Abstract
The mechanism of cognitive dysfunction caused by ischemic white matter lesions is unclear. To explore the effect and mechanism of different cell-derived adenosine A2A receptor (A2AR) in cognitive impairment caused by chronic hypoperfusion white matter lesions (CHWMLs), we destroyed the bone marrow hematopoietic capacity of the recipient mice using radiation irradiation followed by establishing the selectively inactivated or reconstituted A2AR models with the transplanting bone marrow from global A2AR gene knockout or wild-type mice into wild-type or gene knockout mice, respectively. Then Morris Water Maze (MWM), ELISA, immunohistochemistry, and Bielschowsky silver staining were used to assess the effect and mechanism of the cognitive function in chronic cerebral blood flow hypoperfusion (CCH) model. Selectively reconstructing bone marrow-derived cells (BMDCs) A2AR (WT → KO group) and activated total adenosine A2AR with CGS21680 (CCH + CGS group) improved the cognitive related index. Activation of BMDC A2AR suppressed expression of inflammatory cytokines in peripheral blood and reduced the number of activated microglia cells co-localized with cystatin F in local brain, consequently inhibited white matter lesions. On the contrary, selective inactivation of adenosine A2AR (KO → WT group) and activation of non-BMDC A2AR with CGS21680 (KO → WT + CGS group) served the opposite effects. These results suggested that BMDC A2AR could inhibit white matter lesions and attenuate cognitive impairment after CHWMLs, whereas non-BMDC A2ARs aggravate cognitive impairment. The systemic inflammatory response and local activated microglia with cystatin F high expression were involved in the process of cognitive function recovery with BMDC A2AR. The overall trend is that BMDC A2ARs play a leading role.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ischemic leukoencephalopathy refers to a wide range of lesions within the deep perforating arteriole in the hemispheres. The cognitive dysfunction caused by ischemic leukoencephalopathy is the main cause of vascular dementia [1]. Amelioration of cognitive impairment caused by persistent hypoperfusion can significantly improve the survival quality of chronic leukodystrophy patients [2]. However, at present, there is insufficient understanding of the pathogenesis of cognitive dysfunction caused by chronic hypoperfusion white matter lesions (CHWMLs), which greatly hinders the development of therapeutic drugs.
In recent years, the adenosine A2A receptor (A2AR) has been extensively studied in various disease models due to its multiple biological effects, such as pro-inflammation, anti-inflammation, neuroprotection [3,4,5]. In central nervous system diseases, adenosine A2AR has a dual role, with a protective role in some diseases [6,7,8] and aggravation of damage in other diseases [9, 10]. The effect of adenosine A2AR on cognitive function is also contradictory. It has been reported that inhibition of neuron adenosine A2AR can improve cognitive dysfunction caused by Parkinson’s disease [11]. Conditional knockout of astrocyte A2AR can promote long-term memory retention in Alzheimer’s disease patients [7]. Adenosine A2AR aggravates intracranial inflammation through activating microglia, leading to cognitive dysfunction in mice with acute ischemia and hypoxia [12]. However, it has also been reported that promoting adenosine A2AR accumulation in the hippocampus can promote cognitive improvement in schizophrenia [13], that adenosine A2AR knockdown aggravates the motor and cognitive dysfunction of Huntington’s disease by reducing the expression of morphine peptides in the striatum region, and that increasing A2A receptor expression in peripheral blood mononuclear cells can improve the prognosis of mild cognitive function disorder patients by reducing inflammation [14]. These data suggest that adenosine A2AR plays two distinct roles in the recovery of cognitive function in different diseases. However, the role and mechanism of adenosine A2AR in cognitive dysfunction induced by CHWMLs has not been reported, and it is worthy of further exploration.
Our previous research confirmed that BMDC A2ARs can inhibit inflammation [15], while non-BMDC A2ARs activate microglia and promote inflammatory responses [16]. Different sources of adenosine A2AR play contradictory roles. In general, the anti-inflammatory effect mediated by BMDC A2ARs is stronger than the pro-inflammatory effect of non-BMDC A2ARs. Promoting overall adenosine A2A expression can inhibit inflammation and reduce CHWMLs [17]. However, in the persistent chronic hypoperfusion model, the role of different cell-derived adenosine A2AR in cognitive function recovery and its mechanism need to be further studied. In consideration of the effect of the inflammation in the cognitive function recovery, we hypothesize that BMDC A2ARs could improve the cognitive function of chronic white matter ischemia model and non-BMDC A2ARs served the opposite effects.
Therefore, the aims of our study were to apply bone marrow transplantation after irradiating wild-type mice and A2A knockout mice to establish models with selective inactivation or reconstruction of BMDC A2ARs, to explore the effect and mechanism of adenosine A2AR from different tissue sources on cognitive function in CHWMLs, and to find corresponding targets to improve the quality of life of patients with chronic white matter ischemia.
Methods
Experimental Animals and Drugs
Global Adenosine A2AR gene knockout (A2AR gKO) C57BL/6 mice were a gift from Dr. Jiang-Fan Chen (Boston University School of Medicine, Boston, MA). All mice in this experiment were 8–9 weeks old and weighed 23–30 g. Female mice were donors, and male mice were recipients. The adenosine A2AR-specific agonist CGS21680 was purchased from TOCRIS, UK.
Bone Marrow Cell Extraction
Adenosine A2AR KO mice and littermate WT mice were sacrificed by cervical dislocation, and the femur and tibia were separated on a clean platform. The bone marrow cells in the medullary cavity were washed out with RPMI 1640 medium. The cells were collected, filtered through a 200-mesh sieve, and centrifuged at 1500 rpm for 5 min, and the precipitated cells were washed twice with RPMI 1640 medium, centrifuged at 1500 rpm for 5 min, and pulverized in RPMI 1640 medium to form a cell suspension. The cell concentration was adjusted to 2 × 108/mL for use. The cell viability was checked by 0.2% trypan blue staining, and the number of viable cells was greater than 95% for bone marrow transplantation.
