Abstract
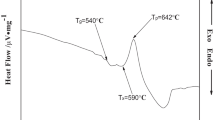
Glass comprising of P2O5–Al2O3–Na2O–ZnO is melted in microwave (MW) heating as an alternate energy efficient heating method. Properties of glasses obtained from MW heating are compared with that of glasses prepared adopting resistive heating. Glass transition temperature (Tg) in MW melted glass is found ~ 15–20 °C lower compared to the glass prepared in resistance furnace. XPS O1s spectra indicate less non-bridging oxygen (NBO) formation in glass obtained from MW melting. This may be due to less evaporation as well as less leaching of alumina from crucible wall during melting. Photoluminescence spectra of Eu3+-doped glass indicate higher asymmetric ratio in the conventional glass. MW melting requires 2 h 20 min, whereas it is ~ 6 h in resistive heating. Comparison of power consumption analysis depicts maximum MW forward power < 1.5 kW with ~ 1 kW average power during melting of glass. Maximum power in resistive heating furnace is recorded 4 kW for identical melting.










Similar content being viewed by others
References
Clark D E, Sutton W H, Microwave processing of materials, Annu. Rev. Mater. Sci. 26 (1996) 299.
Rao K J, Vaidhyanathan B, Ganguli M, and Ramakrishnan P A, Chem. Mater., 11 (1999) 882.
Bhattacharya M, and Basak T, Energy. 97 (2016) 306.
Agrawal D, Trans. Indian Ceram. Soc. 65 (2006) 129.
Fujii T., Kashimura K, and Tanaka H, Hazard. Mater. 369 (2019) 318.
Dube C L, Kashyap S C, Dube D C, and Agrawal D K, J. Alloys Compd. 571 (2013) 75.
Reinosa J J, García-Baños B, Catalá-Civera J M, López-Buendía Á M, Guaita L, and Fernández J F, Int. J. Appl. Glass Sci. 10 (2019) 208.
Singhal C, Murtaza Q, Alam P, and Hasan F, Adv. Mater. Process. Technol. 5 (2019) 559.
Davis III C, Pertuit A L, and Nino J C, J Am. Ceram. Soc. 100 (2017) 765.
Ahmad S, Mahmoud M M, and Hans Jürgen Seifert H, J. Alloys Compd. 797 (2019) 45.
Mahmoud M M, Folz D C, Suchicital C T A, and Clark D E, J. Am. Ceram. Soc. 95 (2012) 579.
Venkateswaran C, Sharma S C, Chauhan V S, and Vaish R, J. Am. Ceram. Soc. 101(1) (2018) 140.
García-Baños B, Reinosa J J, Peñaranda-Foix F L, Fernández J F, and Catalá-Civera J M, Sci. Rep. 9 (2009) 10809.
Siligardi C, D’Arrigo M C, Leonelli C, Pellacani G C, and Cross T E, J. Am. Chem. Soc. 83 (2000) 1001.
Chenu S, Rocherullè J, Lebullenger R, Merdrignac O, Chevire F, Tessier F, and Oudadesse H, J. Non-Cryst. Solids 356 (2010) 87.
Ghussn L and Martinelli J L, J. Mater. Sci. 39 (2004) 1371.
Wang J S, Jeng J S and Ni C T, J. Non. Cryst. Solids. 355 (2009) 780.
Almeida F J M, Martinelli J R, and Partiti C S M, J. Non. Cryst. Solids. 353 (2007) 4783.
Murase I, Imaeda K, Sakurai M and Watanabe M, Phosphorus Res. Bull. 19 (2005) 65.
Chenu S, Rocherullé J, Lebullenger R,. Merdrignac O, Cheviré F, Tessier F, and Oudadesse H, J. Non. Cryst. Solids. 356 (2010) 87.
Mandal A K, and Sen R, Mater. Manuf. Processes. 32 (2017) 1.
Mandal A K, Sen S, Mandal S, Guha C and Sen R, Int. J. Green Energy. 12 (2015) 1280.
Mandal A K, Sinha P K, Das D, Guha C, and Sen R, Mater. Res. Bull. 63 (2015) 141.
Mandal A K, Mandal B, Illath K, Ajithkumar T G, Halder A, Sinha P K, and Sen R, Sci. Rep. 8 (2018) 1.
Mandal A K, and Sen R, Int. J. Appl. Glass Sci. 10 (2019) 83.
Mandal A K, Balaji S, and Sen R, J. Alloys Compd. 615 (2014) 283.
Reddy B N K, Raju B D, Thyagarajan K, Ramanaiah R, Jho Y D and Reddy B S, Ceram. Int. 43 (2017) 8886.
Viswanath C S D, Krishnaiah K V, and Jayasankar C K, Opt. Mater. 83 (2018) 348.
Shirbeeny W E, Aly M H, El-samahy A E and Emad K M, Indian J Pure Appl. Phy. 3 (2007) 122.
Mandal A K, Agrawal D, and Sen R, J. Non-Cryst. Solids 371–372 (2013) 41.
Basak A, Ramrakhiani L, Ghosh S, Mandal A K, and Sen R, J. Non-Cryst. Solids 500 (2018) 11.
Elisa M, Grigorescu C E A, Vasiliu C, Bulinski M, Kuncser V, Predoi D, Filoti G, Meghea A, Lftimie N, and Giurgincal M, Rev. Adv. Mater. Sci. 10 (2005) 367.
Mandal A K, and Sen R, Mater. Res. Bull. 108 (2018) 156.
Pei Z, Su Q, and Li S, J. Lumin. 50 (1991) 123.
Acknowledgements
Authors would like to thank Director of CSIR-Central Glass and Ceramic Research Institute, for his kind encouragement and support to pursue this work. This work is supported by CSIR, FBR Project (MLP 0106/OLP0599).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mandal, A.K. Microwave and Conventional Preparation of P2O5–ZnO–Al2O3–Na2O Glass/Eu3+ Ion as Luminescent Probe. Trans Indian Inst Met 74, 827–837 (2021). https://doi.org/10.1007/s12666-020-02163-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-020-02163-9