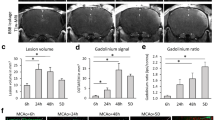
Abstract
Immunity and inflammation play critical roles in the pathogenesis of acute ischemic stroke. Therefore, immune intervention, as a new therapeutic strategy, is worthy of exploration. Here, we tested the inflammation modulator, vinpocetine, for its effect on the outcomes of stroke. For this multi-center study, we recruited 60 patients with anterior cerebral circulation occlusion and onset of stroke that had exceeded 4.5 h but lasted less than 48 h. These patients, after random division into two groups, received either standard management alone (controls) or standard management plus vinpocetine (30 mg per day intravenously for 14 consecutive days, Gedeon Richter Plc., Hungary). Vinpocetine treatment did not change the lymphocyte count; however, nuclear factor kappa-light-chain-enhancer of activated B cell activation was inhibited as seen not only by the increased transcription of IκBα mRNA but also by the impeded phosphorylation and degradation of IκBα and subsequent induction of pro-inflammatory mediators. These effects led to significantly reduced secondary lesion enlargement and an attenuated inflammation reaction. Compared to controls, patients treated with vinpocetine had a better recovery of neurological function and improved clinical outcomes during the acute phase and at 3-month follow-up. These findings identify vinpocetine as an inflammation modulator that could improve clinical outcomes after acute ischemic stroke. This study also indicated the important role of immunity and inflammation in the pathogenesis of acute ischemic stroke and the significance of immunomodulatory treatment. Clinical Trial Registration Information: www.clinicaltrials.gov. Identifier: NCT02878772






Similar content being viewed by others
References
Lapchak PA, Zhang JH. The high cost of stroke and stroke cytoprotection research. Transl Stroke Res. 2016; doi:10.1007/s12975-016-0518-y.
Cassidy JM, Cramer SC. Spontaneous and therapeutic-induced mechanisms of functional recovery after stroke. Transl Stroke Res. 2016; doi:10.1007/s12975-016-0467-5.
Liesz A, Kleinschnitz C. Regulatory T cells in post-stroke immune homeostasis. Transl Stroke Res. 2016;7(4):313–21. doi:10.1007/s12975-016-0465-7.
Xia Y, Cai W, Thomson AW, Hu X. Regulatory T cell therapy for ischemic stroke: how far from clinical translation? Transl Stroke Res. 2016;7(5):415–9. doi:10.1007/s12975-016-0476-4.
Eltzschig HK, Carmeliet P. Hypoxia and inflammation. N Engl J Med. 2011;364(7):656–65. doi:10.1056/NEJMra0910283.
Iadecola C, Anrather J. The immunology of stroke: from mechanisms to translation. Nat Med. 2011;17(7):796–808. doi:10.1038/nm.2399.
Jin R, Liu L, Zhang S, Nanda A, Li G. Role of inflammation and its mediators in acute ischemic stroke. J Cardiovasc Transl Res. 2013;6(5):834–51. doi:10.1007/s12265-013-9508-6.
Tulsulkar J, Glueck B, Hinds TD Jr, Shah ZA. Ginkgo biloba Extract prevents female mice from ischemic brain damage and the mechanism is independent of the HO1/Wnt pathway. Transl Stroke Res. 2016;7(2):120–31. doi:10.1007/s12975-015-0433-7.
Hasegawa Y, Suzuki H, Altay O, Rolland W, Zhang JH. Role of the sphingosine metabolism pathway on neurons against experimental cerebral ischemia in rats. Transl Stroke Res. 2013;4(5):524–32. doi:10.1007/s12975-013-0260-7.
Kim EJ, Raval AP, Hirsch N, Perez-Pinzon MA. Ischemic preconditioning mediates cyclooxygenase-2 expression via nuclear factor-kappa B activation in mixed cortical neuronal cultures. Transl Stroke Res. 2010;1(1):40–7.