Establishment of a Chimeric Model of Bone Marrow Transplant Cells
The recipient mice (male) were subjected to whole body gamma irradiation with a total dose of 12.5 Gy from a 60Co source, which destroyed the bone marrow hematopoietic capacity. The collected bone marrow cell suspension was injected into the recipient mice via the tail vein (the concentration of bone marrow cells was 2 × 108/mL, 0.3 mL/rat), and the bone marrow cell transplantation was completed [18]. Bone marrow cells of female adenosine A2AR KO mice were transplanted into male C57BL/6 mice (KO → WT) to establish a selective inactivated model of BMDC A2ARs; bone marrow cells of female WT mice were transplanted into male C57BL/6 mice as controls (WT → WT); bone marrow cells of female WT mice were transplanted into male adenosine A2A receptor KO mice (WT → KO), which was used as a selective reconstruction model of BMDC A2ARs; and bone marrow cells of female adenosine A2A receptor KO mice were transplanted into male adenosine A2A receptor KO mice (KO → KO), which served as controls. The experiment was carried out 8 weeks after transplantation.
Establishment of a Chronic Cerebral Blood Flow Hypoperfusion Model
A chronic cerebral blood flow hypoperfusion (CCH) model was established with reference to previously described methods [19]. Anesthesia was achieved with isoflurane inhalation, and a supine position was applied. A median longitudinal incision was made after cervical anterior skin depilation and sterilization, the neck muscle was separated, and the bilateral common carotid artery was fully liberated. Two 3-0 nylon threads were used to loosely wrap the near and far end of the right common carotid artery, and two nylon threads were suspended with a hemostat and placed in a specially designed microspring with an inner diameter of 1.8 mm. Then, the spring coil was carefully placed over the common carotid artery; after 30 min, the spring loop was placed over the left common carotid artery with the same method. In the sham-operated control group, the bilateral common carotid arteries were exposed but not clipped. The skin was sutured and disinfected after surgery. The anal temperature of the mice was maintained at 36.5–37.5 °C during the operation. During the whole operation, the Doppler blood flow meter 414-1 probe was fixed at the junction of the scale and the sphenoid wing above the zygomatic arch to measure the cerebral blood flow and evaluate the modeling.
Morris Water Maze Task
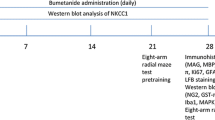
The learning and memory functions of the mice were evaluated using a Morris Water Maze (MWM) task, which was started on the 12th day before modeling and the 21st day after modeling. A black pool with a diameter of 120 cm, a height of 40 cm, and a depth of 25 cm was selected, and warm water with a temperature of 20–23 °C was infused. The pool was divided into 4 quadrants (NW, NE, SE, and SW), and a platform with diameter of 5 cm was placed at the center of the NE quadrant, 1 cm below the horizontal plane. Visual cues of different shapes were given on the NW- and SE-oriented swimming pool walls. The first day was visible platform training (VP), and a marker was placed over the platform to allow the mouse to identify the platform. The mice were placed into the water in a clockwise direction (beginning from the NW quadrant), and the time required for the mouse to find the platform stay on the platform for 10s (escape latency) was recorded. If the mouse did not find the platform within 120s, the experimenter guided it onto the platform, and it stayed on it for 10s. In this situation, the escape latency was recorded as 120s. The second to fifth days were the acquisition phase (AP). At this stage, the mark was removed from the platform, and the above experiment was repeated to record the escape latency and swimming distance. A probe test (PT) was carried out on the sixth day. At this stage, the platform was removed, and the mice were allowed to enter the water from the NW quadrant. The time in the correct quadrant (SE) and the number of passages through the platform location within the 120s were recorded.
Immunohistochemistry
At the 2nd week and 4th week after CCH, 5 mice were randomly selected from each group, anesthetized with 1% sodium pentobarbital (60 mg/kg), and perfused with 4% paraformaldehyde. Their brain tissues were taken out after dehydration and then frozen (10 μm thick per section). After the sections were antigen-repaired, permeated, and blocked, they were incubated at 4 °C overnight with the ZO-1 antibody. The sections were then incubated with biotin-labeled secondary antibody and horseradish peroxidase label for 2h at room temperature, and finally, coloration was performed with a DAB kit. Then, the expression of ZO-1 was observed under a microscope. Semiquantitative analysis of staining results was performed using Image Pro Plus software. The Nishigaya method was used [20]. The staining intensity of the WT mouse in the sham group (normal) was rated as 4. It was rated as 3 when it was slightly lower than normal, rated as 2 when moderately lower than normal, rated as 1 when significantly lower than normal, rated as 0 when the negative result was positive, rated as 5 when slightly stronger than normal, rated as 6 when it was moderately stronger than normal, and rated as 7 when it was significantly stronger than normal.
Measurement of Blood-Brain Barrier Permeability
To analyze the alterations in cerebral vascular permeability, Evans blue dye was used as a marker of albumin extravasation. Briefly, mice were injected with 2% EB (3 mL/kg) via the tail vein 4 weeks after CCH. After an hour, animals were then perfused transcardially with phosphate buffer saline (PBS) to purge the intravascular EB dye. Whole brains were isolated, and the dye was extracted with N,N-dimethylformamide overnight at 50 °C, followed by centrifugation at 5000 rpm for 20 min. The concentration was determined from the OD (610 nm) values according to the standard curve, and then the EB content in the brain tissue was calculated to evaluate blood-brain barrier (BBB) permeability.
Immunofluorescence Staining
At the 4th week after CCH, tissue sections were fixed in 4% paraformaldehyde for 30 min, followed by permeabilization with a 0.1% Triton X-100 solution for 30 min and antigen blocking with 5% goat serum at room temperature for 30 min. The sections were incubated with cystatin F (1:50, Santa Cruz Biotechnology, Inc., USA) and CD11b (1:100, Chemicon, Colorado, USA) overnight at 4 °C. Then, staining with a fluorescein-labeled secondary antibody and DAPI was performed in the dark. The cells were finally mounted on glass slides and observed under a laser confocal microscope.
