Abstract
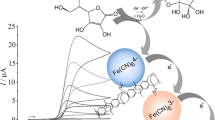
The free radical 4-benzoyloxy-2,2,6,6-tetramethylpiperidine-1-oxyl (4B-TEMPO) is active as an electrocatalyst for primary alcohol oxidations when immobilised at an electrode surface and immersed into an aqueous carbonate buffer solution. In order to improve the catalytic process, a composite film electrode is developed based on (i) carbon microparticles of 2–12 μm diameter to enhance charge transport and (ii) a polymer of intrinsic microporosity (here PIM-EA-TB with a BET surface area of 1027 m2 g−1). The latter acts as a highly rigid molecular framework for the embedded free radical catalyst with simultaneous access to aqueous phase and substrate. The resulting mechanism for the oxidation of primary alcohols is shown to switch in reaction order from first to zeroth with increasing substrate concentration consistent with a kinetically limited process with competing diffusion of charge at the polymer layer-electrode interface (here the “LEk” case in Albery-Hillman notation). Reactivity optimisation and screening for a wider range of primary alcohols in conjunction with DFT-based relative reactivity correlation reveals substrate hydrophobicity as an important factor for enhancing catalytic currents. The PIM-EA-TB host matrix is proposed to control substrate partitioning and thereby catalyst reactivity and selectivity.

The water-insoluble molecular alcohol oxidation catalyst 4-benzoyloxy-TEMPO is employed here embedded in a nano-composite film based on a hydrophobic polymer of intrinsic microporosity (PIM)








Similar content being viewed by others
References
R.A. Sheldon, Catal. Today 247, 4 (2015)
N.B. McKeown, P.M. Budd, Chem. Soc. Rev. 35, 675 (2006)
W.J. Albery, A.R. Hillman, J. Electroanal. Chem. 170, 27 (1984)
J.M. Saveant, Chem. Rev. 108, 2348 (2008)
R. Ciriminna, M. Pagliaro, Org. Process. Res. Dev. 14, 245 (2010)
T. Vogler, A. Studer, Synthesis-Stuttgart 13, 1979 (2008)
K.U. Schoening, Chim. Oggi-Chem. Today 28, 18 (2010)
P. Gallezot, Chem. Soc. Rev. 41, 1538 (2012)
S. Eyley, W. Thielemans, Nanoscale 6, 7764 (2014)
R.B. Grubbs, Polym. Rev. 51, 104 (2011)
S. Park, H. Boo, T.D. Chung, Anal. Chim. Acta 556, 46 (2006)
D.P. Hickey, M.S. McCammant, F. Giroud, M.S. Sigman, S.D. Minteer, J. Amer. Chem. Soc. 136, 15917 (2014)
M. Gilhespy, M. Lok, X. Baucherel, Catal. Today 117, 114 (2006)
R.A. Sheldon, I.W.C.E. Arends, G.T. Brink, A. Dijksman, Acc. Chem. Res. 25, 774 (2002)
J.T. Hill-Cousins, J.K. Kuleshova, R.A. Green, P.R. Birkin, D. Pletcher, T.J. Underwood, S.G. Leach, R.C.D. Brown, ChemSusChem 5, 326 (2012)
Y. Jin, K.J. Edler, F. Marken, J.L. Scott, Green Chem. 16, 3322 (2014)
W.F. Bailey, J.M. Bobbitt, K.B. Wiberg, J. Org. Chem. 72, 4504 (2007)
R.A. Green, J.T. Hill-Cousins, R.C.D. Brown, D. Pletcher, S.G. Leach, Electrochim. Acta 113, 550 (2013)
J.L. Wang, M. Zhang, Z. Zheng, F.W. Yu, J.B. Ji, Chem. Eng. J. 229, 234 (2013)
D.H. Hunter, D.H.R. Barton, W.B. Motherwell, Tetrahedron Lett. 25, 603 (1984)
A. Israeli, M. Patt, M. Oron, A. Samuni, R. Kohen, S. Goldstein, Free Radic. Biol. Med. 38, 317 (2005)
R. Ciriminna, G. Palmisano, M. Pagliaro, ChemCatChem 7, 552 (2015)
P.M. Budd, E.S. Elabas, B.S. Ghanem, S. Makhseed, N.B. McKeown, K.J. Msayib, C.E. Tattershall, D. Wang, Adv. Mater. 16, 456 (2004)
P.M. Budd, B.S. Ghanem, S. Makhseed, N.B. McKeown, K.J. Msayib, C.E. Tattershall, Chem. Commun. 10, 230 (2004)
M. Carta, R. Malpass-Evans, M. Croad, Y. Rogan, J.C. Jansen, P. Bernardo, F. Bazzarelli, N.B. McKeown, Science 339, 303 (2013)
M. Carta, M. Croad, J.C. Jansen, P. Bernardo, G. Clarizia, N.B. McKeown, Polym. Chem. 5, 5255 (2014)
M. Carta, M. Croad, R. Malpass-Evans, J.C. Jansen, P. Bernardo, G. Clarizia, K. Friess, M. Lanc, N.B. McKeown, Adv. Mater. 26, 3526 (2014)
M. Carta, R. Malpass-Evans, M. Croad, Y. Rogan, M. Lee, I. Rose, N.B. McKeown, Polym. Chem. 5, 5267 (2014)
F.J. Xia, M. Pan, S.C. Mu, R. Malpass-Evans, M. Carta, N.B. McKeown, G.A. Attard, A. Brew, D.J. Morgan, F. Marken, Electrochim. Acta 128, 3 (2014)
Y.Y. Rong, R. Malpass-Evans, M. Carta, N.B. McKeown, G.A. Attard, F. Marken, Electrochem. Commun. 46, 26 (2014)
Y.Y. Rong, R. Malpass-Evans, M. Carta, N.B. McKeown, G.A. Attard, F. Marken, Electroanalysis 26, 904 (2014)
D. He, Y.Y. Rong, K.Z. Kui, S.C. Mu, T. Peng, R. Malpass-Evans, M. Carta, N.B. Mckeown, F. Marken, Electrochem. Commun. 59, 72 (2015)
A. Kolodziej, S.D. Ahn, M. Carta, R. Malpass-Evans, N.B. McKeown, R.S.L. Chapman, S.D. Bull, F. Marken, Electrochim. Acta 160, 195 (2015)
Gaussian 09, Revision A.02, M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, Ö. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox. (Gaussian, Inc., Wallingford CT, 2009)
Obtained by data base search and predictive approximation at www.ACDlabs.com. Accessed June 2015
C.Y. Cummings, J.D. Wadhawan, T. Nakabayashi, M. Haga, L. Rassaei, S.E.C. Dale, S. Bending, M. Pumera, S.C. Parker, F. Marken, J. Electroanal. Chem. 657, 196 (2011)
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 2033 kb)
Rights and permissions
About this article
Cite this article
Ahn, S.D., Kolodziej, A., Malpass-Evans, R. et al. Polymer of Intrinsic Microporosity Induces Host-Guest Substrate Selectivity in Heterogeneous 4-Benzoyloxy-TEMPO-Catalysed Alcohol Oxidations. Electrocatalysis 7, 70–78 (2016). https://doi.org/10.1007/s12678-015-0284-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-015-0284-8