Abstract
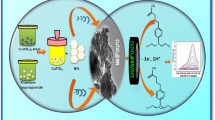
Indinavir (IDV) is a potent and well-tolerated protease inhibitor antiretroviral (ARV) drug used as a component of the highly active antiretroviral therapy (HAART) of human immunodeficiency virus (HIV). It undergoes hepatic first-pass metabolism that is catalysed by microsomal cytochrome P450-3A4 enzyme (CYP3A4), which results in pharmacokinetics that may be favourable or adverse. Therapeutic drug monitoring (TDM) of IDV during HIV treatment is therefore critical, in order to prevent the adverse effects of its first-pass metabolism and optimise an individual’s dosage regime. Biosensors are now the preferred diagnostic tools for TDM assessment at point-of-care, due to their high sensitivity and real-time response. An electrochemical biosensor for IDV was prepared by depositing a thin film of CYP3A4 (a thiolate enzyme) and thioglycolic acid-capped palladium telluride quantum dot (TGA-PdTeQD) on a cysteamine-functionalised gold disk electrode (Cyst|Au) using a combination of thiol and carbodiimide covalent bonding chemistries. The electrochemical signatures of the biosensor (CYP3A4|TGA-PdTeQD|Cyst|Au) were determined by cyclic voltammetry (CV) that was performed at a scan rate of 500 mV s−1, and the sensor responses at the characteristic reduction peak potential value of − 0.26 V were recorded. The sensitivity, linear range (LR) and limit of detection (LOD) values of the indinavir biosensor were 4.45 ± 0.11 μA nM−1 IDV, 0.5–1.0 nM IDV (i.e. 3.6 × 10−4–7.1 × 10−4 mg L−1 IDV) and 4.5 × 10−4 mg L−1 IDV, respectively. The values of the two analytical parameters (LR and LOD) of the biosensor were by up to four orders of magnitude lower than the maximum plasma concentration (Cmax) values of indinavir (0.13–8.6 mg L−1 IDV). The IDV biosensor was successfully used to detect IDV in human serum samples containing dissolved indinavir tablet. This, therefore, indicates the indinavir biosensor’s suitability for TDM applications, using samples obtained within 1–2 h of drug intake at point-of-care, for which very low levels of the drug are expected.

Graphical abstract






Similar content being viewed by others
References
V.V. Shumyantseva, A.V. Kuzikov, R.A. Masamrekh, T.V. Bulko, A.I. Archakov, Biosens Bioelectron 121, 192 (2018)
S.C. Preissner, M.F. Hoffmann, R. Preissner, M. Dunkel, A. Gewiess, S. Preissner, PLoS One 8, e82562 (2013)
S.C. Gay, A.G. Roberts, J.R. Halpert, Future Med Chem 2, 1451 (2010)
J. Ning, Z. Tian, B. Wang, G. Ge, Y. An, J. Hou, C. Wang, X. Zhao, Y. Li, X. Tian, Z. Yu, X. Huo, C. Sun, L. Feng, J. Cui, X. Ma, Mater Chem Front 2, 2013 (2018)
I.G. Denisov, B.J. Baas, Y.V. Grinkova, S.G. Sligar, J Biol Chem 282, 7066 (2007)
F.J. Gonzalez, Drug Metab Rev 35, 319 (2003)
F.P. Guengerich, Mol Interv 3, 194 (2003)
F.P. Guengerich, Chem Res Toxicol 21, 70 (2008)
U.M. Zanger, M.H. Hofmann, Acta Chim Slov 55, 38 (2008)
U.M. Zanger, M. Schwab, Pharmacol Ther 138, 103 (2013)
S. Marchetti, R. Mazzanti, J.H. Beijnen, J.H.M. Schellens, Oncologist 12, 927 (2007)
J.A. Grabowsky, Ann Pharmacother 47, 1055 (2013)