Enzyme-Linked Immune Sorbent Assay
At the 4th week after CCH, 5 mice were randomly selected from each group, and peripheral blood was taken for an enzyme-linked immune sorbent assay (ELISA) to evaluate systemic inflammatory responses. The eBioscience ELISA kit was used, and the reagent instructions were as follows. After preparing the standard, the sample was added and incubated at 37 °C for 30 min. It was then washed repeatedly with PBS buffer, the enzyme standard reagent was added, and the sample then was incubated again at 37 °C for 30 min. Finally, the developer was added, and the absorbance of each well was measured at 450 nm with a microplate reader.
Bielschowsky Silver Staining
Bielschowsky silver staining was carried out according to the instructions. Paraffin sections were dewaxed, 20% silver nitrate solution was dip-coated for 24 h at room temperature, the sections were washed with double distilled water, 10% formaldehyde was reduced, and then 20% silver nitrate solution + ammonia water was applied again for coloration. The sections were then fixed with 5% sodium thiosulfate and sealed after anhydrous ethanol dehydration. The judgment criteria were graded for the degree of damage of white matter nerve fibers [21]: grade 0, normal; grade 1, disordered arrangement of nerve fibers; grade 2, significant vacuolation (nucleus damage); and grade 3, loss of myelinated nerve fibers (neural fiber axon damage).
Statistical Methods
All data are presented as the mean ± standard error. Data analysis was performed using SPSS 22.0. A one-way analysis of variance with the appropriate LSD post hoc test was used for comparison of experimental groups. The MWM test results were analyzed with multivariate analysis of variance with the appropriate LSD post hoc test. P < 0.05 indicated statistical significance.
Results
Adenosine A2AR Gene Knockout Had No Effect on Cognitive Function in Normal Mice
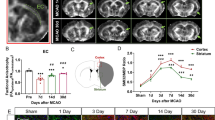
Doppler blood flow monitoring (Fig. 1b) showed the significant differences in the reduction of the cerebral blood flow (CBF) after CCH induction among the groups at each detection time point (Fig. 1c) (P > 0.05). Mouse learning and memory functions were assessed with MWM. The AP stage and the PT stage were used to detect the learning ability and memory function, respectively. After systematic training, the time for both sham (WT) and sham (KO) mice to find the platform hidden was gradually shortened (Fig. S1A). There was no statistical difference in the escape latency (Fig. S1A), the swimming distance (Fig. S1B), the time in correct quadrant (CQ) (Fig. S1C), and the number of passing through platform (Fig. S1D). These data illustrated that the A2AR knockout had no effect on the motor and cognitive function in normal mice.
Adenosine A2AR knockout aggravates cognitive function impairment in CCH mice. a Experimental time axis. b Doppler blood flow was used to monitor the changes in cerebral blood flow (CBF) before modeling and 2 h after modeling. CBF reduction rate (%) = (basic CBF value − post ischemic CBF value) / base CBF value × 100. CBF of bone marrow transplantation mice (c) was statistically analyzed. d The typical trajectory of each group of mice to reach the hidden platform. e Escape latency. f Swimming distance. g The typical heat map of mice in each quadrant. h Time in the correct quadrant (SE, lower right quadrant). i The number of passages through the platform location. *P < 0.05, N.S., no statistical differences, N = 12/group. CBF, cerebral blood flow; CCH, chronic cerebral blood flow hypoperfusion; MWM, Morris water maze task; VP. visible platform training; AP, acquisition phase; PT, probe test
Adenosine A2AR Alleviated Cognitive Impairment in CCH Mice
The escape latency in CCH group was significantly prolonged than that in sham group (Fig. 1e), indicating that CHWMLs caused abnormal learning function. The escape latency of the CCH (WT) group was shorter than that in CCH (KO) group, whereas was longer than that in the CCH + CGS (WT) group (Fig. 1e), demonstrating that A2AR attenuates cognitive impairment. Although the swimming distance (Fig. 1f) was statistically significant between CCH (WT) and CCH (KO) group, the swimming speed of each group showed no statistical significance, which indicated that A2AR inactivation had no effect on motor function. In the space exploration experiment, the time in target quadrant (Fig. 1h) and the number of passing through platforms (Fig. 1i) in CCH (WT) group were statistically significant when compared with these in the CCH + CGS (WT) group and CCH (KO) group. The above data illustrated that CHWMLs led to cognitive dysfunction, and A2AR alleviated cognitive impairment induced by CHWMLs.
Bone Marrow Transplantation Has No Effect on Cognitive Function in Mice
After bone marrow cell transplantation, the time to find the platform hidden underwater in the groups of WT → WT, KO → WT, WT → KO, and KO → KO was gradually shortened, and there was no significant difference in escape latency (Fig. S1E), indicating that the bone marrow transplantation had no effect on the learning ability of normal mice. There was no significant difference in swimming distance (Fig. S1F), in the time in the correct quadrant (Fig. S1G), or the number of passages through the platform location (Fig. S1H) in each group. The above data indicated that bone marrow transplantation had no effect on learning and memory function in mice.
Selective Inactivation of BMDC A2ARs Aggravates Cognitive Impairment and Reconstructed BMDC A2ARs Promote Cognitive Recovery
To further investigate the role of different cell-derived adenosine A2AR in cognitive impairment following CHWMLs, we evaluated the effect of BMDC A2ARs and non-BMDC A2ARs on cognitive recovery by irradiation and bone marrow transplantation. Compared with the WT → WT group, the escape latency of the KO → WT group was longer, the time in the target quadrant was shortened, and the number of passages through the platform location was reduced, indicating that the selective deletion of BMDC A2ARs aggravated cognitive impairment. At the same time, the cognitive related indexes in the WT → KO group were also significantly different from those in the KO → KO group (Fig. 2). These data indicated that BMDC A2AR reconstruction can promote cognitive function recovery after CHWMLs.
