Abstract
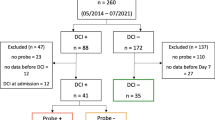
Impaired cerebral autoregulation and neurovascular coupling (NVC) contribute to delayed cerebral ischemia after subarachnoid hemorrhage (SAH). Retinal vessel analysis (RVA) allows non-invasive assessment of vessel dimension and NVC hereby demonstrating a predictive value in the context of various neurovascular diseases. Using RVA as a translational approach, we aimed to assess the retinal vessels in patients with SAH. RVA was performed prospectively in 24 patients with acute SAH (group A: day 5–14), in 11 patients 3 months after ictus (group B: day 90 ± 35), and in 35 age-matched healthy controls (group C). Data was acquired using a Retinal Vessel Analyzer (Imedos Systems UG, Jena) for examination of retinal vessel dimension and NVC using flicker-light excitation. Diameter of retinal vessels—central retinal arteriolar and venular equivalent—was significantly reduced in the acute phase (p < 0.001) with gradual improvement in group B (p < 0.05). Arterial NVC of group A was significantly impaired with diminished dilatation (p < 0.001) and reduced area under the curve (p < 0.01) when compared to group C. Group B showed persistent prolonged latency of arterial dilation (p < 0.05). Venous NVC was significantly delayed after SAH compared to group C (A p < 0.001; B p < 0.05). To our knowledge, this is the first clinical study to document retinal vasoconstriction and impairment of NVC in patients with SAH. Using non-invasive RVA as a translational approach, characteristic patterns of compromise were detected for the arterial and venous compartment of the neurovascular unit in a time-dependent fashion. Recruitment will continue to facilitate a correlation analysis with clinical course and outcome.




Similar content being viewed by others
References
Budohoski KP, Czosnyka M, Kirkpatrick PJ, Smielewski P, Steiner LA, Pickard JD. Clinical relevance of cerebral autoregulation following subarachnoid haemorrhage. Nat Rev Neurol. 2013;9(3):152–63. https://doi.org/10.1038/nrneurol.2013.11.
Foreman B. The pathophysiology of delayed cerebral ischemia. J Clin Neurophysiol. 2016;33(3):174–82. https://doi.org/10.1097/WNP.0000000000000273.
Vergouwen MD, Vermeulen M, van Gijn J, Rinkel GJ, Wijdicks EF, Muizelaar JP, et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke. 2010;41(10):2391–5. https://doi.org/10.1161/STROKEAHA.110.589275.
Jaeger M, Soehle M, Schuhmann MU, Meixensberger J. Clinical significance of impaired cerebrovascular autoregulation after severe aneurysmal subarachnoid hemorrhage. Stroke. 2012;43(8):2097–101. https://doi.org/10.1161/STROKEAHA.112.659888.
Budohoski KP, Czosnyka M, Smielewski P, Kasprowicz M, Helmy A, Bulters D, et al. Impairment of cerebral autoregulation predicts delayed cerebral ischemia after subarachnoid hemorrhage: a prospective observational study. Stroke. 2012;43(12):3230–7. https://doi.org/10.1161/STROKEAHA.112.669788.
Iadecola C. The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron. 2017;96(1):17–42. https://doi.org/10.1016/j.neuron.2017.07.030.
Balbi M, Koide M, Schwarzmaier SM, Wellman GC, Plesnila N. Acute changes in neurovascular reactivity after subarachnoid hemorrhage in vivo. J Cereb Blood Flow Metab. 2015; https://doi.org/10.1177/0271678X15621253.
Balbi M, Koide M, Wellman GC, Plesnila N. Inversion of neurovascular coupling after subarachnoid hemorrhage in vivo. J Cereb Blood Flow Metab. 2017:271678X16686595. doi:https://doi.org/10.1177/0271678X16686595.
Winkler MK, Chassidim Y, Lublinsky S, Revankar GS, Major S, Kang EJ, et al. Impaired neurovascular coupling to ictal epileptic activity and spreading depolarization in a patient with subarachnoid hemorrhage: possible link to blood-brain barrier dysfunction. Epilepsia. 2012;53(Suppl 6):22–30. https://doi.org/10.1111/j.1528-1167.2012.03699.x.
da Costa L, Fierstra J, Fisher JA, Mikulis DJ, Han JS, Tymianski M. BOLD MRI and early impairment of cerebrovascular reserve after aneurysmal subarachnoid hemorrhage. J Magn Reson Imaging. 2014;40(4):972–9. https://doi.org/10.1002/jmri.24474.