N. Wang, C. Gao, F. Xue, Y. Han, T. Li, X. Cao, X. Zhang, Y. Zhang, Z.L. Wang, ACS Nano 9, 3159 (2015)
P. Nowak, M. Woźniakiewicz, P. Kościelniak, TrAC - Trends Anal Chem 59, 42 (2014)
A.K. Udit, M.G. Hill, H.B. Gray, Langmuir 22, 10854 (2006)
V.E.V. Ferrero, L. Andolfi, G. Di Nardo, S.J. Sadeghi, A. Fantuzzi, S. Cannistraro, G. Gilardi, Anal Chem 80, 8438 (2008)
H. Huang, J.-J. Zhu, Analyst 138, 5855 (2013)
E. Nxusani, P.M. Ndangili, R.A. Olowu, A.N. Jijana, T. Waryo, N. Jahed, R.F. Ajayi, P. Baker, E.I. Iwuoha, Nano Hybrids 1, 1 (2012)
A. Ignaszak, N. Hendricks, T. Waryo, E. Songa, N. Jahed, R. Ngece, A. Al-Ahmed, B. Kgarebe, P. Baker, E.I. Iwuoha, J Pharm Biomed Anal 49, 498 (2009)
U. Feleni, U. Sidwaba, H. Makelane, E. Iwuoha, J Nanosci Nanotechnol 19, 1 (2019)
H. Yu, Z. Song, Q. Liu, X. Ji, J. Liu, S. Xu, H. Kan, B. Zhang, J. Liu, J. Jiang, L. Miao, H. Liu, Sensors Actuators. B Chem 248, 1029 (2017)
S. Shahrokhian, A. Mahdavi-Shakib, M. Ghalkhani, R.S. Saberi, Electroanalysis 24, 425 (2012)
N.u.H. Alvi, V.J. Gómez, P.E.D.S. Rodriguez, P. Kumar, S. Zaman, M. Willander, R. Nötzel, Sensors (Basel) 13, 13917 (2013)
T. Teranishi, M. Miyake, Chem Mater 10, 594 (1998)
J.D. Olson, G.P. Gray, S.A. Carter, Sol Energy Mater Sol Cells 93, 519 (2009)
B. Sun, N.C. Greenham, Phys Chem Chem Phys 8, 3557 (2006)
B. Sun, H.J. Snaith, A.S. Dhoot, S. Westenhoff, N.C. Greenham, J Appl Phys 97 (2005)
R. Molaie, K. Farhadi, M. Forough, S. Haijizadeh, J Nanostruct 8, 1 (2018)
M. Rafi Shaik, Z.J.Q. Ali, M. Khan, M. Kuniyil, M.E. Assal, H.Z. Alkhathlan, A. Al-Warthan, M.R.H. Siddiqui, M. Khan, S.F. Adil, Molecules 22 (2017)
P.A. Namini, A.A. Babaluo, B. Bayati, Int J Nanosci Nanotechnol 3, 37 (2007)
M. Masikini, P.M. Ndangili, C.O. Ikpo, U. Feleni, S. Duoman, U. Sidwaba, T.T. Waryo, P.G.L. Baker, E.I. Iwuoha, J Nanopart Res 40, 29 (2016)
S.A. Ivanov, A. Piryatinski, J. Nanda, S. Tretiak, K.R. Zavadil, W.O. Wallace, D. Werder, V.I. Klimov, J Am Chem Soc 129, 11708 (2007)
A. Arshad, R. Akram, S. Iqbal, F. Batool, B. Iqbal, B. Khalid, A.U. Khan, Arab J Chem (2016)
J.K. Cooper, A.M. Franco, S. Gul, C. Corrado, J.Z. Zhang, Langmuir 27, 8486 (2011)
A.E. Vikraman, A.R. Jose, M. Jacob, K.G. Kumar, Anal Methods 7, 16 (2015)
S.S. Rokade, K.A. Joshi, K. Mahajan, G. Tomar, D.S. Dubal, V. Singh, R. Kitture, J. Bellare, S. Ghosh, Glob J Nanomed 2, 5 (2017)
S.H. Shin, J. Bajaj, L.A. Moudy, D.T. Cheung, Appl Phys Lett 43, 68 (1983)
S.J. Byrne, S.A. Corr, T.Y. Rakovich, Y.K. Gun’ko, Y.P. Rakovich, J.F. Donegan, S. Mitchell, Y. Volkov, J Mater Chem 16 (2006)
H. Borchert, D.V. Talapin, N. Gaponik, C. McGinley, S. Adam, A. Lobo, T. Möller, H. Weller, J Phys Chem B 107, 9662 (2003)
S.M. Baesman, T.D. Bullen, J. Dewald, D. Zhang, S. Curran, F.S. Islam, T.J. Beveridge, R.S. Oremland, Appl EnvironMicrobiol 73, 2135 (2007)
A.G. Milekhin, L.L. Sveshnikova, T.A. Duda, N.V. Surovtsev, S.V. Adichtchev, Y.M. Azhniuk, C. Himcinschi, M. Kehr, D.R.T. Zahn, J Phys Conf Ser 245, 012045 (2010)
J. Heo, C.-S. Hwang, Nanomaterials 5, 1955 (2015)
J.S.W. Mak, A.A. Farah, F. Chen, A.S. Helmy, ACS Nano 5, 3823 (2011)
B.E. Conway, Prog Surf Sci 49, 331 (1995)