BMDC A2ARs promoted cognition recovery. a The typical trajectory of mice to reach the hidden platform. b Escape latency. c Swimming distance. d The typical heat map of mice in each quadrant. e Time in the correct quadrant of each group. f The number of passages through the platform location. *P < 0.05, N = 12/group
Non-BMDC A2AR Activation Aggravates Cognitive Impairment
Based on the analysis of the experimental results, we found that the cognitive function-related indexes in the KO → WT group were significantly different from those in the WT → KO group (Fig. 2). These data suggested that non-BMDC A2ARs may aggravate cognitive impairment. To further validate this conclusion, we injected CCH mice with the adenosine A2AR agonist CGS21680. Compared with the KO → WT group, the KO → WT + CGS group had a prolonged escape latency, shortened time in the target quadrant, and reduced number of passages through the platform location (Fig. 3). These data fully demonstrated that non-BMDC A2AR activation aggravates cognitive impairment caused by CCH.
Non-BMDC A2AR activation aggravates cognitive impairment. a The typical trajectory of each group to reach the hidden platform. b Escape latency. c Swimming distance. d The typical heat map of mice in each quadrant. e Time in the correct quadrant of each group. f The number of passages through the platform location. *P < 0.05, N = 12/group
BMDC A2ARs Play a Leading Role in the Recovery of Cognitive Function After CHWMLs
Different sources of adenosine A2AR play different roles in cognitive function recovery. Compared with the WT → KO group, the escape latency was shortened, the time in the target quadrant and the number of passages through the platform location increased in the WT → KO + CGS group (Fig. 3), suggesting that activation of BMDC A2ARs promoted cognitive recovery. The MWM task results in the KO → WT group were significantly different from those in the KO → WT + CGS group, suggesting that further activation of non-BMDC A2ARs aggravated cognitive impairment. The escape latency, time in the target quadrant, and number of passages through the platform location in the WT + Veh group were significantly different from those in the WT + CGS group, indicating that BMDC A2ARs play a leading role in cognitive function recovery.
BMDC A2ARs Suppressed Systemic Inflammation and Non-BMDC A2ARs Aggravated Systemic Inflammatory Response in CHWMLs
We subsequently evaluated the mechanism by which BMDC A2ARs promoted cognitive function recovery after CHWMLs. Systemic inflammatory responses in each group were measured with an ELISA (Fig. 4). Compared with the WT → WT group, the expression of the pro-inflammatory cytokines IL-1β and TNF-α was increased, and that of the anti-inflammatory cytokine IL-10 and TGFβ was decreased in the KO → WT group, indicating that the selective inactivation of BMDC A2ARs leads to a systemic inflammatory reaction. The expression of inflammatory factors in WT → KO and KO → KO groups was significantly different, indicating that selective reconstitution of BMDC A2ARs can inhibit inflammation. The expression of pro-inflammatory cytokines IL-1β and TNF-α in the KO → WT group was higher and the expression of anti-inflammatory cytokines IL-10 and TGFβ was less than that in the KO → KO group, indicating that non-BMDC A2ARs promoted an inflammatory response.
To confirm the effect of adenosine A2AR from different tissue sources on the inflammatory response, we used the A2AR-specific agonist CGS21680 to intervene in different bone marrow cells of transplanted mice. Compared with the WT → KO group, the anti-inflammatory cytokines were suppressed and the pro-inflammatory cytokines were increased in the WT → KO + CGS group, indicating that activation of BMDC A2ARs can inhibit the inflammatory response. The expression of inflammatory factors in the KO → WT group was also significantly different from that in the KO → WT + CGS group, further suggesting that non-BMDC A2ARs aggravated the inflammation. IL-1β and TNF-α expressions in the WT + CGS group were higher, and IL-10 and TGFβ were decreased compared with the WT + Veh group, indicating that the anti-inflammatory effect of BMDC A2ARs plays a leading role in CHWMLs.
BMDC A2ARs Inhibited Microglia Activated in CHWMLs
Firstly, we examined the effect of adenosine A2AR on BBB integrity. We found that A2AR knockout (Fig. S2A, B) and bone marrow transplantation (Fig. S2C, D) had no effect on the BBB dysfunction in normal mice. The ZO-1 immunohistochemistry staining and Evans blue content test demonstrated that the BBB was destroyed and gradually repaired after CCH. However, there was no statistical difference in ZO-1 expression and Evans blue content among each group with different interventions (Fig. S2) at each time point. These data indicated that BBB alterations were not the key points for cognitive function recovery with adenosine A2AR.
Adenosine A2AR involves in immune response during inflammation and infection. In addition, we also reported that cystatin F (CF) in the activated microglia was closely associated with the effect of the A2AR [17]. Therefore, we subsequently investigated whether CF was implicated in the cognitive function recovery mediated with adenosine A2AR (Fig. 5). Immunofluorescence staining revealed the expression of CF was co-localized with CD11b, a specific biomarker of microglia after CCH in WT → WT group. A large number of CF+CD11b+ cells were observed in BMDC A2ARs selective inactivated group (KO → WT). By contrast, BMDC A2ARs reconstruction group (WT → KO) significantly reduced the number of CF+CD11b+ cells, compared to the KO → KO group. Activated BMDC A2ARs with CGS administration could decrease the number of CF+CD11b+ cells, whereas activated non-BMDC A2ARs with CGS increase CF+CD11b+ cells. These data suggested that CF-mediated neuroinflammation involved in cognitive function recovery with adenosine A2AR.
