Abstract
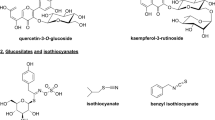
Weedy rice is a semi-wild species that has survived for a very long time in rice fields, so its seeds could contain more functional substances than cultivated rice. Additionally, during the process of seed germination, some physiologically active substances that were not present in the seed are newly synthesized, so the functional compounds of germinated seeds could be improved over those of untreated raw seeds. From this perspective, we evaluated metabolite compounds of the brown rice (BR) and germinated brown rice (GBR) of WD3 compared with a cultivated rice, Sindongjin (SDJ). WD3 is a weedy rice selected for antioxidant activity from 199 accessions of Korea-native weedy rice germplasm (Cho et al., J Crop Sci 63:219–228, 2018). Metabolites were measured by using UPLC coupled with tandem mass spectrometry. Pareto scaling method prior to statistical and chemometric analysis were used for data acquisition and processing for metabolize compounds. OPLS-DA was conducted to develop discriminatory models. To support the potential biomarkers identification and further understanding of this study, the information on compounds was queried on the Chemspider web page. The obvious separation of metabolites between WD3 and SDJ in both BR and GBR were observed, in which a total of 103 compounds were detected. Among them, 52 compounds were known and 51 compounds were unknown. In the known compounds, long aliphatic chains were commonly found and the others were steroids, alkaloids, polyols, peptide and aromatic acid. Some metabolites such as 3 alkaloid compounds, 2 steroids, 1 aromatic acid and 15 long aliphatic chains were significantly increased after germination in WD3 seeds. Among them, the content of C45H84NO7P was increased by more than 1,000,000 times after germination. These compounds found in the GBR of WD3 might also be correlated to the nutritionally qualities claimed to be responsible for the advantageous properties of GBR.







Similar content being viewed by others
Data availability
Readers are encouraged to contact the authors if they require data.
References
Ahsan H, Reagan-Shaw S, Eggert DM, Tan TC, Afaq F, Mukhtar H, Ahmad N (2007) Protective effect of sanguinarine on ultraviolet B-mediated damages in SKH-1 hairless mouse skin: implications for prevention of skin cancer. Photochem Photobiol 83:986–993
Autio K (1996) Functional aspects of cereal cell wall polysaccharides, In Carbohydrate in Food. A. C. Eliasson (ed), 227–266
Banthorpe DV (1994) Natural Products: their chemistry and biological significance, 1st Ed., Longman scientific and technical, Essex. 289
Bouic PJD (2001) The role of phytosterols and phytosterolins in immune modulation: a review of the past 10 years. Curr Opin Clin Nutr Metab Care 4:471–475
Cavazos A, de Mejia EG (2013) Identification of bioactive peptides from cereal storage proteins and their potential role in prevention of chronic diseases. Compr Rev Food Sci Saf 12(4):364–380
Cho EE, Chung NJ (2020) Enhancement of amino acid contents by germination in a Korean weedy rice germplasm. J Crop Sci Biotechnol 23(3):375–384
Cho EE, Baek JS, Chung NJ (2018) Antioxidant activities in germinated and non-germinated seeds of Korean weedy rice. J Crop Sci 63(3):219–228
Chung JW, Park YJ (2010) Population structure analysis reveals the maintenance of isolated sub-populations of weedy rice. Weed Res 50:606–620
Chung HS, Woo WS (2001) A quinolone alkaloid with antioxidant activity from the aleurone layer of anthocyanin-pigmented rice. J Nat Prod 64:1579–1580
Delvin MT (2002) Text Book of Biochemistry with Clinical Correlations, 5th Ed
Ekblad E, Sundler F (2002) Distribution of pancreatic polypeptide and peptide YY. Peptides 23(2):251–261
Fernie AR, Tadmore Y, Zamir D (2006) Natural genetic diversity for improving crop quality. Curr Opin Plant Biol 9:196–202
Ferri M, Graen-Heedfeld J, Bretz K, Guillon F, Michelini E, Calabretta MM, Lamborghini M, Gruarin N, Roda A, Kraft A (2017) Peptide fractions obtained from rice by-products by means of an environment-friendly process show in vitro health-related bioactivities. PLoS ONE 12(1):e0170954
Friedman M (2013) Rice brans, rice bran oils, and rice hulls: Composition, food and industrial uses, and bioactivies in humans, animals, and cells. J Agric Food Chem 61:10626–10641
Hara S (1942) Persistence of an Indica rice in Korea. Agriculture and Horticulture J 17:705–712
Heu MH, Cho YC, Suh HS (1990) Cross affinity of Korean weedy rice to the cultivars. Kor J Crop Sci 35:233–238
Jala RCR, Prasad RBN (2015) Rice bran lecithin: compositional, nutritional, and functional characteristics. Polar Lipids. Biology, Chemistry and Technology. 35–55
Jung HY, Lee DH, Baek HY, Lee YS (2008) Pre- and post-germination changes in pharmaceutical compounds of germinated brown rice. Korean J Crop Sci 53(5):37–43
Kankaanpaa P, Sutas Y, Salminen S, Lichtenstein A, Isolauri E (1999) Dietary fatty acids and allergy. Ann Med 31:282–287
Kaur R, Arora S (2015) Alkaloids important therapeutic secondary metabolites of plant origin. J Crit Rev 2(3):1–8
Keicho N, Kudoh S (2002) Diffuse panbronchiolitis: role of macrolides in therapy. Am J Respir Med 1(2):119–131