K. De Wael, A. Verstraete, S. Van Vlierberghe, W. Dejonghe, P. Dubruel, A. Adriaens, Int J Electrochem Sci 6, 1810 (2011)
A. Ulman, Chem Rev 96, 1533 (1996)
B.R. Kozub, N.V. Rees, R.G. Compton, Sensors Actuators. B Chem 143, 539 (2010)
T. Łuczak, Int J Electrochem 2011, 1 (2011)
Q. Cheng, A. Brajter-Toth, Anal Chem 64, 1998 (1992)
L.H. Wang, W.S. Huang, Sensors 12, 3562 (2012)
A.I. Gopalan, K.P. Lee, K.M. Manesh, P. Santhosh, J.H. Kim, J Mol Catal A Chem 256, 335 (2006)
A. Manickam, C.A. Johnson, S. Kavusi, A. Hassibi, Sensors (Switzerland) 12, 14467 (2012)
M. Grdeń, M. Łukaszewski, G. Jerkiewicz, A. Czerwiński, Electrochim Acta 53, 7583 (2008)
K. Yoshii, Y. Oshino, N. Tachikawa, K. Toshima, Y. Katayama, Electrochem Commun 52, 21 (2015)
C.S. Lim, K. Hola, A. Ambrosi, R. Zboril, M. Pumera, Electrochem Commun 52, 75 (2015)
M. Yang, J.L. Kabulski, L. Wollenberg, X. Chen, M. Subramanian, T.S. Tracy, D. Lederman, P.M. Gannett, N. Wu, Drug Metab Dispos 37, 892 (2009)
N.R. Hendricks, T.T. Waryo, O. Arotiba, N. Jahed, P.G.L. Baker, E.I. Iwuoha, Electrochim Acta 54, 1925 (2009)
B. Dogan, D. Canbaz, S.A. Ozkan, B. Uslu, Pharmazie 61, 409 (2006)
A.C. Sather, H.G. Lee, V.Y. De La Rosa, Y. Yang, P. Müller, S.L. Buchwald, J Am Chem Soc 137, 13433 (2015)
Y. He, X. Yang, Q. Han, J. Zheng, Molecules 22, 1 (2017)
R. Chokkareddy, N.K. Bhajanthri, G.G. Redhi, Biosensors 7, 21 (2017)
C.A. Hasemann, R.G. Kurumbail, S.S. Boddupalli, J.A. Peterson, J. Deisenhofer, Structure 3, 41 (1995)
S.C. Piscitelli, A.H. Burstein, D. Chaitt, R.M. Alfaro, J. Falloon, Lancet 355, 547 (2000)
N. Erk, Pharmazie 59, 183 (2004)
K. Rajitha, N.L. Prasanna, R. Naveen, C.H. Ranjth, A.A. Kumar, Int J Pharm Pharm Sci 6, 8 (2014)
E. Marchei, R. Pacifici, G. Tossini, R. Di Fava, L. Valvo, P. Zuccaro, J Liq Chromatogr Relat Technol 24, 2325 (2001)
J.M. Poirier, P. Robidou, P. Jaillon, Ther Drug Monit 22, 4 (2000)
A.L. Jayewardene, B. Kearney, J.A. Stone, J.G. Gambertoglio, F.T. Aweeka, J Pharm Biomed Anal 25, 309 (2001)
W.K. Kraft, J.B. McCrea, G.A. Winchell, A. Carides, R. Lowry, E.J. Woolf, S.E. Kusma, P.J. Deutsch, H.E. Greenberg, S.A. Waldman, J Clin Pharmacol 44, 305 (2004)
R. Hajian, Z. Tayebi, N. Shams, J Pharm Anal 7, 27 (2017)
T.D. Gibson, ANALUSIS 27, 630 (1999)
C. Sarika, K. Rekha, B. Narasimha Murthy, 3 Biotech 5, 911 (2015)
A. Chaubey, B.D. Malhotra, Biosens Bioelectron 17, 441 (2002)
F. Ricci, A. Amine, G. Palleschi, D. Moscone, Biosens Bioelectron 18, 165 (2003)
D. Ravi Shankaran, N. Uehara, T. Kato, Biosens Bioelectron 18, 721 (2003)
Funding
This work was funded by the South African Department of Science and Technology’s (DST’s) National Nanoscience Postgraduate Teaching and Training Platform (NNPTTP), and the National Research Foundation (NRF) of South Africa Research Chair Initiative Grant Number 85102, for NanoElectrochemistry and Sensor Technology.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Feleni, U., Sidwaba, U., Ntshongontshi, N. et al. Biocompatible Palladium Telluride Quantum Dot-Amplified Biosensor for HIV Drug. Electrocatalysis 11, 68–76 (2020). https://doi.org/10.1007/s12678-019-00563-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-019-00563-0