BMDC A2ARs inhibited CF-mediated microglia activation 4 weeks post CCH. a Confocal microscopy demonstrating the expression of CF co-localization with microglia after selectively inactivated or reconstituted A2AR in BMDCs. The activated microglia marker CD11b was labeled with Alexa Fluor 488 (green), the CF was stained with Alexa Fluor 555 (red), and nuclei were stained with DAPI (blue). b Quantitative analysis for the number of CF+CD+ cells after selectively inactivated or reconstituted A2AR in BMDCs. Confocal microscopy (c) and quantitative analysis (d) for CF+CD+ cells with adenosine A2AR agonist CGS21680 intervention. *P < 0.05, bar = 20 μm, N = 5/group
To further evaluate the mechanism by which BMDC A2ARs promote cognitive function recovery after CHWMLs, we examined the effect of A2AR on blood-brain barrier repair. There was no significant difference in ZO-1 expression between the WT group and KO group (Fig. 4a, b) and no significant difference among the WT → WT, KO → WT, WT → KO, and KO → KO groups (Fig. 4c, d), indicating that adenosine A2AR gene knockout and bone marrow transplantation had no effect on the blood-brain barrier of normal mice. Two weeks after CCH, the blood-brain barrier was destroyed, but there was no significant difference in ZO-1 expression among the groups (Fig. 4e, f). The expression of ZO-1 increased at 4 weeks post CCH compared with that at 2 weeks after modeling, indicating that the blood-brain barrier continued to recover, whereas there was no significant difference in ZO-1 expression among the groups (Fig. 4g, h). These data indicated that adenosine A2AR had no effect on recovery of the blood-brain barrier.
Selective Inactivated BMDC A2ARs Aggravated White Matter Lesions
Bielschowsky silver staining was used to assess white matter lesions in the corpus callosum (Fig. 6). Under normal circumstances, the nerve fibers around the corpus callosum are arranged closely and orderly. After chronic ischemia, the nerve fibers are disordered, suggesting there was white matter damage. The statistical results in the bone marrow cell transplant groups indicated that the degree of corpus callosum white matter nerve fiber damage was ordered from high to low as follows: KO → WT, KO → KO, WT → WT, WT → KO. The silver staining score of the A2AR-specific agonist CGS21680 intervention group had a statistically difference compared with other groups. These results suggested that BMDC A2ARsR knockout aggravated demyelination, the non-BMDC A2AR knockout exerts the opposite effect, and the role of BMDC A2ARs is dominant.
BMDC A2AR reduced white matter lesions. a Bielschowsky silver staining results of BMDC A2AR selective inactivated or reconstruction group. b Statistical analysis of silver staining in each group. c Bielschowsky silver staining results for each group after adenosine A2A receptor-specific agonist CGS21680 intervention. d Silver staining statistical analysis with CGS21680 intervention. *P < 0.05, bar = 20 μm, N = 5/group
Discussion
Our results confirmed that BMDC A2ARs promoted cognitive function recovery in a CCH mouse model, while non-BMDC A2ARs aggravated cognitive impairment. In general, the function of BMDC A2ARs to promote cognitive recovery plays a leading role, and its mechanism is mainly achieved by reducing inflammation and white matter lesions.
Dementia caused by vascular cognitive impairment accounts for more than 20% of the number of dementia patients, which is the second leading cause of dementia, after Alzheimer’s disease (AD) [22, 23]. Chronic hypoperfusion is an important factor leading to vascular cognitive impairment, and it has received increasing attention in recent years. The phosphodiesterase inhibitor roflumilast [24] and the transient receptor potential melastatin 2 (TRPM2) inhibitor [25] can suppress microglial activity after CCH injury and promote cognitive function recovery through anti-inflammatory effects. IL-1β receptor inhibitors and IL-1β knockout mice block the IL-1β signaling pathway, thereby inducing oligodendrocyte premature cell migration to the periphery of the corpus callosum, and avoiding white matter damage and reversing chronic hypoperfusion-induced cognitive dysfunction by improving the local inflammatory environment [26]. MiR-181c [27] and MiR-96 [28] can ameliorate cognitive dysfunction by regulating autophagy in CCH [29]. In general, demyelination, inflammatory responses, and mitochondrial dysfunction are common pathways leading to cognitive dysfunction [30, 31], which can ameliorate the prognosis of chronic cortical hypoperfusion by reducing white matter lesions.
Adenosine is an intermediate product of energy metabolism, which is widely distributed in various tissues and organs. It is also an important neuromodulator in the nervous system that plays an important role in regulating inflammation. Our previous studies reported that the anti-inflammatory effects of BMDC A2ARs are stronger than the pro-inflammatory effects of non-BMDC A2ARs [15], but it is not clear whether its role in cognitive impairment is caused by CHWMLs. MWM showed that the cognitive function-related indexes in the CCH (WT) group were significantly different from those in the CCH (KO) group. These data suggested that adenosine A2AR involved in cognitive impairment after CHWMLs. To further evaluate the role of adenosine A2AR in cognitive impairment in different tissue sources, we used irradiation and bone marrow transplantation to establish a selective inactivated or reconstruction model of BMDC A2ARs. The results of the MWM test suggested that after selective deletion of BMDC A2ARs, the escape latency was prolonged, the time in the target quadrant was shortened, the number of passages through the platform location was reduced, and the trend was consistent with the trend of A2AR whole gene knockout. The selective reconstruction of BMDC A2ARs can reverse the above results, further indicating that BMDC A2ARs have protective effects on cognitive function. In the course of the experiment, we also found an interesting phenomenon; that is, the cognitive function-related index in the KO → WT group had a significant difference in comparison with the WT → KO group, which suggested that non-BMDC A2ARs may aggravate cognitive function damage. To further confirm this hypothesis, we used the adenosine A2AR agonist CGS21680 to interfere in different bone marrow transplantation groups. The experimental results suggested that only activated non-BMDC A2ARs prolong the escape latency, reduce the time in the target quadrant, and decrease the number of passages through the platforms. Only activated BMDC A2ARs will shorten the escape latency and increase the time in the target quadrant and the number of passages through the platform location. These data confirmed our hypothesis that BMDC A2ARs promote cognitive function recovery and that non-BMDC A2ARs aggravate cognitive impairment. However, the overall trend is that adenosine A2AR provides a protective effect on cognitive impairment in CHWMLs.
