Abstract
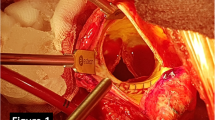
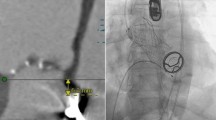
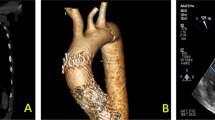
Redo surgery is regarded as the first-line treatment option for patients presenting with prosthetic valve degeneration. However, many patients have concomitant co-morbidities and this option is associated with significant risk. Transcatheter valve-in-valve implantation is an alternative strategy depending on the bioprosthetic valve that is being treated. The Sorin Mitroflow bioprosthetic aortic valve has been regarded as a contraindication to valve-in-valve treatment due to the high risk of coronary obstruction. We here present the case of a patient with small peripheral vasculature who underwent successful transfemoral valve-in-valve implantation and subsequently discuss the challenges and technical aspects that require consideration.



Similar content being viewed by others
References
Brown JM, O’Brien SM, Wu C, Sikora JA, Griffith BP, Gammie JS. Isolated aortic valve replacement in North America comprising 108,687 patients in 10 years: changes in risks, valve types, and outcomes in the Society of Thoracic Surgeons National Database. J Thorac Cardiovasc Surg. 2009;137(1):82–90.
Vesely I. The evolution of bioprosthetic heart valve design and its impact on durability. Cardiovasc Pathol. 2003;12(5):277–86.
Maganti M, Rao V, Armstrong S, Feindel CM, Scully HE, David TE. Redo valvular surgery in elderly patients. Ann Thorac Surg. 2009;87(2):521–5.
Balsam LB, Grossi EA, Greenhouse DG, Ursomanno P, Deanda A, Ribakove GH, Culliford AT, Galloway AC. Reoperative valve surgery in the elderly: predictors of risk and long-term survival. Ann Thorac Surg. 2010;90(4):1195–2000 (discussion 1201).
Hamm CW, Mollmann H, Holzhey D, Beckmann A, Veit C, Figulla HR, Cremer J, Kuck KH, Lange R, Zahn R, et al. The German Aortic Valve Registry (GARY): in-hospital outcome. Eur Heart J. 2014;35(24):1588–98.
Eggebrecht H, Schafer U, Treede H, Boekstegers P, Babin-Ebell J, Ferrari M, Mollmann H, Baumgartner H, Carrel T, Kahlert P, et al. Valve-in-valve transcatheter aortic valve implantation for degenerated bioprosthetic heart valves. JACC Cardiovasc Interv. 2011;4(11):1218–27.
Dvir D, Webb J, Brecker S, Bleiziffer S, HildickSmith D, Colombo A, Descoutures F, Hengstenberg C, Moat NE, Bekeredjian R, et al. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in-valve registry. Circulation. 2012;126(19):2335–44.
Dvir D, Leipsic J, Blanke P, Ribeiro HB, Kornowski R, Pichard A, Rodes-Cabau J, Wood DA, Stub D, Ben-Dor I, et al. Coronary obstruction in transcatheter aortic valve-in-valve implantation: preprocedural evaluation, device selection, protection, and treatment. Circ Cardiovasc Interv. 2015;8(1):e002079.
Gurvitch R, Cheung A, Bedogni F, Webb JG. Coronary obstruction following transcatheter aortic valve-in-valve implantation for failed surgical bioprostheses. Catheter Cardiovasc Interv. 2011;77(3):439–44.
Bapat V, Mydin I, Chadalavada S, Tehrani H, Attia R, Thomas M. A guide to fluoroscopic identification and design of bioprosthetic valves: a reference for valve-in-valve procedure. Catheter Cardiovasc Interv. 2013;81(5):853–61.
Linke A, Woitek F, Merx MW, Schiefer C, Mobius-Winkler S, Holzhey D, Rastan A, Ender J, Walther T, Kelm M, et al. Valve-in-valve implantation of Medtronic CoreValve prosthesis in patients with failing bioprosthetic aortic valves. Circ Cardiovasc Interv. 2012;5(5):689–97.
Yankah CA, Pasic M, Musci M, Stein J, Detschades C, Siniawski H, Hetzer R. Aortic valve replacement with the Mitroflow pericardial bioprosthesis: durability results up to 21 years. J Thorac Cardiovasc Surg. 2008;136(3):688–96.
Ribeiro HB, Webb JG, Makkar RR, Cohen MG, Kapadia SR, Kodali S, Tamburino C, Barbanti M, Chakravarty T, Jilaihawi H, et al. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. J Am Coll Cardiol. 2013;62(17):1552–62.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Dr. Azeem Latib is a consultant for Medtronic and Direct Flow Medical. Dr. Antonio Colombo is a Minor Shareholder of Direct Flow. Inc.
Human and animal informed consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Full informed consent was obtained from the patient.
Rights and permissions
About this article
Cite this article
Ruparelia, N., Colombo, A. & Latib, A. Valve-in-valve transcatheter aortic valve implantation overcoming hostile anatomy: Evolut R for the treatment of Mitroflow bioprosthesis dysfunction. Cardiovasc Interv and Ther 31, 292–295 (2016). https://doi.org/10.1007/s12928-015-0354-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12928-015-0354-0