Kalra L, Irshad S, Hodsoll J, Simpson M, Gulliford M, Smithard D, et al. Prophylactic antibiotics after acute stroke for reducing pneumonia in patients with dysphagia (STROKE-INF): a prospective, cluster-randomised, open-label, masked endpoint, controlled clinical trial. Lancet. 2015; doi:10.1016/s0140-6736(15)00126-9.
Westendorp WF, Vermeij JD, Zock E, Hooijenga IJ, Kruyt ND, Bosboom HJ, et al. The Preventive Antibiotics in Stroke Study (PASS): a pragmatic randomised open-label masked endpoint clinical trial. Lancet. 2015;385(9977):1519–26. doi:10.1016/S0140-6736(14)62456-9.
Meisel A, Smith CJ. Prevention of stroke-associated pneumonia: where next? Lancet. 2015; doi:10.1016/s0140-6736(15)00127-0.
Fu Y, Zhang N, Ren L, Yan Y, Sun N, Li YJ, et al. Impact of an immune modulator fingolimod on acute ischemic stroke. Proc Natl Acad Sci U S A. 2014;111(51):18315–20. doi:10.1073/pnas.1416166111.
Fu Y, Hao J, Zhang N, Ren L, Sun N, Li YJ, et al. Fingolimod for the treatment of intracerebral hemorrhage: a 2-arm proof-of-concept study. JAMA Neurol. 2014;71(9):1092–101. doi:10.1001/jamaneurol.2014.1065.
Zhu Z, Fu Y, Tian D, Sun N, Han W, Chang G, et al. Combination of an immune modulator fingolimod with alteplase in acute ischemic stroke: a pilot trial. Circulation. 2015; doi:10.1161/circulationaha.115.016371.
Fu Y, Liu Q, Anrather J, Shi FD. Immune interventions in stroke. Nat Rev Neurol. 2015;11(9):524–35. doi:10.1038/nrneurol.2015.144.
Vas A, Gulyas B, Szabo Z, Bonoczk P, Csiba L, Kiss B, et al. Clinical and non-clinical investigations using positron emission tomography, near infrared spectroscopy and transcranial Doppler methods on the neuroprotective drug vinpocetine: a summary of evidences. J Neurol Sci. 2002;203-204:259–62.
Gulyas B, Halldin C, Karlsson P, Chou YH, Swahn CG, Bonoczk P, et al. Cerebral uptake and metabolism of (11C) vinpocetine in monkeys: PET studies. Orv Hetil. 1999;140(30):1687–91.
Gulyas B, Toth M, Schain M, Airaksinen A, Vas A, Kostulas K, et al. Evolution of microglial activation in ischaemic core and peri-infarct regions after stroke: a PET study with the TSPO molecular imaging biomarker [((11))C]vinpocetine. J Neurol Sci. 2012;320(1–2):110–7. doi:10.1016/j.jns.2012.06.026.
Vas A, Shchukin Y, Karrenbauer VD, Cselenyi Z, Kostulas K, Hillert J, et al. Functional neuroimaging in multiple sclerosis with radiolabelled glia markers: preliminary comparative PET studies with [11C]vinpocetine and [11C]PK11195 in patients. J Neurol Sci. 2008;264(1–2):9–17. doi:10.1016/j.jns.2007.07.018.
Szobor A, Klein M. Ethyl apovincaminate therapy in neurovascular diseases. Arzneimittelforschung. 1976;26(10a):1984–9.
Bonoczk P, Gulyas B, Adam-Vizi V, Nemes A, Karpati E, Kiss B, et al. Role of sodium channel inhibition in neuroprotection: effect of vinpocetine. Brain Res Bull. 2000;53(3):245–54.
Feigin VL, Doronin BM, Popova TF, Gribatcheva EV, Tchervov DV. Vinpocetine treatment in acute ischaemic stroke: a pilot single-blind randomized clinical trial. Eur J Neurol. 2001;8(1):81–5.