As we previously mentioned, the inflammatory response is the basic pathway for cognitive impairment caused by CHWMLs. We therefore further verified whether anti-inflammation is the mechanism by which adenosine A2AR promotes the recovery of neurological function. Selective inactivated BMDC A2ARs or activated non-BMDC A2ARs led to systemic inflammatory response, and increased the expression of CF, accordingly activated microglia, whereas selective reconstitution reversed the above phenomenon. CF, a potent endogenous cysteine protease inhibitor, was substantially up-regulated in regions of white matter rarefaction that occurred in various demyelinating diseases of the CNS [32, 33]. In this study, we found that BMDC A2ARs suppressed CF-mediated microglia activated and non-BMDC A2ARs promoted CF-mediated microglia activated, which was consistent with our previous research [16]. The immunohistochemistry suggested BBB alterations was not the key point for cognitive function recovery with adenosine A2ARs. However, elevated BBB permeability was the pathological basis of systemic inflammatory cytokines acting on brain parenchyma cells. Silver staining results suggested that activation of BMDC A2ARs can alleviate corpus callosum white matter damage. These data confirmed our hypothesis that adenosine A2AR promotes the recovery of cognitive function in mice by inhibiting inflammatory responses and reducing white matter lesions. In this process, BMDC A2ARs play a leading role.
In the AD and PD disease models, adenosine A2AR aggravates cognitive dysfunction, whereas adenosine A2AR can improve cognitive function in the HD and schizophrenia disease models. It is worth further consideration to determine the reason for the exact opposite effect of A2AR. Literature reports have suggested that adenosine A2AR had two main pathways for regulating cognitive function: one is via integrating dopamine, glutamate, N-methyl-D-aspartate (NMDA), and brain-derived neurotrophic factor signaling pathways, and the other is regulating inflammatory responses [34]. Adenosine A2AR mediates cognitive dysfunction in AD and PD disease models mainly through the first pathway [9, 35, 36], while in the HD and schizophrenia disease models, it mainly protects through the second pathway [13, 14, 37, 38]. This study showed that adenosine A2AR also improved cognitive function through anti-inflammatory effects in CHWMLs, and this mechanism of action is consistent with the above conjecture. However, at the same time, we must also realize that in other disease models [12, 39,40,41,42], the role of adenosine A2AR in cognitive function cannot be explained by the above mechanism, indicating that there may be other functional ways, which requires further study.
Although the FDA has approved acetylcholinesterase inhibitors and an NMDA receptor antagonist, memantine, as a drug for the treatment of cognitive dysfunction since 1993, these drugs currently only play a role in delaying the course of cognitive dysfunction and cannot effectively stop or reverse the course of the disease [43]. Moreover, specific drugs for cognitive impairment of chronic hypoperfusion leukoencephalopathy are lacking. Adenosine A2AR has multiple biological effects and can play different roles in different disease models. This study found that BMDC A2ARs inhibit cognitive function after CHWMLs through anti-inflammatory effects. Non-BMDC A2ARs can aggravate cognitive impairment through inflammation. Overall, BMDC A2ARs play a leading role. This study initially explored the role and mechanism of adenosine A2AR in the pathogenesis of cognitive impairment in CHWMLs and provides a new treatment strategy for CHWMLs.
Conclusions
This study finds that BMDC A2ARs inhibit cognitive impairment after CHWMLs through anti-inflammatory effects. Non-BMDC A2ARs can aggravate cognitive impairment through pro-inflammation. Overall, global A2ARs activation could alleviate cognitive impairment after CHWMLs. This study initially explores the role and mechanism of adenosine A2AR in the pathogenesis of cognitive impairment in CHWMLs, and provides a new treatment strategy for CHWMLs.
References
Wang Y, Liu G, Hong D, Chen F, Ji X, Cao G. White matter injury in ischemic stroke. Prog Neurobiol. 2016;141:45–60. https://doi.org/10.1016/j.pneurobio.2016.04.005.
Joutel A, Monet-Lepretre M, Gosele C, Baron-Menguy C, Hammes A, Schmidt S, et al. Cerebrovascular dysfunction and microcirculation rarefaction precede white matter lesions in a mouse genetic model of cerebral ischemic small vessel disease. J Clin Invest. 2010;120(2):433–45. https://doi.org/10.1172/JCI39733.
Cai Y, Li H, Liu M, Pei Y, Zheng J, Zhou J, et al. Disruption of adenosine 2A receptor exacerbates NAFLD through increasing inflammatory responses and SREBP1c activity. Hepatology. 2018;68(1):48–61. https://doi.org/10.1002/hep.29777.
Jespers W, Schiedel AC, Heitman LH, Cooke RM, Kleene L, van Westen GJP, et al. Structural mapping of adenosine receptor mutations: ligand binding and signaling mechanisms. Trends Pharmacol Sci. 2018;39(1):75–89. https://doi.org/10.1016/j.tips.2017.11.001.
Beavis PA, Henderson MA, Giuffrida L, Mills JK, Sek K, Cross RS, et al. Targeting the adenosine 2A receptor enhances chimeric antigen receptor T cell efficacy. J Clin Invest. 2017;127(3):929–41. https://doi.org/10.1172/JCI89455.
Kim DG, Bynoe MS. A2A adenosine receptor modulates drug efflux transporter P-glycoprotein at the blood-brain barrier. J Clin Invest. 2016;126(5):1717–33. https://doi.org/10.1172/JCI76207.
Orr AG, Hsiao EC, Wang MM, Ho K, Kim DH, Wang X, et al. Astrocytic adenosine receptor A2A and Gs-coupled signaling regulate memory. Nat Neurosci. 2015;18(3):423–34. https://doi.org/10.1038/nn.3930.
Newell EA, Exo JL, Verrier JD, Jackson TC, Gillespie DG, Janesko-Feldman K, et al. 2′,3′-cAMP, 3′-AMP, 2′-AMP and adenosine inhibit TNF-alpha and CXCL10 production from activated primary murine microglia via A2A receptors. Brain Res. 2015;1594:27–35. https://doi.org/10.1016/j.brainres.2014.10.059.
Viana da Silva S, Haberl MG, Zhang P, Bethge P, Lemos C, Goncalves N, et al. Early synaptic deficits in the APP/PS1 mouse model of Alzheimer’s disease involve neuronal adenosine A2A receptors. Nat Commun. 2016;7:11915. https://doi.org/10.1038/ncomms11915.
