Abstract
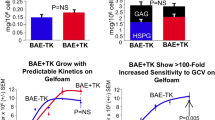
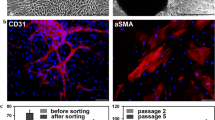
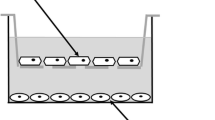
Carotid artery disease is a widespread cause of morbidity and mortality. Porcine models of vascular disease are well established in vivo, but existing endothelial systems in vitro (e.g. human umbilical vein endothelial cells, rat aortic endothelial cultures) poorly reflect carotid endothelium. A reliable in vitro assay would improve design of in vivo experiments and allow reduction and refinement of animal use. This study aimed (1) to develop ex vivo endothelial cultures from porcine carotid and (2) to test whether these were suitable for lentivector-mediated transgene delivery. Surplus carotid arteries were harvested from young adult female Large White pigs within 10 min post-mortem. Small sectors of carotid artery wall (approximately 4 mm × 4 mm squares) were immobilised in a stable gel matrix. Cultures were exposed to HIV-derived lentivector (LV) encoding a reporter transgene or the equivalent integration-deficient vector (IDLV). After 7–14 days in vitro, cultures were fixed and labelled histochemically. Thread-like multicellular outgrowths were observed that were positive for endothelial cell markers (CD31, VEGFR2, von Willebrand factor). A minority of cells co-labelled for smooth muscle markers. Sensitivity to cytotoxic agents (paclitaxel, cycloheximide, staurosporine) was comparable to that in cell cultures, indicating that the gel matrix permits diffusive access of small pharmacological molecules. Transgene-expressing cells were more abundant following exposure to LV than IDLV (4.7, 0.1 % of cells, respectively). In conclusion, ex vivo adult porcine carotid artery produced endothelial cell outgrowths that were effectively transduced by LV. This system will facilitate translation of novel therapies to clinical trials, with reduction and refinement of in vivo experiments.





Similar content being viewed by others
Abbreviations
- EGFP:
-
Enhanced green fluorescent protein
References
Chen D, Morgan F, Berton I, et al. Developing a porcine transplantation model: efficient gene transfer into porcine vascular cells. Transplantation. 2004;77:1443–51.
Pfeiffer S, Zorn III GL, Blair KS, et al. Hyperacute lung rejection in the pig-to-human model 4: evidence for complement and antibody independent mechanisms. Transplantation. 2005;79:662–71.
Herbert PE, Morgan MF, Berton I, et al. A pig allograft model of antibody-mediated rejection. Transpl Immunol. 2008;19:167–72.
Tanaka Y, Imai H, Konno K, et al. Experimental model of lacunar infarction in the gyrencephalic brain of the miniature pig: neurological assessment and histological, immunohistochemical, and physiological evaluation of dynamic corticospinal tract deformation. Stroke. 2008;39:205–12.
Versari D, Gossl M, Mannheim D, et al. Hypertension and hypercholesterolemia differentially affect the function and structure of pig carotid artery. Hypertension. 2007;50:1063–8.
De Salles AA, Solberg TD, Mischel P, et al. Arteriovenous malformation animal model for radiosurgery: the rete mirabile. AJNR Am J Neuroradiol. 1996;17:1451–8.
Lind NM, Moustgaard A, Jelsing J, Vajta G, Cumming P, Hansen AK. The use of pigs in neuroscience: modeling brain disorders. Neurosci Biobehav Rev. 2007;31:728–51.
McGrath P. Observations on the intracranial carotid rete and the hypophysis in the mature female pig and sheep. J Anat. 1977;124:689–99.
Moss RF, Parmar NK, Tighe D, Davies DC. Adrenergic agents modify cerebral edema and microvessel ultrastructure in porcine sepsis. Crit Care Med. 2004;32:1916–21.
Whitelaw CB, Lillico SG, King T. Production of transgenic farm animals by viral vector-mediated gene transfer. Reprod Domest Anim. 2008;43 Suppl 2:355–8.
Zhu WH, MacIntyre A, Nicosia RF. Regulation of angiogenesis by vascular endothelial growth factor and angiopoietin-1 in the rat aorta model: distinct temporal patterns of intracellular signaling correlate with induction of angiogenic sprouting. Am J Pathol. 2002;161:823–30.
Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8:e1000412.