Patyar S, Prakash A, Modi M, Medhi B. Role of vinpocetine in cerebrovascular diseases. Pharmacol Rep: PR. 2011;63(3):618–28.
Bagoly E, Feher G, Szapary L. The role of vinpocetine in the treatment of cerebrovascular diseases based in human studies. Orv Hetil. 2007;148(29):1353–8. doi:10.1556/oh.2007.28115.
Jeon KI, Xu X, Aizawa T, Lim JH, Jono H, Kwon DS, et al. Vinpocetine inhibits NF-kappaB-dependent inflammation via an IKK-dependent but PDE-independent mechanism. Proc Natl Acad Sci U S A. 2010;107(21):9795–800. doi:10.1073/pnas.0914414107.
Medina AE. Vinpocetine as a potent antiinflammatory agent. Proc Natl Acad Sci U S A. 2010;107(22):9921–2. doi:10.1073/pnas.1005138107.
Wang H, Zhang K, Zhao L, Tang J, Gao L, Wei Z. Anti-inflammatory effects of vinpocetine on the functional expression of nuclear factor-kappa B and tumor necrosis factor-alpha in a rat model of cerebral ischemia-reperfusion injury. Neurosci Lett. 2014;566:247–51. doi:10.1016/j.neulet.2014.02.045.
Balestreri R, Fontana L, Astengo F. A double-blind placebo controlled evaluation of the safety and efficacy of vinpocetine in the treatment of patients with chronic vascular senile cerebral dysfunction. J Am Geriatr Soc. 1987;35(5):425–30.
Savman K, Heyes MP, Svedin P, Karlsson A. Microglia/macrophage-derived inflammatory mediators galectin-3 and quinolinic acid are elevated in cerebrospinal fluid from newborn infants after birth asphyxia. Transl Stroke Res. 2013;4(2):228–35. doi:10.1007/s12975-012-0216-3.
Wu LJ. Microglial voltage-gated proton channel Hv1 in ischemic stroke. Transl Stroke Res. 2014;5(1):99–108. doi:10.1007/s12975-013-0289-7.
Shi Y, Leak RK, Keep RF, Chen J. Translational stroke research on blood-brain barrier damage: challenges, perspectives, and goals. Transl Stroke Res. 2016;7(2):89–92. doi:10.1007/s12975-016-0447-9.
Reuter B, Rodemer C, Grudzenski S, Meairs S, Bugert P, Hennerici MG, et al. Effect of simvastatin on MMPs and TIMPs in human brain endothelial cells and experimental stroke. Transl Stroke Res. 2015;6(2):156–9. doi:10.1007/s12975-014-0381-7.
Garg M, Gupta RK, Husain M, Chawla S, Chawla J, Kumar R, et al. Brain abscesses: etiologic categorization with in vivo proton MR spectroscopy. Radiology. 2004;230(2):519–27. doi:10.1148/radiol.2302021317.
Gaudinski MR, Henning EC, Miracle A, Luby M, Warach S, Latour LL. Establishing final infarct volume: stroke lesion evolution past 30 days is insignificant. Stroke. 2008;39(10):2765–8. doi:10.1161/strokeaha.107.512269.
Zhu Z, Fu Y, Tian D, Sun N, Han W, Chang G, et al. Combination of the immune modulator fingolimod with Alteplase in acute ischemic stroke: a pilot trial. Circulation. 2015;132(12):1104–12. doi:10.1161/CIRCULATIONAHA.115.016371.
Young AC, Yiannoutsos CT, Hegde M, Lee E, Peterson J, Walter R, et al. Cerebral metabolite changes prior to and after antiretroviral therapy in primary HIV infection. Neurology. 2014;83(18):1592–600. doi:10.1212/wnl.0000000000000932.
Voevodskaya O, Sundgren PC, Strandberg O, Zetterberg H, Minthon L, Blennow K, et al. Myo-inositol changes precede amyloid pathology and relate to APOE genotype in Alzheimer disease. Neurology. 2016;86(19):1754–61. doi:10.1212/wnl.0000000000002672.