Kim GR, Jung ES, Lee S, Lim SH, Ha SH, Lee CH (2014) Combined mass spectrometry-based metabolite profiling of different pigmented rice (Oryza sativa L.) seeds and correlation with antioxidant activities. Molecules 19:15673–15686
Lim DK, Mo C, Long NP, Kim G, Kwon SW (2017) Simultaneous profiling of lysoglycerophospholipids in rice (Oryza sativa L.) using direct infusion-tandem mass spectrometry with multiple reaction monitoring. J Agric Food Chem 65:2628–2634
Long NP, Lim DK, Mo C, Kim G, Kwon SW (2017) Development and assessment of a lysophospholipid-based deep learning model to discriminate geographical origins of white rice. Sci Rep 7:1
López-Boado YS, Rubin BK (2008) Macrolides as immunomodulatory medications for the therapy of chronic lung diseases. Curr Opin Pharmacol 8(3):286–291
Lou Q, Ma C, Wen W, Zhou J, Chen L, Feng F, Xu X, Luo L, Mei H, Xu G (2011) Profiling and association mappimg of grain metabolites in a subsel of the core collection of Chinese rice germplasm (Oryza sativa L.). J Agric Food Chem 39:9257–9264
Machowinski A, Krämer H, Hort W, Mayser P (2006) Pityriacitrin – a potent UV filter produced by Malassezia furfur and its effect on human skin microflora. Mycoses 49:388–392
Maestri E, Marmiroli M, Marmiroli N (2016) Bioactive peptides in plant-derived food stuffs. J Proteomics 1:1
Matsuura HN, Fett-Neto AG (2013) The major indole alkaloid N, b-D-glucopyranosyl vincosamide from leaves of Psychotria leiocarpa Cham. & Schltdl. is not an antifeedant but shows broad antioxidant activity. Nat Prod Res 27:402–411
Matsuura HN, Rau MR, Fett-Neto AG (2014) Oxidative stress and production of bioactive monoterpene indole alkaloids: biotechnological implications. Biotechnol Lett 36:191–200
Moreau RA, Whitaker BD, Hicks KB (2002) Phytosterols, phytostanols, and their conjugates in foods: Structural diversity, quantitative analysis, and health-promoting uses. Prog Lipid Res 41:457–500
Nadir S, Xiong HB, Zhu Q, Zhang XL, Xu HY, Li J, Dongchen W, Henry D, Guo XQ, Khan S, Suh HS, Lee DS, Chen LJ (2017) Weedy rice in sustainable rice production. A review. Agron Sustain Dev 2017:37–46
Nakagawa K, Onota A (1996) Accumulation of γ- aminobutyric acid(GABA) in the rice germ. Shokuhin and Kaihatsu 31:43–46
Oikawa A, Matsuda F, Kusano M, Okazaki Y, Saito K (2008) Rice Metab Rice 1(1):63–71
Patil SB, Khan MK (2011) Germinated brown rice as a value added rice product: a review. J Food Sci Technol 48(6):661–667
Petković M (2019) Alternatives for sugar replacement in food technology: Formulating and processing key aspects. Food Eng 1:822–851
Porto DD, Matsuura HN, Vargas LRB, Henriques AT, Fett-Neto AG (2014) Shoot accumulation kinetics and effects on herbivores of the wound-induced antioxidant indole alkaloid brachycerine of Psychotria brachyceras. Nat Prod Commun 9:629–632
Quı´lez J, Garcı´a-Lorda P, Salas-Salvado´ J. (2003) Potential uses and benefits of phytosterols in diet: present situation and future directions. Clin Nutr 22:343–351
Rincón-León F (2003) Functional foods in Encyclopedia of Food Sciences and Nutrition (2th Edition)
Roudier F, Gissot L, Beaudoin F, Haslam R, Michaelson L, Marion J, Molino D, Lima A, Bach L, Morin H (2010) Very-long-chain fatty acids are involved in polar auxin transport and developmental patterning in Arabidopsis. Plant Cell 22:364–375
Schaller H (2004) New aspects of sterol biosynthesis in growth and development of higher plants. Plant Physiol Biochem 42:465–476
Simopoulos AP (1991) Omega-3 fatty acids in health and disease and in growth and development. Am J Clin Nutr 54:438–463
Suh HS, Park SZ, Heu MH (1992) Collection and evaluation of Korean red rices I. Regional distribution and seed characteristics. Korean J Crop Sci 37:425–430
Vieira AT, Pinho V, Lepsch LB, Scavone C, Ribeiro IM (2005) Mechanisms of the anti-inflammatory effects of the natural secosteroids physalins in a model of intestinal ischaemia and reperfusion injury. Br J Pharmacol 146:244–251
Vriet C, Russinova E, Reuzeaua C (2012) Boosting crop yields with plant steroids. Plant Cell 24(3):842–857
Wold S, Sjostrom M, Eriksson L (2001) PLS-regression: a basic tool of chemometrics. Chemom Intell Lab Syst 58:109–130
Wu F, Yang N, Toure A, Jin Z, Xu X (2013) Germinated brown rice and its role in human health. Crit Rev Food Sci Nutr 53:451–463
Yang L, Stöckigt J (2010) Trends for diverse production strategies of plant medicinal alkaloids. Nat Prod Rep 27:1469–1479
Zhang S, Tian L, Li J, Wang C, Lee D, Peng R, Chen L (2017) Morphological characterization of weedy rice populations from different regions of Asia. Mol Plant Breed 8(6):52–64
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
To the best of our knowledge, the named authors have no conflict of interest, financial or otherwise. The authors whose names are listed in this manuscript certify that they have no affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cho, E.E., Chung, NJ. Comparison of metabolites between brown rice and germinated brown rice in a Korean weedy rice germplasm. J. Crop Sci. Biotechnol. 27, 235–247 (2024). https://doi.org/10.1007/s12892-023-00225-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-023-00225-0