Hainsworth AH, Bermpohl D, Webb TE, et al. Expression of cellular FLICE inhibitory proteins (cFLIP) in normal and traumatic murine and human cerebral cortex. J Cereb Blood Flow Metab. 2005;25:1030–40.
Yanez-Munoz RJ, Balaggan KS, MacNeil A, et al. Effective gene therapy with nonintegrating lentiviral vectors. Nat Med. 2006;12:348–53.
Follenzi A, Ailles LE, Bakovic S, Geuna M, Naldini L. Gene transfer by lentiviral vectors is limited by nuclear translocation and rescued by HIV-1 pol sequences. Nat Genet. 2000;25:217–22.
Wanisch K, Yanez-Munoz RJ. Integration-deficient lentiviral vectors: a slow coming of age. Mol Ther. 2009;17:1316–32.
Roger VL, Go AS, Lloyd-Jones DM, et al. Heart disease and stroke statistics—2011 update: a report from the American Heart Association. Circulation. 2011;123:e18–e209.
Ederle J, Dobson J, Featherstone RL, et al. Carotid artery stenting compared with endarterectomy in patients with symptomatic carotid stenosis (International Carotid Stenting Study): an interim analysis of a randomised controlled trial. Lancet. 2010;375:985–97.
van Lammeren GW, Peeters W, de Vries JP, et al. Restenosis after carotid surgery: the importance of clinical presentation and preoperative timing. Stroke. 2011;42:965–71.
Hooper WC, Catravas JD, Heistad DD, Sessa WC, Mensah GA. Vascular endothelium summary statement I: health promotion and chronic disease prevention. Vascul Pharmacol. 2007;46:315–7.
Marshall Jr WG, Boone BA, Burgos JD, et al. Electroporation-mediated delivery of a naked DNA plasmid expressing VEGF to the porcine heart enhances protein expression. Gene Ther. 2010;17:419–23.
Raake PW, Hinkel R, Muller S, et al. Cardio-specific long-term gene expression in a porcine model after selective pressure-regulated retroinfusion of adeno-associated viral (AAV) vectors. Gene Ther. 2008;15:12–7.
Bandopadhyay R, Orte C, Lawrenson JG, Reid AR, De SS, Allt G. Contractile proteins in pericytes at the blood–brain and blood–retinal barriers. J Neurocytol. 2001;30:35–44.
Rowinsky EK, Jiroutek M, Bonomi P, Johnson D, Baker SD. Paclitaxel steady-state plasma concentration as a determinant of disease outcome and toxicity in lung cancer patients treated with paclitaxel and cisplatin. Clin Cancer Res. 1999;5:767–74.
Perree J, van Leeuwen TG, Kerindongo R, Spaan JA, Vanbavel E. Function and structure of pressurized and perfused porcine carotid arteries: effects of in vitro balloon angioplasty. Am J Pathol. 2003;163:1743–50.
Gambillara V, Chambaz C, Montorzi G, Roy S, Stergiopulos N, Silacci P. Plaque-prone hemodynamics impair endothelial function in pig carotid arteries. Am J Physiol Heart Circ Physiol. 2006;290:H2320–8.
Muller-Hulsbeck S, Stolzmann P, Liess C, et al. Vessel wall damage caused by cerebral protection devices: ex vivo evaluation in porcine carotid arteries. Radiology. 2005;235:454–60.
Stiffey-Wilusz J, Boice JA, Ronan J, Fletcher AM, Anderson MS. An ex vivo angiogenesis assay utilizing commercial porcine carotid artery: modification of the rat aortic ring assay. Angiogenesis. 2001;4:3–9.
Acknowledgments
We are grateful to Mr RW Chang FRCS, Mr M Morsy FRCS and Professor G StJ Whitley for valuable advice and access to resources, and to the staff of the Biological Research Facility of St George’s for their expert assistance. We thank Dr AG Clifton FRCRadiol for the helpful comments throughout the project.
Funding
AHH gratefully acknowledges funding from The National Centre for the Replacement, Refinement and Reduction of Animals in Research (www.nc3rs.org.uk), the St George’s Hospital Charity and The Neuroscience Research Foundation. RJYM and AHH acknowledge funding from the South West London Academic Network.
Conflict of Interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andoh, J., Sawyer, B., Szewczyk, K. et al. Transgene Delivery to Endothelial Cultures Derived from Porcine Carotid Artery Ex Vivo. Transl. Stroke Res. 4, 507–514 (2013). https://doi.org/10.1007/s12975-013-0261-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-013-0261-6