Cousins JP. Clinical MR spectroscopy: fundamentals, current applications, and future potential. AJR Am J Roentgenol. 1995;164(6):1337–47. doi:10.2214/ajr.164.6.7754871.
Provencher SW. Automatic quantitation of localized in vivo 1H spectra with LCModel. NMR Biomed. 2001;14(4):260–4.
Valenzuela MJ, Sachdev P. Magnetic resonance spectroscopy in AD. Neurology. 2001;56(5):592–8.
Valcour V, Chalermchai T, Sailasuta N, Marovich M, Lerdlum S, Suttichom D, et al. Central nervous system viral invasion and inflammation during acute HIV infection. J Infect Dis. 2012;206(2):275–82. doi:10.1093/infdis/jis326.
Valero IP, Baeza AG, Hernandez-Tamames JA, Monge S, Arnalich F, Arribas JR. Cerebral volumes, neuronal integrity and brain inflammation measured by MRI in patients receiving PI monotherapy or triple therapy. J Int AIDS Soc. 2014;17(4 Suppl 3):19578. doi:10.7448/ias.17.4.19578.
Ciccarelli O, Barkhof F, Bodini B, De Stefano N, Golay X, Nicolay K, et al. Pathogenesis of multiple sclerosis: insights from molecular and metabolic imaging. Lancet Neurol. 2014;13(8):807–22. doi:10.1016/s1474-4422(14)70101-2.
Roricht S, Meyer BU, Grafin von Einsiedel H, Sander B. A solitary toxoplasmosis focus simulating a brain tumor as the first manifestation of AIDS. RoFo: Fortschritte auf dem Gebiete der Rontgenstrahlen und der Nuklearmedizin. 1997;167(2):201–3. doi:10.1055/s-2007-1015517.
Luyten PR, Marien AJ, Heindel W, van Gerwen PH, Herholz K, den Hollander JA, et al. Metabolic imaging of patients with intracranial tumors: H-1 MR spectroscopic imaging and PET. Radiology. 1990;176(3):791–9. doi:10.1148/radiology.176.3.2389038.
Marik PE. Aspiration pneumonitis and aspiration pneumonia. N Engl J Med. 2001;344(9):665–71. doi:10.1056/nejm200103013440908.
Meisel C, Meisel A. Suppressing immunosuppression after stroke. N Engl J Med. 2011;365(22):2134–6. doi:10.1056/NEJMcibr1112454.
Perry VH, Gordon S. Macrophages and microglia in the nervous system. Trends Neurosci. 1988;11(6):273–7.
Mahad DH, Trapp BD, Lassmann H. Pathological mechanisms in progressive multiple sclerosis. Lancet Neurol. 2015;14(2):183–93. doi:10.1016/s1474-4422(14)70256-x.
Fernando KT, McLean MA, Chard DT, MacManus DG, Dalton CM, Miszkiel KA, et al. Elevated white matter myo-inositol in clinically isolated syndromes suggestive of multiple sclerosis. Brain. 2004;127(Pt 6):1361–9. doi:10.1093/brain/awh153.
Chamorro A, Meisel A, Planas AM, Urra X, van de Beek D, Veltkamp R. The immunology of acute stroke. Nat Rev Neurol. 2012;8(7):401–10. doi:10.1038/nrneurol.2012.98.
Perez JM, Chirieleison SM, Abbott DW. An IkappaB kinase-regulated feedforward circuit prolongs inflammation. Cell Rep. 2015; doi:10.1016/j.celrep.2015.06.050.