Nishiyama K, Suzuki H, Maruyama M, Yoshihara T, Ohta H. Genetic deletion of GPR52 enhances the locomotor-stimulating effect of an adenosine A2A receptor antagonist in mice: a potential role of GPR52 in the function of striatopallidal neurons. Brain Res. 1670;2017:24–31. https://doi.org/10.1016/j.brainres.2017.05.031.
Li Z, Chen X, Wang T, Gao Y, Li F, Chen L, et al. The corticostriatal adenosine A2A receptor controls maintenance and retrieval of spatial working memory. Biol Psychiatry. 2018;83(6):530–41. https://doi.org/10.1016/j.biopsych.2017.07.017.
Chen PZ, He WJ, Zhu ZR, GJ E, Xu G, Chen DW, et al. Adenosine A2A receptor involves in neuroinflammation-mediated cognitive decline through activating microglia under acute hypobaric hypoxia. Behav Brain Res. 2018;347:99–107. https://doi.org/10.1016/j.bbr.2018.02.038.
Shen HY, Singer P, Lytle N, Wei CJ, Lan JQ, Williams-Karnesky RL, et al. Adenosine augmentation ameliorates psychotic and cognitive endophenotypes of schizophrenia. J Clin Invest. 2012;122(7):2567–77. https://doi.org/10.1172/JCI62378.
Mievis S, Blum D, Ledent C. A2A receptor knockout worsens survival and motor behaviour in a transgenic mouse model of Huntington’s disease. Neurobiol Dis. 2011;41(2):570–6. https://doi.org/10.1016/j.nbd.2010.09.021.
Ran H, Duan W, Gong Z, Xu S, Zhu H, Hou X, et al. Critical contribution of adenosine A2A receptors in bone marrow-derived cells to white matter lesions induced by chronic cerebral hypoperfusion. J Neuropathol Exp Neurol. 2015;74(4):305–18. https://doi.org/10.1097/NEN.0000000000000174.
Duan W, Wang H, Fan Q, Chen L, Huang H, Ran H. Cystatin F involvement in adenosine A2A receptor-mediated neuroinflammation in BV2 microglial cells. Sci Rep. 2018;8(1):6820. https://doi.org/10.1038/s41598-018-25031-5.
Duan W, Ran H, Zhou Z, He Q, Zheng J. Adenosine A2A receptor deficiency up-regulates cystatin F expression in white matter lesions induced by chronic cerebral hypoperfusion. PLoS One. 2012;7(12):e52566. https://doi.org/10.1371/journal.pone.0052566.
Yu L, Huang Z, Mariani J, Wang Y, Moskowitz M, Chen JF. Selective inactivation or reconstitution of adenosine A2A receptors in bone marrow cells reveals their significant contribution to the development of ischemic brain injury. Nat Med. 2004;10(10):1081–7. https://doi.org/10.1038/nm1103.
Jiwa NS, Garrard P, Hainsworth AH. Experimental models of vascular dementia and vascular cognitive impairment: a systematic review. J Neurochem. 2010;115(4):814–28. https://doi.org/10.1111/j.1471-4159.2010.06958.x.
Nishigaya K, Yagi S, Sato T, Kanemaru K, Nukui H. Impairment and restoration of the endothelial blood-brain barrier in the rat cerebral infarction model assessed by expression of endothelial barrier antigen immunoreactivity. Acta Neuropathol. 2000;99(3):231–7. https://doi.org/10.1007/pl00007432.
Wakita H, Tomimoto H, Akiguchi I, Kimura J. Glial activation and white matter changes in the rat brain induced by chronic cerebral hypoperfusion: an immunohistochemical study. Acta Neuropathol. 1994;87(5):484–92. https://doi.org/10.1007/bf00294175.
Gorelick PB, Scuteri A, Black SE, Decarli C, Greenberg SM, Iadecola C, et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42(9):2672–713. https://doi.org/10.1161/STR.0b013e3182299496.
Dichgans M, Leys D. Vascular cognitive impairment. Circ Res. 2017;120(3):573–91. https://doi.org/10.1161/CIRCRESAHA.116.308426.
Santiago A, Soares LM, Schepers M, Milani H, Vanmierlo T, Prickaerts J, et al. Roflumilast promotes memory recovery and attenuates white matter injury in aged rats subjected to chronic cerebral hypoperfusion. Neuropharmacology. 2018;138:360–70. https://doi.org/10.1016/j.neuropharm.2018.06.019.
Miyanohara J, Kakae M, Nagayasu K, Nakagawa T, Mori Y, Arai K, et al. TRPM2 channel aggravates CNS inflammation and cognitive impairment via activation of microglia in chronic cerebral hypoperfusion. J Neurosci. 2018;38(14):3520–33. https://doi.org/10.1523/JNEUROSCI.2451-17.2018.
Zhou Y, Zhang J, Wang L, Chen Y, Wan Y, He Y, et al. Interleukin-1beta impedes oligodendrocyte progenitor cell recruitment and white matter repair following chronic cerebral hypoperfusion. Brain Behav Immun. 2017;60:93–105. https://doi.org/10.1016/j.bbi.2016.09.024.
Fang C, Li Q, Min G, Liu M, Cui J, Sun J, et al. MicroRNA-181c ameliorates cognitive impairment induced by chronic cerebral hypoperfusion in rats. Mol Neurobiol. 2017;54(10):8370–85. https://doi.org/10.1007/s12035-016-0268-6.
Liu P, Liu P, Wang Z, Fang S, Liu Y, Wang J, et al. Inhibition of MicroRNA-96 ameliorates cognitive impairment and inactivation autophagy following chronic cerebral hypoperfusion in the rat. Cell Physiol Biochem. 2018;49(1):78–86. https://doi.org/10.1159/000492844.
Zou W, Song Y, Li Y, Du Y, Zhang X, Fu J. The role of autophagy in the correlation between neuron damage and cognitive impairment in rat chronic cerebral Hypoperfusion. Mol Neurobiol. 2018;55(1):776–91. https://doi.org/10.1007/s12035-016-0351-z.