Patton N, Aslam T, Macgillivray T, Pattie A, Deary IJ, Dhillon B. Retinal vascular image analysis as a potential screening tool for cerebrovascular disease: a rationale based on homology between cerebral and retinal microvasculatures. J Anat. 2005;206(4):319–48. https://doi.org/10.1111/j.1469-7580.2005.00395.x.
London A, Benhar I, Schwartz M. The retina as a window to the brain-from eye research to CNS disorders. Nat Rev Neurol. 2013;9(1):44–53. https://doi.org/10.1038/nrneurol.2012.227.
Heitmar R, Cubbidge RP, Lip GY, Gherghel D, Blann AD. Altered blood vessel responses in the eye and finger in coronary artery disease. Invest Ophthalmol Vis Sci. 2011;52(9):6199–205. https://doi.org/10.1167/iovs.10-6628.
Wong TY, Klein R, Couper DJ, Cooper LS, Shahar E, Hubbard LD, et al. Retinal microvascular abnormalities and incident stroke: the atherosclerosis risk in communities study. Lancet. 2001;358(9288):1134–40. https://doi.org/10.1016/S0140-6736(01)06253-5.
Fountas KN, Kapsalaki EZ, Lee GP, Machinis TG, Grigorian AA, Robinson JS, et al. Terson hemorrhage in patients suffering aneurysmal subarachnoid hemorrhage: predisposing factors and prognostic significance. J Neurosurg. 2008;109(3):439–44. https://doi.org/10.3171/JNS/2008/109/9/0439.
Obuchowska I, Turek G, Mariak Z, Kochanowicz J, Mariak Z. Late ophthalmological assessment of patients with subarachnoid hemorrhage and clipping of cerebral aneurysm. Acta Neurochir. 2011;153(11):2127–36. https://doi.org/10.1007/s00701-011-1161-8.
Newman EA. Functional hyperemia and mechanisms of neurovascular coupling in the retinal vasculature. J Cereb Blood Flow Metab. 2013;33(11):1685–95. https://doi.org/10.1038/jcbfm.2013.145.
Garhofer G, Bek T, Boehm AG, Gherghel D, Grunwald J, Jeppesen P, et al. Use of the retinal vessel analyzer in ocular blood flow research. Acta Ophthalmol. 2010;88(7):717–22. https://doi.org/10.1111/j.1755-3768.2009.01587.x.
Kotliar KE, Vilser W, Nagel E, Lanzl IM. Retinal vessel reaction in response to chromatic flickering light. Graefes Arch Clin Exp Ophthalmol. 2004;242(5):377–92. https://doi.org/10.1007/s00417-003-0847-x.
Polak K, Dorner G, Kiss B, Polska E, Findl O, Rainer G, et al. Evaluation of the Zeiss retinal vessel analyser. Br J Ophthalmol. 2000;84(11):1285–90.
Albanna W, Conzen C, Weiss M, Clusmann H, Fuest M, Mueller M, et al. Retinal vessel analysis (RVA) in the context of subarachnoid hemorrhage—a proof of concept study. PLoS One. 2016;11(7):e0158781. https://doi.org/10.1371/journal.pone.0158781.
MacCormick IJ, Beare NA, Taylor TE, Barrera V, White VA, Hiscott P, et al. Cerebral malaria in children: using the retina to study the brain. Brain. 2014;137(Pt 8):2119–42. https://doi.org/10.1093/brain/awu001.
Diringer MN, Bleck TP, Claude Hemphill J 3rd, Menon D, Shutter L, Vespa P, et al. Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocrit Care. 2011;15(2):211–40. https://doi.org/10.1007/s12028-011-9605-9.
Rossini PM, Altamura C, Ferretti A, Vernieri F, Zappasodi F, Caulo M, et al. Does cerebrovascular disease affect the coupling between neuronal activity and local haemodynamics? Brain. 2004;127(Pt 1):99–110. https://doi.org/10.1093/brain/awh012.