Acknowledgements
We thank our patients for participating this study and to Yao YR and Lu HY for facilitating recruitment of the patients; we also thank Shi HL for technical support. This work was financially supported by the National Basic Research Program of China (2013CB966900 to FDS), the National Natural Science Foundation of China (81571600, 81322018, 81273287, and 81100887 to JWH), the Youth Top-Notch Talent Support Program, and the National Key Clinical Specialty Construction Project of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
This work was financially supported by the National Basic Research Program of China (2013CB966900 to FDS), the National Natural Science Foundation of China (81571600, 81322018, 81273287, and 81100887 to JWH), the Youth Top-Notch Talent Support Program, and the National Key Clinical Specialty Construction Project of China.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Electronic Supplementary Material
Supplemental Figure 1
Comparison of infection rates in recipients of vinpocetine or standard treatment. In another retrospective study of infection associated with stroke, we collected records from 200 patients with acute ischemic stroke who received only standard treatment, then compared the scores with those from 200 age-, gender- and infarct volume-matched patients with acute ischemic stroke (control vs vinpocetine: Female, n, 74 vs 79; age, year, 61.3 ± 16.7 vs 65.1 ± 19.2) who received standard treatment and vinpocetine (30 mg per day intravenously for 14 consecutive days). The inclusion and exclusion criteria were as follows: Inclusions: (1) >18 years of age; (2) Anterior-circulation ischemic stroke: All patients had symptoms of focal neurological deficits and radiological evidence (nuclear magnetic resonance, NMR) of a concurrent ischemic brain lesion; (3) measurable neurological deficit (NIHSS ≥3). Exclusion criteria were (1) evidence of other diseases of the central nervous system (CNS); (2) diabetes mellitus; (3) tumor and hematological system diseases; (4) any infection before acute ischemic stroke; (5) concomitant use of antineoplastic or immune modulating therapies. (A) The infection rate of vinpocetine group (13%) was lower than that of the control group (28%). Statistically significant differences were found between these groups (P = 0.014 Chi-square text). (B) Among the infected patients of vinpocetine group, 88% were respiratory infection and the last 12% were urinary infection. In the control group, respiratory infections account for 74%, urinary infections account for 20%, oral and ocula infections account for 3% respectively. (DOCX 48.1 kb).
Supplemental Figure 2
Dynamics of lymphocyte subsets during vinpocetine treatment. The dynamic changes of lymphocyte subsets were monitored at baseline (<48 h after stroke onset) in whole-blood samples from all patients to receive vinpocetine, preceding the first dose, and then at days 3 and 7 after the first vinpocetine dose. These values were compared with those from control patients (no vinpocetine) at the same time points. Peripheral blood mononuclear cells were isolated from the whole-blood specimens and stained with antibodies to CD4-FITC, CD8-PE, CD3-PerCP, CD19-APC, CD11c-FITC and CD56-APC (BD Biosciences, Franklin Lakes, NJ, USA). Data were acquired using a FACS Calibur (Becton Dickinson Immunocytometry Systems, San Jose, CA, USA) and analyzed with Flow Jo software (Tree Star, Ashland, OR, USA). At baseline, day 3 and day 7 after symptom onset, the lymphocyte counts and percentages of CD4+ T, CD8+ T, CD19+ B, CD56+ NK and CD11c+ DC cells in the vinpocetine group were similar to those of controls. These results suggest that vinpocetine exerts anti-inflammatory effects but not by decreasing the number of lymphocytes. Data represent means ± SE (DOCX 68.9 kb).
Supplementary Figure 3
Baseline characteristics. (a) NIHSS score and (b) lesion volume of two goups at baseline. Values are means ± SE. (DOCX 67.3 kb).
Supplementary Figure 4
Lesion volumes compared between two groups. Lesion volumes did not differ significantly between the vinpocetine-treated group and control group at baseline and day 7. Values are means ± SE. (DOCX 46.4 kb).
Supplemental Table 1
(DOCX 30 kb).
Rights and permissions
About this article
Cite this article
Zhang, F., Yan, C., Wei, C. et al. Vinpocetine Inhibits NF-κB-Dependent Inflammation in Acute Ischemic Stroke Patients. Transl. Stroke Res. 9, 174–184 (2018). https://doi.org/10.1007/s12975-017-0549-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-017-0549-z