Choi BR, Kim DH, Back DB, Kang CH, Moon WJ, Han JS, et al. Characterization of white matter injury in a rat model of chronic cerebral hypoperfusion. Stroke. 2016;47(2):542–7. https://doi.org/10.1161/STROKEAHA.115.011679.
Du SQ, Wang XR, Xiao LY, Tu JF, Zhu W, He T, et al. Molecular mechanisms of vascular dementia: what can be learned from animal models of chronic cerebral hypoperfusion? Mol Neurobiol. 2017;54(5):3670–82. https://doi.org/10.1007/s12035-016-9915-1.
Liang J, Li N, Zhang Y, Hou C, Yang X, Shimizu T, et al. Disinhibition of cathepsin C caused by cystatin F deficiency aggravates the demyelination in a cuprizone model. Front Mol Neurosci. 2016;9:152. https://doi.org/10.3389/fnmol.2016.00152.
Ma J, Tanaka KF, Shimizu T, Bernard CC, Kakita A, Takahashi H, et al. Microglial cystatin F expression is a sensitive indicator for ongoing demyelination with concurrent remyelination. J Neurosci Res. 2011;89(5):639–49. https://doi.org/10.1002/jnr.22567.
Chen JF. Adenosine receptor control of cognition in normal and disease. Int Rev Neurobiol. 2014;119:257–307. https://doi.org/10.1016/B978-0-12-801022-8.00012-X.
Faivre E, Coelho JE, Zornbach K, Malik E, Baqi Y, Schneider M, et al. Beneficial effect of a selective adenosine A2A receptor antagonist in the APPswe/PS1dE9 mouse model of Alzheimer’s disease. Front Mol Neurosci. 2018;11:235. https://doi.org/10.3389/fnmol.2018.00235.
Temido-Ferreira M, Ferreira DG, Batalha VL, Marques-Morgado I, Coelho JE, Pereira P, et al. Age-related shift in LTD is dependent on neuronal adenosine A2A receptors interplay with mGluR5 and NMDA receptors. Mol Psychiatry. 2018:1–25. https://doi.org/10.1038/s41380-018-0110-9.
Moscoso-Castro M, Gracia-Rubio I, Ciruela F, Valverde O. Genetic blockade of adenosine A2A receptors induces cognitive impairments and anatomical changes related to psychotic symptoms in mice. Eur Neuropsychopharmacol. 2016;26(7):1227–40. https://doi.org/10.1016/j.euroneuro.2016.04.003.
Li W, Silva HB, Real J, Wang YM, Rial D, Li P, et al. Inactivation of adenosine A2A receptors reverses working memory deficits at early stages of Huntington’s disease models. Neurobiol Dis. 2015;79:70–80. https://doi.org/10.1016/j.nbd.2015.03.030.
Gui L, Duan W, Tian H, Li C, Zhu J, Chen JF, et al. Adenosine a 2A receptor deficiency reduces striatal glutamate outflow and attenuates brain injury induced by transient focal cerebral ischemia in mice. Brain Res. 2009;1297:185–93. https://doi.org/10.1016/j.brainres.2009.08.050.
Zhao ZA, Zhao Y, Ning YL, Yang N, Peng Y, Li P, et al. Adenosine A2A receptor inactivation alleviates early-onset cognitive dysfunction after traumatic brain injury involving an inhibition of tau hyperphosphorylation. Transl Psychiatry. 2017;7(5):e1123. https://doi.org/10.1038/tp.2017.98.
Sandau US, Colino-Oliveira M, Jones A, Saleumvong B, Coffman SQ, Liu L, et al. Adenosine kinase deficiency in the brain results in maladaptive synaptic plasticity. J Neurosci. 2016;36(48):12117–28. https://doi.org/10.1523/JNEUROSCI.2146-16.2016.
Hu Q, Ren X, Liu Y, Li Z, Zhang L, Chen X, et al. Aberrant adenosine A2A receptor signaling contributes to neurodegeneration and cognitive impairments in a mouse model of synucleinopathy. Exp Neurol. 2016;283(Pt A):213–23. https://doi.org/10.1016/j.expneurol.2016.05.040.
Antonini A, Poewe W. Adenosine A2A receptor antagonists in Parkinson’s disease: still in the running. The Lancet Neurology. 2014;13(8):748–9. https://doi.org/10.1016/S1474-4422(14)70153-X.
Acknowledgments
We would like to thank Dr. Jiang-Fan Chen (Boston University School of Medicine, USA) for providing the adenosine A2A receptor knockout mice.
Availability of Data and Materials
Data supporting the conclusions are presented in the manuscript. Any information regarding the animal model, the experimental methods used, and the data in this paper are available to scientific communities upon direct contact to the authors.
Funding
This work was supported by grants from Chongqing Natural Science Foundation (cstc2018jcyjAX0203), National Natural Science Foundation of China (No. 81701146), Third Military Medical University “Miaopu” talents scheme (XZ-2019-505-064), and Southwest Hospital Science Foundation (SWH2018QNKJ-02).
Author information
Authors and Affiliations
Contributions
This study was based on the original idea of ZHZ and KNC. HR, JCY, JLH, and JW conducted the experiments. JCY and ZHZ performed data analyses. HR and JCY drafted the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
All animal experiments and care were approved by the Third Military Medical University Committee on Ethics in the Care and Use of Laboratory Animals.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare that they have no conflict of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. BMDC A2ARs attenuate cognitive impairment after CHWMLs through anti-inflammatory effects.
2. Non-BMDC A2ARs can aggravate cognitive impairment through promoting inflammation.
3. Global A2ARs activation could alleviate cognitive impairment after CHWMLs.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ran, H., Yuan, J., Huang, J. et al. Adenosine A2A Receptors in Bone Marrow-Derived Cells Attenuate Cognitive Impairment in Mice After Chronic Hypoperfusion White Matter Injury. Transl. Stroke Res. 11, 1028–1040 (2020). https://doi.org/10.1007/s12975-019-00778-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-019-00778-9