Albanna W, Weiss M, Muller M, Brockmann MA, Rieg A, Conzen C, et al. Endovascular rescue therapies for refractory vasospasm after subarachnoid hemorrhage: a prospective evaluation study using multimodal, continuous event neuromonitoring. Neurosurgery. 2017;80(6):942–9. https://doi.org/10.1093/neuros/nyw132.
Nagel E, Vilser W, Fink A, Riemer T. Static vessel analysis in nonmydriatic and mydriatic images. Klin Monatsbl Augenheilkd. 2007;224(5):411–6. https://doi.org/10.1055/s-2007-963093.
Yu PK, Balaratnasingam C, Cringle SJ, McAllister IL, Provis J, Yu DY. Microstructure and network organization of the microvasculature in the human macula. Invest Ophthalmol Vis Sci. 2010;51(12):6735–43. https://doi.org/10.1167/iovs.10-5415.
Dorner GT, Garhofer G, Kiss B, Polska E, Polak K, Riva CE, et al. Nitric oxide regulates retinal vascular tone in humans. Am J Physiol Heart Circ Physiol. 2003;285(2):H631–6. https://doi.org/10.1152/ajpheart.00111.2003.
Metea MR, Newman EA. Glial cells dilate and constrict blood vessels: a mechanism of neurovascular coupling. J Neurosci. 2006;26(11):2862–70. https://doi.org/10.1523/JNEUROSCI.4048-05.2006.
Petzold GC, Murthy VN. Role of astrocytes in neurovascular coupling. Neuron. 2011;71(5):782–97. https://doi.org/10.1016/j.neuron.2011.08.009.
Luksch A, Garhofer G, Imhof A, Polak K, Polska E, Dorner GT, et al. Effect of inhalation of different mixtures of O(2) and CO(2) on retinal blood flow. Br J Ophthalmol. 2002;86(10):1143–7.
Nagaoka T, Mori F, Yoshida A. Retinal artery response to acute systemic blood pressure increase during cold pressor test in humans. Invest Ophthalmol Vis Sci. 2002;43(6):1941–5.
Liew G, Sharrett AR, Wang JJ, Klein R, Klein BE, Mitchell P, et al. Relative importance of systemic determinants of retinal arteriolar and venular caliber: the atherosclerosis risk in communities study. Arch Ophthalmol. 2008;126(10):1404–10. https://doi.org/10.1001/archopht.126.10.1404.
Obuchowska I, Turek G, Mariak Z, Mariak Z. Early intraocular complications of subarachnoid haemorrhage after aneurysm rupture. Neuro-Ophthalmology. 2014;38(4):199–204. https://doi.org/10.3109/01658107.2014.911918.
Sehba FA, Hou J, Pluta RM, Zhang JH. The importance of early brain injury after subarachnoid hemorrhage. Prog Neurobiol. 2012;97(1):14–37. https://doi.org/10.1016/j.pneurobio.2012.02.003.
Ogoh S. Relationship between cognitive function and regulation of cerebral blood flow. J Physiol Sci. 2017; https://doi.org/10.1007/s12576-017-0525-0.
Al-Khindi T, Macdonald RL, Schweizer TA. Cognitive and functional outcome after aneurysmal subarachnoid hemorrhage. Stroke. 2010;41(8):e519–36. https://doi.org/10.1161/STROKEAHA.110.581975.
Acknowledgements
The authors are grateful to Mrs. Jasmin Dell’Anna and Professor Klaus Rademacher (Faculty of Medical Engineering, Helmholtz-Institute for Biomedical Engineering, RWTH Aachen University) for supporting the constructional set up of the imaging device.
Funding
CC was supported by the START-Program of the Faculty of Medicine (RWTH Aachen University) and by the Foundation of Neurosurgical Research (German Society of Neurosurgery, 2016). WA was supported by funding from the German Society of Neurointensive and Emergency Medicine (Junior research award 2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
IMEDOS Systems provided the Retinal Vessel Analyzer for research purposes only. WV is affiliated to IMEDOS Systems as the company’s CEO. WV did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
The other authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Conzen, C., Albanna, W., Weiss, M. et al. Vasoconstriction and Impairment of Neurovascular Coupling after Subarachnoid Hemorrhage: a Descriptive Analysis of Retinal Changes. Transl. Stroke Res. 9, 284–293 (2018). https://doi.org/10.1007/s12975-017-0585-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-017-0585-8