cataudella T, Conti L, Cattaneo E. Neural stem and progenitor cells: choosing the right Shc. Prog Brain Res. 2004;146:127–33.
Wary KK, Mainiero F, Isakoff SJ, Marcantonio EE, Giancotti FG. The adaptor protein Shc couples a class of integrins to the control of cell cycle progression. Cell. 1996;87(4):733–43.
Stevenson LE, Ravichandran KS, Frackelton AR Jr. Shc dominant negative disrupts cell cycle progression in both G0-G1 and G2-M of ErbB2-positive breast cancer cells. Cell Growth Differ. 1999;10(1):61–71.
Migliaccio E, Giorgio M, Mele S, Pelicci G, Reboldi P, Pandolfi PP, et al. The p66shc adaptor protein controls oxidative stress response and life span in mammals. Nature. 1999;402(6759):309–13.
Talley Watts L, Zheng W, Garling RJ, Frohlich VC, Lechleiter JD. Rose Bengal photothrombosis by confocal optical imaging in vivo: a model of single vessel stroke. J Vis Exp. 2015;100:e52794.
Yang G, Chan PH, Chen J, Carlson E, Chen SF, Weinstein P, et al. Human copper-zinc superoxide dismutase transgenic mice are highly resistant to reperfusion injury after focal cerebral ischemia. Stroke. 1994;25(1):165–70.
Shibata M, Yamasaki N, Miyakawa T, Kalaria RN, Fujita Y, Ohtani R, et al. Selective impairment of working memory in a mouse model of chronic cerebral hypoperfusion. Stroke. 2007;38(10):2826–32.
Noh MY, Koh SH, Kim SM, Maurice T, Ku SK, Kim SH. Neuroprotective effects of donepezil against Aβ42-induced neuronal toxicity are mediated through not only enhancing PP2A activity but also regulating GSK-3β and nAChRs activity. J Neurochem. 2013;127:562–74.
Shin J, Yin Y, Park H, Park S, Triantafillu UL, Kim Y, et al. p38 siRNA-encapsulated PLGA nanoparticles alleviate neuropathic pain behavior in rats by inhibiting microglia activation. Nanomedicine (London). 2018;13(13):1607–21.
Carmichael ST. Rodent models of focal stroke: size, mechanism, and purpose. NeuroRx. 2005;2(3):396–409.
Reers M, Smith TW, Chen LB. J-Aggregate formation of a carbocyanine as a quantitative fluorescent indicator of membrane potential. Biochemistry. 1991;30(18):4480–6.
Di Lisa F, Blank PS, Colonna R, Gambassi G, Silverman HS, Stern MD, et al. Mitochondrial membrane potential in single living adult rat cardiac myocytes exposed to anoxia or metabolic inhibition. J Physiol. 1995;486(Pt 1):1–13.
Menon JU, Kuriakose A, Iyer R, Hernandez E, Gandee L, Zhang S, et al. Dual-drug containing core-shell nanoparticles for lung cancer therapy. Sci Rep. 2017;7(1):13249.
Watson BD, Dietrich WD, Busto R, Wachtel MS, Ginsberg MD. Induction of reproducible brain infarction by photochemically initiated thrombosis. Ann Neurol. 1985;17(5):497–504.
Zheng W, Watts LT, Holstein DM, Prajapati SI, Keller C, Grass EH, et al. Purinergic receptor stimulation reduces cytotoxic edema and brain infarcts in mouse induced by photothrombosis by energizing glial mitochondria. PLoS One. 2010;5(12):e14401.
Zheng W, Talley Watts L, Holstein DM, Wewer J, Lechleiter JD. P2Y1R-Initiated, IP3R-dependent stimulation of astrocyte mitochondrial metabolism reduces and partially reverses ischemic neuronal damage in mouse. J Cereb Blood Flow Metab. 2013;33(4):600–11.
Witte OW, Stoll G. Delayed and remote effects of focal cortical infarctions: secondary damage and reactive plasticity. Adv Neurol. 1997;73:207–27.
Hagemann G, Redecker C, Neumann-Haefelin T, Freund HJ, Witte OW. Increased long-term potentiation in the surround of experimentally induced focal cortical infarction. Ann Neurol. 1998;44(2):255–8.
Labat-gest V, Tomasi S. Photothrombotic ischemia: a minimally invasive and reproducible photochemical cortical lesion model for mouse stroke studies. J Vis Exp. 2013;9(76):50370.
Kleinschnitz C, et al. Blocking of platelets or intrinsic coagulation pathway-driven thrombosis does not prevent cerebral infarctions induced by photothrombosks. Stroke. 2008;39:1262–8.
Uzdensky AB. Photothrombotic stroke as a model of ischemic stroke. Transl Stroke Res. 2018;9(5):437–51.
Sato K, Kimoto M, Kakumoto M, Horiuchi D, Iwasaki T, Tokmakov AA, et al. Adaptor protein Shc undergoes translocation and mediates upregulation of the tyrosine kinase c-Src in EGF-stimulated A431 cells. Genes Cells. 2000;5(9):749–64.
Ventura A, Maccarana M, Raker VA, Pelicci PG. A cryptic targeting signal induces isoform-specific localization of p46Shc to mitochondria. J Biol Chem. 2004;279(3):2299–306.
Nemoto S, Combs CA, French S, Ahn BH, Fergusson MM, Balaban RS, et al. The mammalian longevity-associated gene product p66shc regulates mitochondrial metabolism. J Biol Chem. 2006;281(15):10555–10,560.
Lang JY, Ma K, Guo JX, Sun H. Oxidative stress induces B lymphocyte DNA damage and apoptosis by upregulating p66shc. Eur Rev Med Pharmacol Sci. 2018;22(4):1051–60.
Xie ZZ, Shi MM, Xie L, Wu ZY, Li G, Hua F, et al. Sulfhydration of p66Shc at cysteine 59 mediates the antioxidant effect of hydrogen sulfide. Antioxid Redox Signal. 2014;21(18):2531–42.
Piao S, Nagar H, Kim S, Lee I, Choi SJ, Kim T, et al. CRIF1 deficiency induced mitophagy via p66shc-regulated ROS in endothelial cells. Biochem Biophys Res Commun. 2020;522(4):869–875
Yoo SM, Jung YK. A molecular approach to mitophagy and mitochondrial dynamics. Mol Cell. 2018;41(1):18–26.
Funding
This research was supported by the Ministry of Science, ICT & Future Planning (NRF-2019R1C1C1005585, NRF-2018R1D1A1B07045664), and the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2019R1A2C2004884).
Author information
Authors and Affiliations
Contributions
JH participated in the design of the study, carried out pain behavior testing and immunostaining studies, participated in data analysis, and drafted the manuscript. NS and HJS carried out real-time PCR. YY and HHK participated in the RB model and behavior tests. HP and SK participated in in vitro experiments. DWK and HS participated in the design of the study and data analysis and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All protocols were performed in accordance with the Chungnam National University hospital Guide for the Care and Use of Laboratory Animals and were approved by the University of Chungnam Institutional Animal Care and Use Committee.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PPTX 865 kb)
Rights and permissions
About this article
Cite this article
Hwang, JA., Shin, N., Shin, H.J. et al. Protective Effects of ShcA Protein Silencing for Photothrombotic Cerebral Infarction. Transl. Stroke Res. 12, 866–878 (2021). https://doi.org/10.1007/s12975-020-00874-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-020-00874-1